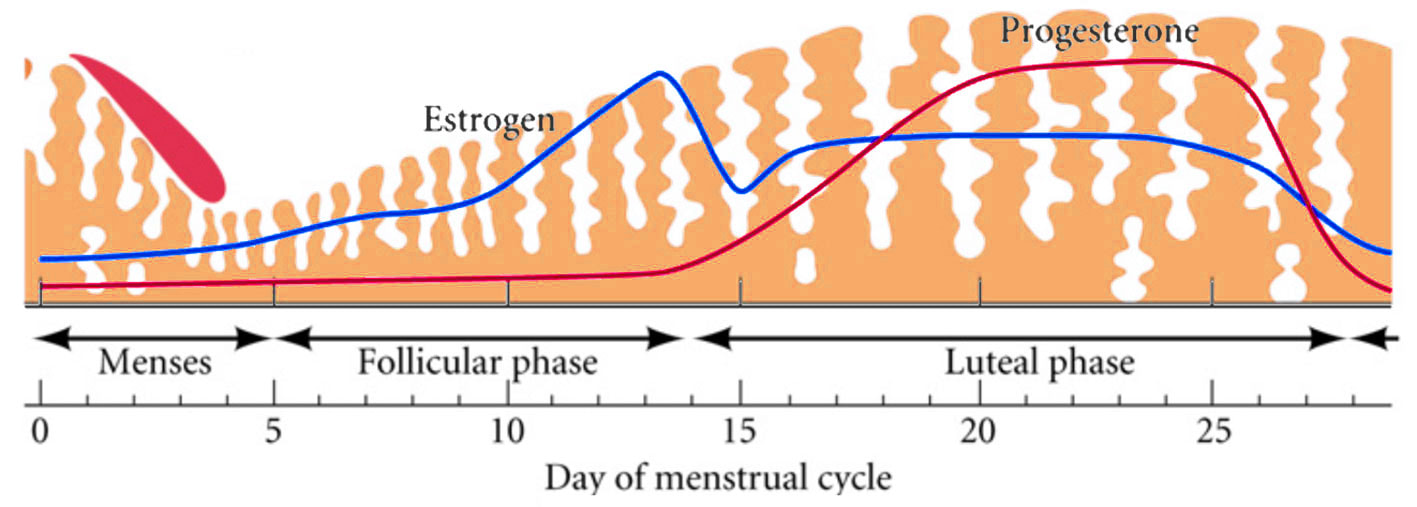
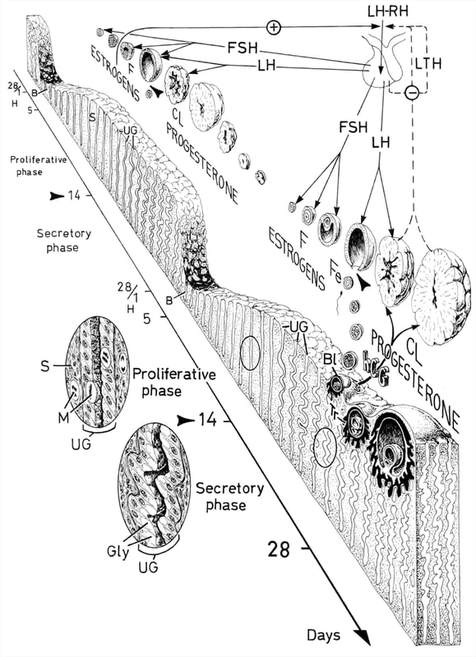
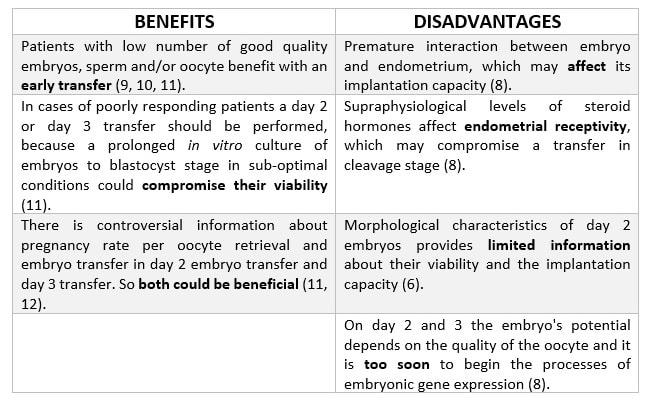
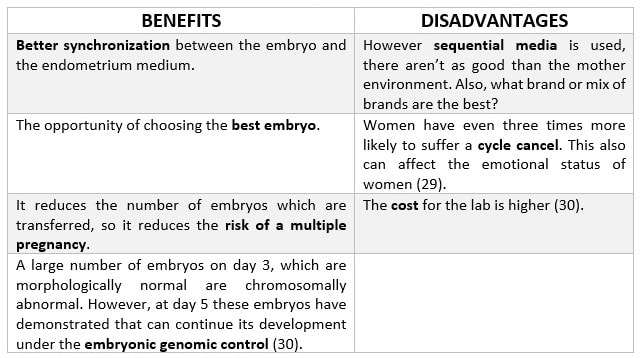
|
"The underlying cause for endometriosis is likely to present a multifactorial origin" Authors: Javier del Río, Noemi Díaz & Edel Rocher Endometriosis is defined as the presence of endometrial tissue outside the uterus, which induces a chronic inflammatory reaction. It is generally assumed that lesions arise through retrograde endometrial tissue loss during menstruation, coelomic metaplasia and lymphatic spread in immunologically and genetically susceptible individuals. Associated symptoms usually have an impact on the general physical, mental and social well-being (2,3,4). This condition is found in women from all ethnic and social groups, predominantly in those of reproductive age, which may represent an estimation of up to 176 million women all over the world. The prevalence of endometriotic disease seems to be around 5%, normally reaching a peak somewhere between ages 25 and 35. Among women aged 15-49, an annual incidence of endometriosis of 0.1% has been reported (2,3,4). Which symptoms may be indicative of the disease? Women who suffer from endometriosis typically experiment abdominal pain and related symptoms such as dysmenorrhoea, dyspareunia, heavy menstrual bleeding, non-menstrual pelvic pain, painful ovulation, dyschezia and dysuria, as well as chronic fatigue. Endometriotic lesions are followed by denervation and reinnervation, which represent accompanying changes in the central nervous system (central sensitization), thus generating a chronic pain syndrome. The extent of the disease varies from a few, small lesions on otherwise normal pelvic organs to large, ovarian endometriotic cysts (endometriomas) and/or extensive fibrosis and adhesion formation, causing marked distortion of the pelvic anatomy (2,3). As a consequence, establishing the diagnosis of endometriosis on the basis of symptoms alone may result difficult, not only due to their high degree of variation, but also because there is so much overlapping with other conditions such as irritable bowel syndrome or pelvic inflammatory disease. Therefore, it is common to come across a delay of several years between the onset of symptoms and a definitive diagnosis. In this respect, the revised scoring system of the American Society for Reproductive Medicine is normally employed to determine the disease stage (ranging from I, indicating minimal disease, to IV, indicating severe disease). This system is based on the type, location and appearance of the lesions, as well as the depth of the invasion caused and the extent of disease and adhesions (2,5,6). However, endometriosis symptoms do not always correspond to observations from laparoscopic exploration. The severity of these symptoms increase with age, along with the probability of a proper diagnosis. Moreover, the incidence of endometriosis peaks in women in their 40s (7). Which are the possible causes of the disease? Even though the underlying cause for endometriosis remains uncertain, it is likely to present a multifactorial origin. This includes inflammation enhanced by genetic causes, which may be epigenetically regulated, as well as triggered by exposure to environmental factors (3). So, it seems evident why pathophysiology of endometriosis keeps being a subject of debate. It remains unclear whether endometrial and endometriotic cells are genetically different, or whether such differences are the consequence of distinct environments or due to preexisting immunological defects (8). However, several hypotheses have been proposed to explain the origin of the disease:
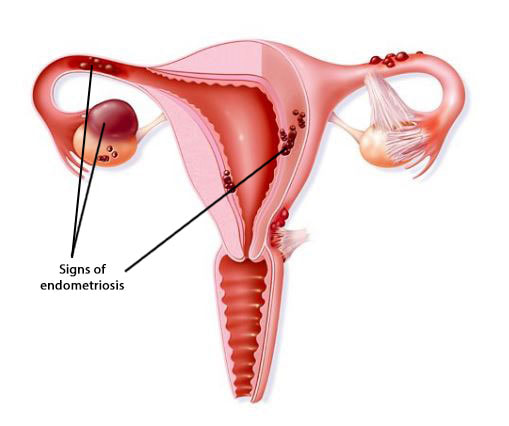
This was the first hypothesis proposed, put forth as the main cause for the pathogenesis of endometriosis. Retrograde menstruation appears when viable endometrial fragments are driven through the Fallopian tubes, possibly by a pressure gradient originating from dys-synergic uterine contractions. Once these fragments reach the peritoneal cavity they can implant, grow and invade pelvic structures. The likelihood of this event is influenced epidemiologically by any menstrual, reproductive or personal factor that may augment pelvic contamination by regurgitated endometrium (Figure 1). Further support for this aetiology is derived from studies of obstructed or compromised outflow tracts (9,10,11). However, this event occurs in 76–90% of women with patent Fallopian tubes, and not all of them suffer from endometriosis (11).
The monthly regeneration of the endometrium after menstrual shedding and re-ephitelialization after parturition or surgical curettage supports the existence of a stem cell pool. The involvement of these cells in the formation of endometriotic deposits could be the result of abnormal translocation of the normal stratum basalis of the endometrium via retrograde menstruation. Some group of cells may deposit and survive in the peritoneal cavity after retrograde flow, and may reactivate during adolescence in response to ovarian hormones. However, there are currently no data on the amount of endometrial stem/progenitor cells in neonatal period compared to the adult endometrium (11).
This theory postulates that endometriosis originates from the metaplasia of specialised cells present in the mesothelial lining of the visceral and abdominal peritoneum. Hormonal or immunological factors are thought to stimulate the transformation of normal peritoneal tissue/cells into endometrium-like tissue. The theory may explain the occurrence of endometriosis in pre-pubertal girls. However, the usual driving force for endometrial growth, oestrogen, is not present in pre-pubertal girls, and therefore this condition may be different from the endometriosis found in women of reproductive age. Nevertheless, this theory is imperfect due to endometriotic lesions being found in areas outside the course of the Müllerian duct (4,11).
Genetic factors represent about half of the variation in endometriosis risk, with an estimate of heritability of 51%. Meta-analyses of the few genome-wide studies performed in the past few years have provided evidence of a robust association of endometriosis with seven risk loci. Among them, WNT4, CDKN2B-AS1 and GREB1 are of particular interest due to their gene-based ranking, known pathophysiology and proximity to SNPs of genome-wide significance. Therefore, these genes represent strong candidates for further studies on endometriosis (4,12,13). Inflammation is an additional typical feature of endometriosis, since the presence of ectopic tissue in the peritoneal cavity is associated with overproduction of prostaglandins, cytokines and chemokines. Macrophages infiltrating the ectopic lesions express typical markers of alternative activation, favouring the growth of the lesions and promoting associated angiogenesis. Overproduction of reactive oxygen species (ROS) is also accompanied by a decreased level of antioxidants that usually eliminates these molecules. Thus, the resulting accumulation of ROS may contribute to the propagation and maintenance of endometriosis and associated symptoms (4,10,11). Oestrogens fuel ectopic endometrium growth, and alterations in oestrogen signalling have been associated with the disease. In fact, it is known that oestradiol produced both in the steroidogenic organs and locally in the endometriotic implants through aromatase expression promote the growth of endometrial tissue ectopically (4,11). This ectopic tissue has been consistently shown to express different levels of oestrogen receptors (ER) α and β than eutopic tissue, being ERβ highly present in ectopic tissue (4). Deficient methylation of the promoter of the ERβ-encoding gene has been suggested to result in pathological overexpression of ERβ in endometriosis, which in turn suppresses ERα expression and diminishes oestradiol-mediated induction of the progesterone receptor in endometriotic cells. This mechanism is thought to contribute to resistance to selective actions of progesterone in these cells, which is manifested by perturbations in a number of downstream progesterone target genes (4,11). Progesterone normally triggers a uterine endometrial response characterized by inhibition of oestrogen-dependent proliferation of epithelial cells, secretory maturation of the glands, and transformation of stromal cells into specialized decidual cells. Moreover, progesterone transiently induces a receptive phenotype in endometrial epithelial cells essential for embryo implantation [you can read more about the role of these hormones in the endometrium in our previous publication here]. As a consequence of progesterone resistance, genes critical to these events, such as prolactin for decidual response or glycodelin for embryo implantation, are dysregulated in the endometrium of affected women. Endometriosis-derived inflammation, in turn, could induce progesterone resistance by altering the signalling pathway of this hormone through mechanisms of competition or interference with proinflammatory transcriptional factors (4,11,14,15,16). The observation that autoimmune diseases tend to be more common in women with endometriosis supports the hypothesis that pathogenesis of this condition may involve a defective immune response in these patients. Women suffering from endometriosis present higher concentration of activated macrophages, decreased cellular immunity and repressed NK cell function (11). Endometriosis and infertility The relationship between endometriosis and infertility has been debated for many years. Infertile women are 6-8 times more likely to suffer from endometriosis than fertile women (7). Despite extensive research no agreement has been reached, and several mechanisms have been proposed to explain the association between endometriosis and infertility. These mechanisms include distorted pelvic anatomy, endocrine and ovulatory abnormalities, altered peritoneal function and altered hormonal and cell-mediated functions in the endometrium. Major pelvic adhesions or peritubal adhesions disturbing the tube-ovarian liaison and tube patency can impair the release of oocytes from the ovary, inhibit oocyte pickup, or impede the transport of the ovum itself (7). Women with endometriosis may exhibit endocrine and ovulatory disorders, including luteinized unruptured follicle (LUF) syndrome, impaired folliculogenesis, luteal phase defect, and premature or multiple LH surges. Moreover, a complex network of humoral and cellular immunity factors modulates the growth and inflammatory behaviour of ectopic endometrial implants, and so it may have adverse effects on the function of the oocyte, sperm, embryo, or Fallopian tubes (7,16). Management of endometriosis by assisted reproductive technologies There is currently no clear answer to the question of whether endometriosis has a negative impact on IVF outcomes. Several studies have previously suggested poorer outcomes in comparison to control cases, whereas other have shown no significant differences (17). Clinical outcomes for different stages of endometriosis Patients with endometriosis can present different levels of severity, which have a strong influence in the outcomes. Thus, it would be common to find a case of disease at a more severe phase with worse results than a different one presenting less extensive endometriosis (18,19). In stage III/IV endometriosis the prognosis for IVF/ICSI treatments is lower compared to milder stages I/II. (19). In their retrospective study, Opøien et al. highlighted the fact that patients with a more severe form of the disease had fewer oocytes retrieved, despite the higher gonadotropin doses these women had been administered (20). This may be due to endometriosis affecting both oestrogen and progesterone mRNA receptors in granulosa cells (21). Likewise, patients suffering from stage III/IV endometriosis have been reported to show a decrease in implantation and clinical pregnancy rates (22). Endocrinology of endometriosis Measurements of circulating hormone concentrations have shown statistically higher levels of hMG/FSH in endometriosis patients than in control patients. However, both high-quality embryo and clinical pregnancy rates were found to be lower in such patients (23). Similarly, the high expression of PR-A and ER-α in patients with endometriosis might be a leading cause of ovarian dysfunction due to this condition. This means that stages III-IV patients required higher gonadotropin stimulation doses to prevent ovarian dysfunction (24). Endometriomas Endometriomas represent an additional variable that may also affect ART outcomes. They have been reported to affect 17-44% of patients with endometriosis, who exhibit reduced ovarian response due to the low response to gonadotropin stimulation (25). Such response has been widely discussed by different authors. Conclusions from these reports suggest a reduced number of retrieved oocytes in women with bilateral endometriomas, as well as the rates of top-quality embryos, implantation, clinical pregnancy and live birth, when compared to control groups (26,27,28). Gonadotropin stimulation for IVF in the endometriosis context Patients with stage III/IV endometriosis normally undergo stimulation by higher doses of gonadotropin, which consequently results in higher circulating oestradiol levels (24). D'Hooghe and coworkers performed an analysis of this kind of patients and demonstrated that the cumulative endometriosis recurrence rate (CERR) was lower after ovarian hyperstimulation for IVF than after lower-dose ovarian stimulation for IUI. This suggests that temporary exposure to high oestradiol levels during ovarian hyperstimulation for IVF is not a risk factor for endometriosis recurrence in women subjected to ART (29). Previous reports from Benaglia et al. have shown that after 3-6 months of IVF cycles 77% of patients experimented an improvement of the condition, whereas about 11% reported worsening. Also, it should be noted that endometrioma size also remained stable after IVF cycles, which suggests they are not affected by gonadotropin stimulation (30). IVF outcomes after medical therapy for endometriosis Medical interventions have been demonstrated to have benefit in alleviating, if not eliminating, symptoms associated with endometriosis (31). The first thing to control is the prolonged use of GnRHa prior to initiation of gonadotropin stimulation for ART. The extension of GnRHa use up to about 3 months has been reported to exhibit higher implantation rates and significantly higher clinical pregnancy rates (32). This is due to the fact that endometriosis patients were more likely to present aberrant endometrial expression of β3 integrin, and that a 3-month course of GnRHa allowed to rescue up to 64% of the expression (33). Another approach employed to increase integrin expression is the of danazol as a post-treatment after the ovulatory cycle. This has been reported to result in both similar clinical pregnancy and live birth rates to those from patients with normal integrin expression (34). A different medical therapy is the administration of oral contraceptives in women with endometriosis before the initiation of the actual ART treatment. This approach seems to improve clinical outcomes, keeping them comparable to women of similar age without endometriosis. In contrast, ART outcomes are markedly compromised in endometriosis patients who are not pretreated with this method. It is worth to mention that ovarian responsiveness to stimulation was not altered by 6-8 weeks use of oral contraceptives prior to initiation of ART treatment, including poor responders with endometriomas (35). So far, it has not been possible to establish the optimal duration of medical therapy for endometriosis by means of comparative trials. As a consequence, ideal patients who could benefit from medical intervention have not yet been depicted. In spite of this, it would be reasonable to think that those patients suffering from more severe stages of the disease and/or with prior implantation failure might be the best candidates (36). Infertility in women affected by endometriosis may be related to alterations in follicles, poor oocyte quality (...), or even decreased endometrial receptivity. Impact of surgical management of endometriosis on IVF outcomes Previous reports have shown that pre-cycle surgical intervention may result beneficial for the final outcome. Opøien et al. demonstrated that patients with stage I/II endometriosis treated with surgical resection presented higher clinical pregnancy rates than those who had only been subjected to diagnostic laparoscopy before IVF/ICSI (37). Similar data were obtained when evaluating results from a group of 825 endometriosis patients; those treated with surgical resection presented overall significantly higher pregnancy and IVF rates when compared to those who had been subjected to IVF alone, and also to those with no treatment whatsoever (38). Implantation and pregnancy rates have also been reported to increase after resection in patients with deeply invasive endometriosis, although this group needed higher gonadotropin doses for stimulation, and yet fewer oocytes were retrieved (39). However, it is important to keep in mind that research is limited by a variety of factors, such as variations in surgical techniques (i.e., ablation vs. resection), degree of removal of the endometriotic tissue or differences in IVF laboratories (40). Interestingly, previous reports indicate that IVF implantation rates are not affected by the time intervals between surgical interventions for resection of endometriosis in the absence of endometriomas (41). On the other hand, certain studies have reported the negative impact of surgical management of endometriomas on IVF treatments, like the inability to access follicles at oocyte retrieval following precycle resection of endometriomas, or the harmful effect on oocytes after exposure to endometrioma fluid. Nevertheless, most authors refuse these arguments (42), and few studies assert the benefits of surgery in endometriomas larger than 3 cm in order to treat painful symptoms or to facilitate access to the ovary (43). Current data from clinical outcomes suggest that, under controlled circumstances, IVF cycles are not compromised by the presence of endometriosis. The exception to this finding is the fact that patients with ovarian endometriomas showed lower response to gonadotropin stimulation (17). Thorough meta-analyses through early studies revealed lower number of oocytes obtained after egg collection, as well as decreasing fertilization, implantation and pregnancy rates in patients affected by endometriosis after ART treatments (18). However, it is important to note that pregnancy rates were extremely low in those years. Effects of endometriosis on ART outcomes Since endometriosis patients have sometimes been studied simultaneously to other groups of patients such as women suffering from Fallopian tube-related infertility, it is not clear whether endometriosis actually affects implantation rates. Barcelo and coauthors’ explanation is the similar percentage of meiotic abnormalities in in vitro matured oocytes between endometriosis patients and the control group after ovarian stimulation (44). An additional factor that might contribute to confusion is the presence of adenomyosis, which is frequently found in endometriosis patients and could have a deleterious impact on the implantation process (45). Altered ovulation and oocyte production, as well as increased inflammatory cells in the peritoneal fluid of endometriomas have been observed in endometriosis patients (46). In fact, infertility in women affected by endometriosis may be related to alterations on follicles, poor oocyte quality and related failures during subsequent embryogenesis, or even decreased endometrial receptivity. Such hypothesis is supported by the altered progesterone and cytokine concentrations found in the follicular fluid from these patients (44). Embryos derived from affected women are likely to develop more slowly, compared to embryos derived from women suffering from tubal disease (45). It is worth to mention that women with moderate to severe endometriosis who receive oocytes from healthy donors seem to present normal endometrial receptivity and pregnancy rates. Conversely, when donor oocytes from endometriosis-affected women are transferred into healthy women, implantation rates are lower and embryo quality is reduced (46). Further studies are needed, though, in order to determine pregnancy rates from donors in different conditions and disease stages (16). Delayed histological maturation or biochemical disturbances may lead to endometrial dysfunction, too. This is concluded from reports that show uterine implantation being affected by changes in receptivity on an endometriosis background (17). Endometrial expression of the adhesion molecule αvβ integrin has been observed to be reduced during the time of implantation in some endometriosis-affected women. Additionally, the synthesis of the endometrial ligand for L-selectin has also been observed to be affected in some patients (16). L-selectin is a trophoblast surface-coating protein, which may explain the altered implantation rates observed in such cases (47). The observation of luteal phase disruption associated to endometriosis may be due to dysregulation of the progesterone receptor, as well as an effect on progesterone target genes that consequently results in a decrease in endometrial receptivity (17,47). Sperm quality and/or function is also reduced, which has been proposed to be due to inflammatory/toxic effects of the peritoneal fluid, along with a higher amount of activated macrophages. Not only are these effects harmful to oocytes and sperm, but toxic to the embryo (17). Treatment of endometriosis-associated infertility IVF is currently the most effective treatment for endometriosis-associated infertility (17). However, comparison of data on the effectiveness of IVF for endometriosis patients vs. patients suffering from other causes of infertility is not without controversy. Reports from the Society of Assisted Reproductive Technology (SART) have shown that the average delivery rate per retrieval for patients undergoing IVF-ET is 39.1% for endometriosis-affected women, as opposed to 33.2% for women with other causes of infertility (48). Pre-treatment ovulation suppression is a possibility to be considered in order to enhance suppression of inflammatory cytokines, as well as to reduce the presence of disease signs prior to any form of ART. Nevertheless, further research will be required on patients with endometriomas to assess their effect on IVF/ICSI, and to elucidate whether pre-ART surgical intervention may increase success rates (48). Potential treatments in the future There is currently no consensus on whether performing surgery prior to undergoing ART is fundamental for achieving pregnancy. Either way, there is a priority in terms of age in endometriosis cases (17). There are, however, some novel medical therapies, such as immunoconjugate (ICON) and aromatase inhibitors. ICON targets aberrantly expressed tissue factor on endometriotic endothelium, causing regression of the established disease (most likely by devascularization), which seems to improve fertility rates (17,49). Aromatase inhibitors, absent under normal circumstances, are found to be present in ectopic endometrial tissue; this may have a direct impact on oestradiol levels and implantation rates in endometriosis patients (50,51). Current treatment of endometriosis-associated infertility focuses on improving fecundity by removing or reducing ectopic endometrial implants, thus restoring normal pelvic anatomy (47). There are several possibilities that practitioners can adopt when facing this kind of events, depending on the particular case and the patient: expectant management, medical treatment and/or surgical treatment (48,52). The reality is that the optimal method of choice to treat endometriosis-associated infertility is an individualized decision that should be made on the basis of the specificity of the patient (18,50). CONCLUSIONS Despite lower ovarian response, reduced embryo quality and impaired implantation in moderate/severe cases, endometriosis patients have been able to obtain IVF/ICSI success rates similar to those with tubal factor-related infertility. As it has been widely explained, the combination of aggressive but controlled ovarian hyperstimulation, appropriate hypophysis suppression and efficient surgery before initiating cycles seemed to be crucial and significantly efficient for IVF/ICSI success on patients suffering from endometriosis (50,52). Within the last few years, scientific knowledge have made it possible to develop certain clinical improvements that have surely opened new possibilities for endometriosis patients. REFERENCES 1. Available from: https://www.womensmd.org/wp-content/uploads/2017/06/Endometriosis-causing-heavy-periods-with-blood-clots.jpg [Cited November 4 2017].
2. Kennedy S, Bergqvist A, Chapron C, d’Hooghe T, Dunselman G, Greb R, et al. ESHRE guideline for the diagnosis and treatment of endometriosis. Hum Reprod. 2005;20:2698–704. 3. Johnson NP, Hummelshoj L, World Endometriosis Society Montpellier Consortium. Consensus on current management of endometriosis. Hum Reprod. 2013;28:1552–68. 4. Vercellini P, Viganò P, Somigliana E, Fedele L. Endometriosis: pathogenesis and treatment. Nat Rev Endocrinol. 2014;10(5):261–75. 5. Giudice L. Endometriosis. N Engl J Med. 2010;362:2389–98. 6. Practice Committee of the American Society for Reproductive Medicine. Endometriosis and Infertility. Fertil Steril. 2006;14:S156–60. 7. Bulletti C, Coccia ME, Battistoni S, Borini A. Endometriosis and infertility. J Assist Reprod Genet. 2010; 27:441–7. 8. Koninckx P, Ussia A, Adamyan L, Wattiez A, Donnez J. Deep endometriosis: definition, diagnosis, and treatment. Fertil Steril. 2012;98(3):564-571. 9. Burney R, Giudice L. Pathogenesis and pathophysiology of endometriosis. Fertil Steril. 2012;98(3):511-519. 10. Lebovic D, Mueller M, Taylor R. Immunobiology of endometriosis. Fertil Steril. 2001;75(1):1-10. 11. Sourial S, Tempest N, Hapangama D. Theories on the Pathogenesis of Endometriosis. Int J Reprod Med. 2014; 179515.1-9. 12. Nyholt DR, Low SK, Anderson CA, Painter JN, Uno S, Morris AP et al. Genome-wide association meta-analysis identifies new endometriosis risk loci. Nat Genet. 2012;44(12):1355–1359. 13. Pagliardini L, Gentilini D, Vigano’ P, Panina-Bordignon P, Busacca M, Candiani M et al. An Italian association study and meta-analysis with previous GWAS confirm WNT4, CDKN2BAS and FN1 as the first identified susceptibility loci for endometriosis. J Med Genet. 2013;50:43–46. 14. Aghajanova L, Hamilton A, Kwintkiewicz J, Vo KC, Giudice LC. Steroidogenic enzyme and key decidualization marker dysregulation in endometrial stromal cells from women with versus without endometriosis. Biol Reprod. 2009;80:105 –114. 15. Wei Q, St Clair JB, Fu T, Stratton P, Nieman LK. Reduced expression of biomarkers associated with the implantation window in women with endometriosis. Fertil Steril. 2009;91: 1686–91. 16. Practice Committee of the American Society for Reproductive Medicine. Endometriosis and infertility: a committee opinion. Fertil Steril. 2012; 98: 591-598. 17. Surrey ES. Endometriosis and assisted reproductive technologies: maximizing outcomes. Semin Reprod Med. 2013;31:154–63. 18. Barnhart K, Dunsmoor-Su R, Coutifaris C. Effect of endometriosis on in vitro fertilization. Fertil Steril. 2002;77:1148 –1155. 19. Kuivasaari P, Hippelainen M, Anttila M, Heinonen S. Effect of endometriosis on IVF/ICSI outcome: stage III/IV endometriosis worsens cumulative pregnancy and live-born rates. Hum Reprod. 2005;20:3130–3135. 20. Opøien HK, Fedorcsak P, Omland AK, Abyholm T, Bjercke S, Ertzeid G, et al. In vitro fertilization is a successful treatment in endometriosis-associated infertility. Fertil Steril. 2012;97:912–8. 21. Karita M, Yamashita Y, Hayashi A, Yoshida Y, Hayashi M, Yamamoto H et al. Does advanced-stage endometriosis affect the gene expression of estrogen and progesterone receptors in granulosa cells? Fertil Steril. 2011;95:889 – 894. 22. Harb H, Gallos I, Chu J, Harb M, Coomarasamy A. The effect of endometriosis on in vitro fertilisation outcome: a systematic review and meta-analysis. BJOG. 2013;120:1308–20. 23. Revelli A, Pettinau G, Basso G, Carosso A, Ferrero A, Dallan C et al. Controlled Ovarian Stimulation with recombinant-FSH plus recombinant-LH vs. human Menopausal Gonadotropin based on the number of retrieved oocytes: results from a routine clinical practice in a real-life population. Reprod Biol Endocrinol. 2015;13:77. 24. Tan CW, Lee IH, Choolani M, Tan HH, Griffith L, Chan J. Endometriosis, endometrium, implantation and fallopian tube. Hum Reprod. 2013;28:206-226. 25. Somigliana E, Infantino M, Benedetti F, Arnoldi M, Calanna G, Ragni G. The presence of ovarian endometriomas is associated with a reduced responsiveness to gonadotropins. Fertil Steril. 2006;86:192–196. 26. Benaglia L, Bermejo A, Somiglianaetal E. Invitrofertilization outcome in women with unoperated bilateral endometriomas. Fertil Steril. 2013;99:1714–1719. 27. Suzuki T, Izumi SI, Matsubayashi H, Awaji H, Yoshikata K, Makino T. Impact of ovarian endometrioma on oocytes and pregnancy outcome in in vitro fertilization. Fertil Steril. 2005;83:908–913. 28. Almog B, Shehata F, Sheizaf B, Tan SL, Tulandi T. Effects of ovarian endometrioma on the number of oocytes retrieved for in vitro fertilization. Fertil Steril. 2011;95:525–527. 29. Hooghe MD, Denys B, Spiessens C, Meuleman C, Debrock S. Is the endometriosis recurrence rate increased after ovarian hyperstimulation? Fertil Steril. 2006;86:283–290. 30. Benaglia L, Somigliana E, Santi G, Scarduelli C, Ragni G, Fedele L. IVF and endometriosis-related symptom progression: insights from a prospective study. Hum Reprod. 2011;26:2368–2372. 31. Mehedintu C, Plotogea M, Ionescu S, Antonovici M. Endometriosis still a challenge. J Med Life. 2014;7:349-57. 32. Surrey ES, Silverberg KM, Surrey MW, Schoolcraft WB. Effect of prolonged gonadotropin-releasing hormone agonist therapy on the outcome of in vitro fertilization-embryo transfer in patients with endometriosis. Fertil Steril. 2002;78:699–704. 33. Lessey BA. Medical management of endometriosis and infertility. Fertil Steril. 2000;73:1089–1096. 34. Tei C, Maruyama T, Kuji N, Miyazaki T, Mikami M, Yoshimura Y. Reduced expression of alphavbeta3 integrin in the endometrium of unexplained infertility patients with recurrent IVF-ET failures: improvement by danazol treatment. J Assist Reprod Genet. 2003;20:13-20. 35. Ziegler D, Gayet V, Aubriotetal FX. Useoforalcontraceptives in women with endometriosis before assisted reproduction treatment improves outcomes. Fertil Steril. 2010;94:2796–2799. 36. Furness S, Yap C, Farquhar C, Cheong Y. Pre and post-operative medical therapy for endometriosis surgery. Cochrane Database Syst Rev. 2004:CD003678. 37. Opøien HK, Fedorcsak P, Byholm T, Tanbo T. Complete surgical removal of minimal and mild endometriosis improves outcome of subsequent IVF/ICSI treatment. Reprod Biomed Online. 2011;23:389 – 395 38. Barri PN, Coroleu B, Tur R, Barri-Soldevila PN, Rodríguez I. Endometriosis-associated infertility: surgery and IVF, a comprehensive therapeutic approach. Reprod Biomed Online. 2010;21:179–185. 39. Bianchi PH, Pereira RM, Zanatta A, Alegretti JR, Motta EL, Serafini PC. Extensive excision of deep infiltrative endometriosis before in vitro fertilization significantly improves pregnancy rates. J Minim Invasive Gynecol. 2009;16:174–180. 40. Surrey ES, Schoolcraft WB. Does surgical management of endometriosis within 6 months of an in vitro fertilization–embryo transfer cycle improve outcome? J Assist Reprod Genet. 2003;20:365–70. 41. Khamsi F, Yavas Y, Lacanna IC, Roberge S, Endman M, Wong JC. Exposure of human oocytes to endometrioma fluid does not alter fertilization or early embryo development. J Assist Reprod Genet. 2001;18:106-109. 42. Benschop L, Farquhar C, Van der Poel N, Heineman MJ. Interventions for women with endometrioma prior to assisted reproductive technology. Cochrane Database Syst Rev. 2010;11:CD008571. 43. Elter K, Oral E. Surgical treatment before assisted reproductive technologies. Semin Reprod Med. 2014; 32(04): 253-261. 44. Barcelos ID, Vieira RC, Ferreira EM, Martins WP, Ferriani RA, Navarro PA. Comparative analysis of the spindle and chromosome configurations of in vitro-matured oocytes from patients with endometriosis and from control subjects: a pilot study. Fertil Steril. 2009:92;1749–1752. 45. Salim R, Riris S, Saab W, Abramov B, Khadum I, Serhal P. Adenomyosis reduces pregnancy rates in infertile women undergoing IVF. Reprod BioMed Online. 2012;25:273– 277. 46. Dmowski WP, Pry M, Ding J, Rana N. Cycle specific and cumulative fecundity in patients with endometriosis who are undergoing controlled ovarian hyperstimulation-intrauterine insemination or in vitro fertilization-embryo transfer. Fertil Steril. 2002;78:750-756. 47. Macer ML, Taylor HS. Endometriosis and infertility: a review of the pathogenesis and treatment of endometriosis-associated infertility. Obstet Gynecol Clin North Am. 2012;39: 535-549. 48. Ruiz Flores FJ, Garcia-Velasco JA. Is there a benefit for surgery in endometrioma associated infertility?. Curr Opin Obstet Gynecol. 2012;24(3):13640. 49. Hadman M, Omar SZ, Dunselman G, Chenog Y. Influence of endometriosis on assisted reproductive technology outcomes: a systematic review and meta-analysis. Obstet Gynecol. 2015;125(1):79-88. 50. Wahd SA, Alaf SK, Al-Shawaf T, AlTawil NG. Ovarian reserve markers and assisted reproductive technique (ART) outcomes in women with advanced endometriosis. Reprod Biol Endocrinol. 2014;12:120. 51. Kawwass JF, Crawford S, Session DR, Kissin DM, Jamieson DJ. Endometriosis and Assisted Reproductive Technology: United States Trends and Outcomes: 2000–2011. National ART Surveillance System Group. Fertil Steril. 2015;103(6):1537-43. 52. D Xiyuan, L Xiuhua, Wang R, Zhang H. The impact of endometriosis on IVF/ICSI outcomes. Int J Clin Exp Pathol. 2013;6(9):1911-1918.
4 Comments
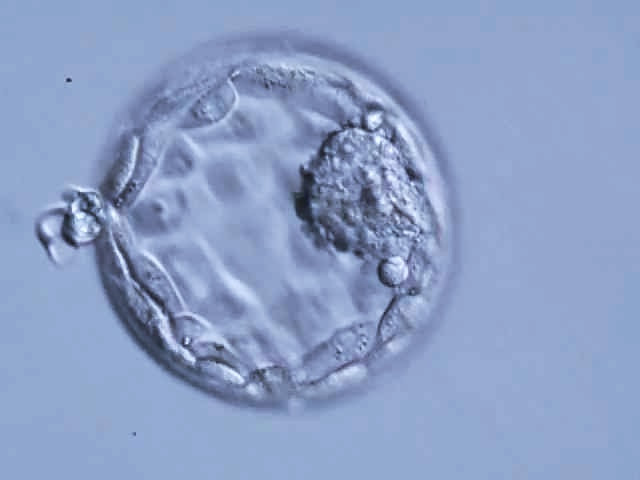
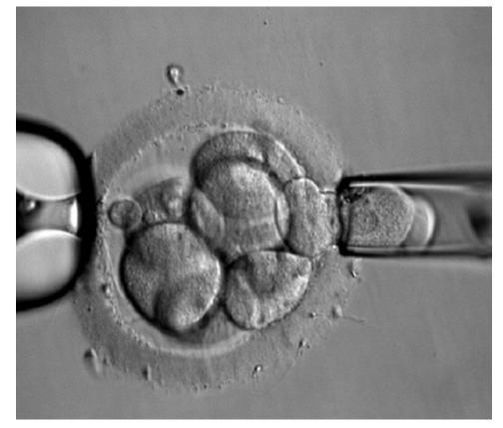
Authors: Iñaki Arroyos, Lidón Carretero Vilarroig & Belén Gómez Giménez INTRODUCTION Cryopreservation is a basic procedure in the daily work with assisted reproduction techniques. It is routinely and successfully applied to sperm samples, spare embryos from in vitro fertilization (IVF) cycles and oocytes. In the past, slow freezing procedures were carried out with no reliable clinical outcomes, especially in egg freezing programs. After the recent introduction of vitrification it is now possible to cryopreserve embryos and oocytes, and to assure patients that this process will not decrease the quality and the biological potential of the cells. At present, there is a growing trend towards a “vitrify-all” strategy after IVF with a single embryo transfer of a warmed embryo in a subsequent cycle (1, 2). There have been reports of improved implantation and pregnancy rates with frozen embryo transfers as compared to fresh autologous embryo transfer, suggesting superior endometrial receptivity in the absence of ovarian stimulation (2, 3). Oocyte cryopreservation The mature oocyte is the biggest cell in the body and contains a high proportion of water. The amount of water compromises the viability of the ovocyte during the slow freezing because ice crystals can be formed. The spindle of the mature oocyte is very sensitive to damage from ice crystal formation, and so it may easily become disrupted. Also, the oocyte is extremely vulnerable to mild chilling, which may severely compromise its integrity, and even cause complete degeneration. Innovation and improvement of vitrification was not easy in the beginning. It took a lot of research and experimentation on different protocols for the developers, as well as long time to set the right process. The procedure itself is complex, and it involves numerous critical steps for successful performance (4). Egg freezing is an increasing phenomenon because of its many advantages. It may provide an easier solution in cases of legal and/or ethical issues between the parents, such as divorce or decease. Freezing eggs rather than embryos can also avoid dealing with the complexities of having an increasing number of frozen embryos in nitrogen tanks unclaimed by the progenitors. Moreover, certain medical treatments to battle cancer can jeopardize the ovarian function, and so an egg freezing program seems suitable for such patients. Regarding social aspects, egg freezing allows women to postpone their motherhood. In addition, egg freezing also makes synchronization between donors and recipients much easier. Finally, this technique is suitable for patients who have low ovarian reserve and need PGD treatments: it allows patients to accumulate oocytes from several cycles, which increase the success of the treatments (5). Sperm cryopreservation This technique offers the possibility of freezing sperm to preserve fertility in those men subjected to treatments such as chemotherapy or radiotherapy, which may compromise gametogenesis. Additionally, sperm cryopreservation makes it possible to create sperm banks, which offer a valuable alternative to azoospermic patients willing to father children. Embryo cryopreservation Embryo freezing represents an essential method for any assisted reproduction centre, since it allows to save spare embryos from an IVF cycle and preserve them in liquid nitrogen for future uses. Ovarian Tissue Cryopreservation Ovarian tissue cryopreservation (OTC) represents another technique to preserve fertility in women. It has been carried out for more than 18 years now (6, 7) and it represents the main option available for women who require cancer treatment but cannot delay chemotherapy. Moreover, it is the only possibility for pre-pubertal patients (8). Currently, embryo and oocyte cryopreservation are the only fertility preservation techniques that are considered to be non-experimental by the American Society for Reproductive Medicine (9). Unfortunately, both approaches require previous preparation and stimulation with gonadotropins for oocyte retrieval, which usually requires 2-4 weeks or longer (10). Furthermore, oocyte harvest typically involves the use of transvaginal ultrasound and needle oocyte retrieval techniques, which could require general anaesthesia. This delay is often not possible or appropriate for females requiring urgent therapy or in breast cancer patients, because high estrogen levels might have detrimental effects on the primary tumor. Additionally, not all patients have partners with whom to create embryos for cryopreservation (10). Most clearly, these techniques are not indicated for young and pre-pubertal female patients due to the inability of their immature hypothalamic-pituitary axis to produce mature eggs (11) Under these circumstances, the possibility of cryopreservation of ovarian tissue (cortex) has become an urgent and highly-demanded technology for two types of young female cancer patients. First, those who must undergo advanced chemotherapy and/or radiotherapy. Second, those with non-oncological systemic diseases such as autoimmune or haematological conditions, that sometimes require chemotherapy, radiotherapy, or bone marrow transplantation (12). OTC does not require ovarian stimulation, and it allows to preserve gonadal function in pre-pubertal and adult patients. This procedure offers a promising option for women at high risk of premature ovarian failure and sterility (12). Furthermore, transplanting ovarian tissue not only restores fertility but also restores endocrine function (13). Ovarian cortical tissue contains the primordial follicles, which are located in the ovarian cortex, near the surface epithelium. These are the smallest female fertility unit, including 90% of the ovarian follicular reservoir. Although there are other types of follicles present in ovaries removed from the patient, primordial follicles are the ones that are considered for ovarian cryopreservation. Among other reasons, these are the most resistant follicles to cryoinjury due to of the small size of the oocytes, the reduced cytoplasmic content and the absence of meiotic spindle that could potentially be damaged (14). OVARIAN CORTEX CRYOPRESERVATION TECHNIQUE The most common steps to be followed for this approach are summarized below: 1. Biopsy. The removal of ovarian tissue is a relatively simple procedure. In order to ensure optimal penetration of cryoprotectants, approximately twelve small biopsies of ovarian cortical tissue (3x3x1 mm) are collected (15). This procedure can be performed by using minimally invasive techniques like laparoscopy, or even in conjunction with other open procedures such as primary tumor resection. Regardless of the technique, any area away from the hilum and lacking predominant follicles and luteal tissue is preferred for biopsy. The goal is to maximize the number of primordial follicles per specimen without compromising vascular supply to the remaining ovarian tissue. In order to optimise results, it is best to remove the ovarian cortex from the medulla, which helps extreme penetration of cryoprotectants into the cortical tissue (14). Normally, extraction of a single block of cortical tissue through partial excision technique can be performed in either one or both ovaries (11). 2. Histological examination. Ovarian fragments are immediately observed by a pathologist to simultaneously confirm the presence of follicles and the absence of a massive cancer infiltration (16). 3. Cryopreservation. Ovarian tissue can be cryopreserved independently of the menstrual phase, which represents no delay for the oncological therapy. Centres offering OTC allow for the procedure to be performed even the first day after the patient's first visit (17), and it can be carried out using one of two established techniques: slow freezing or rapid freezing (vitrification) (Fig. 3). The slow freezing method has already resulted in dozens of live births worldwide, whereas vitrified tissue has only led to a few reported live births to date (18, 19). The well-established standard method for human OTC is slow equilibrium freezing. It begins with exposing cells to low concentrations of one of the cryoprotective agents (CPAs) (propanediol, dimethyl sulfoxide or ethylene glycol) in combination with human serum albumin, and temperature must be slowly decreased. In contrast, vitrification requires higher concentrations of CPAs, which reduces the risk of ice nucleation, and it is significantly faster. When the solution is rapidly cooled, the tissue turns into a glassy, vitrified state, avoiding extracellular and intracellular ice crystallization (19). You can read our previous article to know more about these techniques here. The current main problem of ovarian banking is to restore ovarian function and fertility using frozen/thawed tissue with both high efficiency and minimal risks (16). 4. Biopsies are stored in the vapour phase of liquid nitrogen at -176 °C, thus allowing for long-term storage of potentially large numbers of primordial follicles (15). 5. Re-implantation. The stored ovarian tissue can be re-implanted orthotopically (into the remaining ovary, ovarian fossa, or broad ligament), heterotopically (into the subcutaneous space of the forearm, subcutaneous tissue of the abdomen, anterior wall of the abdomen, just beneath the peritoneum, or in the rectus muscle), or as recent studies have reported, xenografted or matured entirely in vitro (15, 19). Orthotopic transplantation may provide the possibility to achieve natural pregnancy; however, it requires abdominal surgery with general anaesthesia. In contrast, heterotopic is advantageous in cases of severe pelvic adhesions, distorted pelvic anatomy, and poor pelvic vasculature due to previous irradiation. Furthermore, it implies the possibility of creating long-term ovarian endocrine function with a less invasive surgical approach for transplantation, which does not require general anaesthesia, and thereby creating a more cost-effective option. However, this option may produce oocytes (and therefore, embryos) with reduced quality, as compared to orthotopic transplantation sites. This outcome is likely related to the suboptimal environment of heterotopic sites in regard to local factors. These include: external pressure, temperature, vascularization (decreased blood supply), oxygen tension and reduced paracrine factors, which could affect the possibility of sustaining the growth and development of normal follicles and oocytes (16). Thawed ovarian fragments do not need vascular anastomosis when transplanted into a well-vascularized tissue. Genes for angiogenesis factors seem to be upregulated in the ovary, compared to other tissues (16). However, there is a significant risk of the tissue suffering from hypoxia until neovascularization starts, normally following the first 48 hours after the procedure. Survival of primordial follicles (around 90% after thawing) ranges between 5 and 50% after grafting, the most crucial factor being the degree of ischaemic injury after transplantation (16). Although restoration of ovarian function has been reported for both approaches, live births following bilateral oophorectomy have only been documented from orthotopic transplantations (19). CASE REPORTS Successful studies in experimental animals with live births following transplantation of cryostored ovarian tissue have been previously reported in rodents and sheep (20, 21, 22, 23). Ovarian tissue cryopreservation was first described by Hovatta et al in 1996 (6). The first human ovarian transplantation with cryopreserved ovarian tissue was later performed by Oktay in 2000 (24). Donnez and colleagues reported the first human live birth from orthotropic transplantation of frozen human ovarian tissue in 2004 (25), with another successful live birth achieved by Meirow in 2005 (26). Live births following cryopreservation of pre-pubertal ovarian tissue had not been described until the 2015 case report by Demeestere et al (27). During all these years, authors have been discussing about whether vitrification is better than slow freezing. Some of them found apoptotic cells in vitrified ovarian tissue, meanwhile others showed higher primordial follicle density using the slow freezing method. Sanfilippo et al found no significant differences between both techniques (28). The American Society of Clinical Oncology advises that OTC for fertility preservation is still considered an experimental technique (9). Consequently, it should only be performed in centres with the appropriate expertise. This means performance under the Institutional Review Board (IRB) approved protocols, which include follow-up for recurrent cancer (29). However, in some clinical situations it remains the only available option. Here we describe some reported cases in different scenarios: fertility wishes and puberty induction, autotransplantation and allotransplantation. Fertility preservation OTC is the only available option for fertility preservation before menarche. Demesteree et al (2015) reported a spontaneous pregnancy case of a woman who had undergone ovarian tissue transplantation shortly before turning 14 (27). She had been diagnosed with sickle-cell anaemia at the age of 5 and treated with hematopoietic stem cell transplantation at 11. Her parents and herself were offered OTC in order to preserve fertility. The patient started puberty at the age of 10 (breast development) and the procedure was performed when she was 13 years and 11 months. At the age of 25 she interrupted hormonal supplementation and underwent ovarian tissue transplantation. Two years later she got pregnant and gave birth to a healthy boy in November 2014. Andersen 2012 et al described a case of ovarian tissue allotransplantation between identical twins at the age of 38 (30). One of them had undergone menopause at the age of 22, whereas the other had already had 2 children. The woman who was transferred the ovarian tissue had three natural pregnancies: eight months after transplantation, at age 42 and at 45. She gave birth to a healthy girl and two healthy boys. Puberty induction A.K. Jensen et al recommend OTC to all young girls who present a high risk of developing ovarian insufficiency and/or infertility following high-dose chemotherapy and/or irradiation (31). The ovary serves a double function: folliculogenesis and production of sex hormones, which play a main role in the woman’s body such as the development of secondary sexual characteristics. Ernst et al (2013) described a case of puberty induction in a girl treated with chemo- and radiotherapy at the age of 9 (32). Due to the treatment, her ovary had been damaged, leading to a pre-pubertal stage with postmenopausal levels of FSH. Four and a half years after OTC, tissue was grafted and she recovered normal levels of FSH and oestradiol, which let the ovaries reach the pubertal stage. Clinical effectiveness and outcomes Since the lifespan of a piece of transferred ovary varies from a few to several years (33), hormone levels can turn to those corresponding to a pre-pubertal stage some months after transplantation. The effectiveness of this technique also depends on the woman’s own ovarian reserve, which is linked to the age at which her ovary tissue was cryopreserved. In all cases reported in which pregnancy has been achieved, patients had only cryopreserved tissue of one of their ovaries, whereas the other one had been kept during the chemotherapy treatment. Thus, it has not been possible to determine whether pregnancy was a product of the cryopreserved and transferred tissue or to the remaining intact ovary. On the other hand, cases have been described in which infertility due to hormone failure could be restored by ovarian tissue allotransplantation, thus leading to healthy pregnancies (34). Even though numerous successful births have been reported after OTC, re-implantation of malignant cells (potentially causing metastatic processes), along with grafted ovarian tissue in a subject previously treated for the same cancer, still remains a serious concern. CONCLUSIONS - It is well known that treatments that expose patients to gonadotoxic therapies such as alkylating agents, pelvic irradiation, and/or stem cell transplant could increase the risk for post-therapy infertility, because the ovaries are very sensitive to these cytotoxic drugs. - Although currently OTC still remains an experimental technique, there has been an increasing amount of successfully case reports since 1996. - OTC remains an alternative and the only suitable option to preserve fertility in some special cases such as young female cancer patients. - It is necessary to make efforts in order to develop the best protocol to preserve ovarian tissue, as well as to study some unclear aspects such as the possibility of reintroducing the original disease (no cases reported, through), or to try to improve the survival of primordial follicles after grafting. - Finally, it would be recommended to follow up individuals from live births after OTC in order to analyze any abnormal findings. REFERENCES
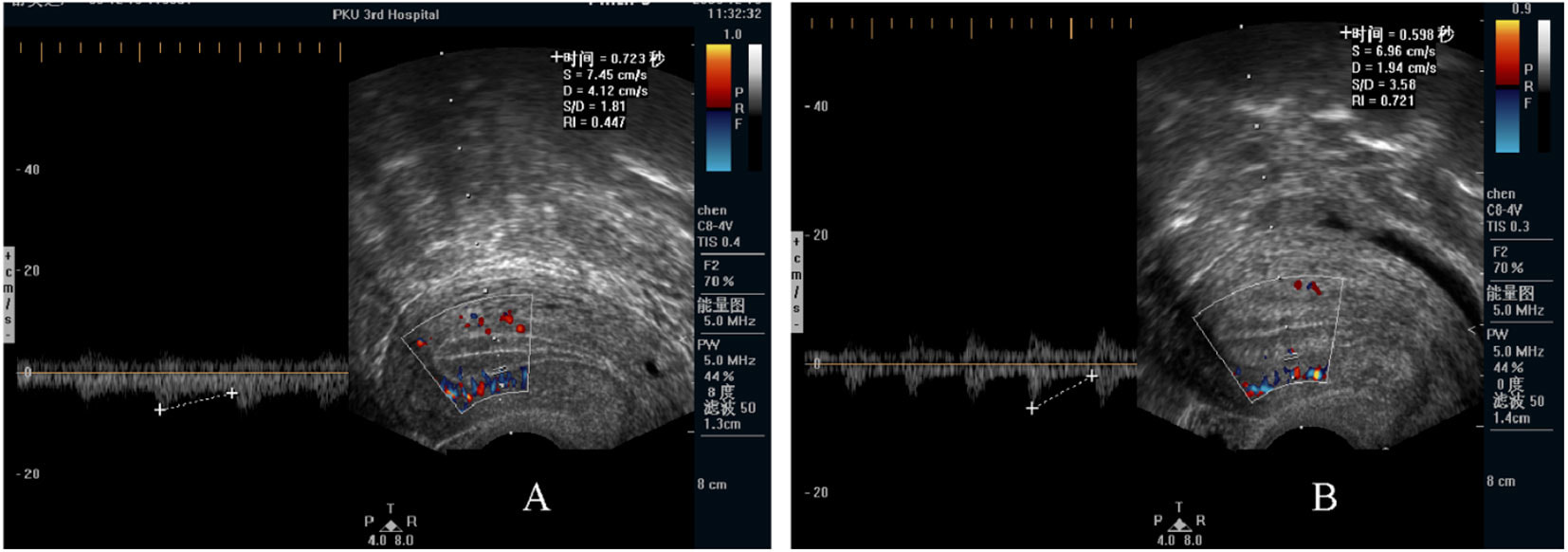
1- Roy T, Bradley C, Bowman M et al. Single-embryo transfer of vitrified-warmed blastocysts yields equivalent live-birth rates and improved neonatal outcomes compared with fresh transfers. Fertil Steril 2014;101:1294-301 2- Barnhart K. Are we ready to eliminate the transfer of fresh embryos in in vitro fertilization? Fertil Steril 2014;102:1-2 3- Roque M, Lattes K, Serra S, et al. Fresh embryo transfers versus frozen embryo transfer in in vitro fertilization cycles: A systematic review and meta-analysis. Fertil Steril 2013;99:156-162. 4- Silber S. Foreword. In: Michael J. Tucker and Juergen Liebermann. Vitrification in Assisted Reproduction.Second ed. Boca Raton FL:CRC Press; 2016 5- Chatziparasidou A, Martine N, Moisidou M, Chara O, Loakeimidou C, Pappas C, Christoforidis N. Accumulation of oocytes and/or embryos by vitrification: a new strategy for managing poor responder patients undergoing pre implantation diagnosis. F1000Research 2014,2:240. Last updated 03 JUL 2014. 6- Hovatta O, Silye R, Krausz T et al. Cryopreservation of human ovarian tissue using dimethylsulphoxide and propanediol-sucrose as cryoprotectants. Hum Reprod 1996;11:1268-72. 7- Newton H, Aubard Y, Rutherford A, Sharma V, Gosden R. Low temperature storage and grafting of human ovarian tissue. Hum Reprod 1996;11:1487-91. 8- The Ethics Committee of American Society for Reproductive Medicine. Fertility preservation and reproduction in patients facing gonadotoxic therapies: a committee opinion. Fertil Steril 2013;100:1224-12315. 9- Practice Committee of American Society for Reproductive M. Fertility preservation in patients undergoing gonadotoxic therapy or gonadectomy: a committee opinion. Fertil Steril. 2013;100(5):1214e1223. 10- Medicine PCotASfR, Technology PCotSfAR. Ovarian tissue and oocyte cryopreservation. Fertil Steril. 2006;86(5 Suppl 1):S142-7. 11- Corkum KS, Laronda MM, Rowell EE. A review of reported surgical techniques in fertility preservation for prepubertal and adolescent females facing a fertility threatening diagnosis or treatment. Am J Surg. 2017. [Epub ahead of print] 12- Xiao Z, Zhang Y, Fan W. Cryopreservation of human ovarian tissue using the silver closed vitrification system. J Assist Reprod Genet. 2017. [Epub ahead of print] 13- Silber S. Ovarian tissue cryopreservation and transplantation: scientific implications. J Assist Reprod Genet. 2016;33(12):1595-603. 14- Fathi R, Rezazadeh Valojerdi M, Ebrahimi B, Eivazkhani F, Akbarpour M, Tahaei LS, et al. Fertility Preservation in Cancer Patients: In Vivo and In Vitro Options. Cell J. 2017;19(2):173-83. 15- Anderson RA, Wallace WH, Baird DT. Ovarian cryopreservation for fertility preservation: indications and outcomes. Reproduction. 2008;136(6):681-9. 16- Moffa F, Biacchiardi CP, Fagioli F, Biasin E, Revelli A, Massobrio M, et al. Ovarian tissue cryostorage and grafting: an option to preserve fertility in pediatric patients with malignancies. Pediatr Hematol Oncol. 2007;24(1):29-44. 17- Lotz L, Maktabi A, Hoffmann I, Findeklee S, Beckmann MW, Dittrich R. Ovarian tissue cryopreservation and retransplantation--what do patients think about it? Reprod Biomed Online. 2016;32(4):394-400. 18- Herraiz S, Novella-Maestre E, Rodríguez B, Díaz C, Sánchez-Serrano M, Mirabet V, et al. Improving ovarian tissue cryopreservation for oncologic patients: slow freezing versus vitrification, effect of different procedures and devices. Fertil Steril. 2014;101(3):775-84. 19- Ladanyi C, Mor A, Christianson MS, Dhillon N, Segars JH. Recent advances in the field of ovarian tissue cryopreservation and opportunities for research. J Assist Reprod Genet. 2017;34(6):709-22. 20- Deanesly R. Inmature rat ovaries grafted after freezing and thawing. J Endocrinol 1954;11:197-200. 21- Green SH, Smith AU, Zuckerman S. The number of oocytes in ovarian autografts after freezing and thawing. J Endocrinol 1956;13:330-4. 22- Carroll J, Gosden RG,. Transplantation of frozen thawed mouse primordial follicles. Human Reprod 1993;8:1163-7. 23- Gosden RG, Baird DT, Wade JC, Webb R. Restoration of fertility to oophorectomized sheep by ovarian autografts stored at -196ºC. Human Reprod 1994;9:597-603. 24- Oktay K, Karlikaya G. Ovarian function after transplantation of frozen, banked autologous ovarian tissue. N Engl J Med. 2000;342(25):1919. 25- Donnez J, Dolmans MM, Demylle D, Jadoul P, Pirard C, Squifflet J, et al. Livebirth after orthotopic transplantation of cryopreserved ovarian tissue. Lancet. 2004; 364 (9443): 1405-10. 26- Meirow D, Levron J, Eldar-Geva T, Hardan I, Fridman E, Zalel Y, et al. Pregnancy after transplantation of cryopreserved ovarian tissue in a patient with ovarian failure after chemotherapy. N Engl J Med. 2005;353(3):318-21. 27- Demeestere I, Simon P, Dedeken L, Moffa F, Tsépélidis S, Brachet C, et al. Live birth after autograft of ovarian tissue cryopreserved during childhood. Hum Reprod. 2015;30(9):2107-9. 28- Sanfilippo S, Canis M, Smitz J, Sion B, Darcha C, Janny L, et al. Vitrification of human ovarian tissue: a practical and relevant alternative to slow freezing. Reprod Biol Endocrinol [Internet]. Reproductive Biology and Endocrinology; 2015;13(1):67. 29- Loren AW, Mangu PB, Beck LN, Brennan L, Magdalinski AJ, Partridge AH, et al. Fertility preservation for patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2013;31(19):2500–10. 30- Andersen CY, Silber SJ, Berghold SH, Jorgensen JS, Ernst E. Long-term duration of function of ovarian tissue transplants: Case reports. Reprod Biomed Online [Internet]. Reproductive Healthcare Ltd.; 2012;25(2):128–32. 31- Jensen AK, Rechnitzer C, Macklon KT, Ifversen MRS, Birkebaek N, Clausen N, et al. Cryopreservation of ovarian tissue for fertility preservation in a large cohort of young girls: focus on pubertal development. Hum Reprod. 2016;32(1):154–64. 32- Ernst E, Kjærsgaard M, Birkebæk NH, Clausen N, Andersen CY. Case report: Stimulation of puberty in a girl with chemo- and radiation therapy induced ovarian failure by transplantation of a small part of her frozen/thawed ovarian tissue. Eur J Cancer. 2013;49(4):911–4. 33- Ernst E, Bergholdt S, Jørgensen JS, Andersen CY. The first woman to give birth to two children following transplantation of frozen/thawed ovarian tissue. Hum Reprod. 2010;25(5):1280–1. 34- Silber SJ, Derosa M, Pineda J, Lenahan K, Grenia D, Gorman K, et al. A series of monozygotic twins discordant for ovarian failure: Ovary transplantation (cortical versus microvascular) and cryopreservation. Hum Reprod. 2008;23(7):1531–7. Decidualization is the combination of changes on endometrium structure and hormone profiles, along with gene expression, cell differentiation and tissue modification, that allow for implantation of the embryo. Authors: Inés Abad, Roberto de la Fuente and Sara Sanz INTRODUCTION The menstrual cycle in humans lasts 28 days in regular terms, being the first day of menses considered the first day of the cycle. But in actuality, cycle refers to a series of intermingled events mainly related to three aspects: the ovary, the uterus (or specifically the endometrium) and the hormones participating in the process. As a consequence, one may refer to either the follicular or the luteal phase regarding to the ovary, the proliferative or the secretory phase of the endometrium or even to the hormone with highest blood levels. Any of them refers to a specific moment of the cycle (Fig. 2). The window of implantation (WOI) is the term used for that phase in which the uterus is ready to receive the conceptus, around day 9 after ovulation (depending on when fertilization actually occurs). In an ideal 28-day cycle, the window of implantation is considered to start at day 20 and to last for about 5 days (3). During this interval, there is a series of physiological changes in the woman (see below) aimed to facilitate the implantation of the embryo and its survival and development in the endometrial microenvironment. Because many cases of infertility have been associated with a reduced receptivity of the endometrium, a proper WOI is currently considered a fertility-determining factor (4, 5). Hormone fluctuation within the cycle The entire menstrual cycle is controlled by levels of different hormones, some of the most important being the gonadotropins. These hormones are glycoproteins secreted by the anterior pituitary through GnRH (gonadotropin-releasing hormone) stimulation, which is strictly controlled by the hypothalamus (reviewed in (6)). The main gonadotropins involved in regulation of the menstrual cycle are FSH (follicle-stimulating hormone) and LH (luteinizing hormone). Follicular/Proliferative phase The beginning of the cycle is regarded as the beginning of the follicular phase of the ovary and the proliferative phase of the endometrium. During this phase, cohorts of follicles at different stages progress and develop due to an increase in FSH levels. The largest follicles, called Graafian follicles (~18mm), present granulosa cells that are able to produce not only oestrogen, like early smaller follicles, but also progesterone (7). The granulosa cells are the cells that conform the follicle structure, nourishing and providing support to the oocyte. Whereas progesterone levels are relatively low and constant during the first half of the cycle, there is an increment in the blood levels of oestrogen (mainly oestradiol 17β) due to its production from the follicles. However, during the last days before ovulation, largest follicles also acquire the ability to produce progesterone, and so its levels increase significantly at that time. Oestradiol secretion creates a positive feedback that stimulates LH production; but increased levels of LH are also the result of progesterone-mediated regulation (8). Follicle growth and development means that granulosa cells not only respond to FSH-stimulation, but also to LH, as well. As a consequence, levels of oestradiol that had been increasing from ~day 5 become so high that at the preovulatory phase they trigger a rapid and highly significant release of LH from the pituitary (LH surge) (9). The LH surge causes a negative feedback on the steroidogenic pathway from the ovary, and so there is a switch from the production of oestrogen to the production of progesterone (10) (Fig. 3). Luteal/Secretory phase The LH surge coincides with ovulation (~day 14) and promotes the final stages of egg maturation to occur. At this moment, the huge amount of cells left at the follicle after ovulation form the so-called corpus luteum (CL), highly active in the production of progesterone, mainly from the granulosa cells. Oestrogen production, in turn, is back to low levels although maintained relatively constant. The rapid increase in progesterone produced by the CL has another consequence: a negative feedback that causes release of LH to cease due to inhibition of translation of the mRNA into protein, and also suppression of the growth of antral follicles due to the depression of FSH production. By the end of this phase, in the absence of conception, steroid levels are reduced at luteolysis. This results in the relaxation of the negative feedback originated by progesterone, and the restoration of positive feedback that leads to the production of oestrogens, and to FSH and LH levels reaching a plateau. As a consequence, preantral follicle development is resumed along with the next cycle (7). 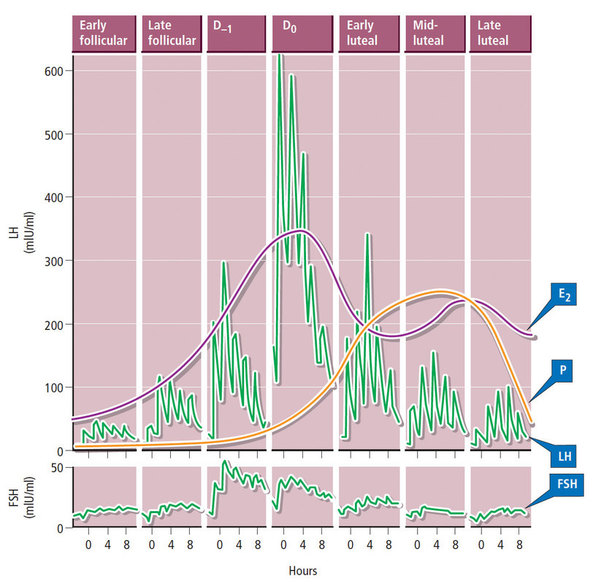 Fig 3. Profile of the main hormones involved in the menstrual cycle, with details on the different phases. Pulsatile stimulation of GnRH is seen, triggering release of LH and FSH (green profile lines). The overall levels of steroids, E2 and P, are depicted as purple and yellow lines, respectively. This image shows the clear predominance of E2 (oestrogen) in the proliferative/follicular phase vs. the main action of P (progesterone) in the secretory/luteal phase. Modified from (7). The endocrine basis of implantation It has been mentioned earlier that oestrogen (E2) is a key player during the proliferative phase of the endometrium. Besides its role in promoting stromal cell proliferation, oestrogen also induces the expression of the progesterone receptor (PR) (11), which will be of utmost importance during the secretory phase. The invasion of the endometrium by the blastocyst entails a rupture of the luminal epithelium in order to achieve implantation. For this to occur, it is necessary a series of changes in the uterine structure, as well as in the profile of several transcription factors and hormones. This way the mother adapts to the progression of the conceptus and, at the same time, controls its invasion. The development of all these changes is a process known as decidualization, which in humans starts by the mid-luteal phase, ahead of potential implantation (12). The significantly increased production of progesterone by the CL makes it the main character of the second half of the menstrual cycle. The action of progesterone has different simultaneous effects: it reduces proliferation of epithelial cells (regulated by oestrogen) while inducing their differentiation (13), and it promotes stromal cells proliferation, stimulating the glandular secretory activity. There is also a potential increase in the action of specific immune cell types (like uNKs, see below) to respond to local production of chemokines (14). Additionally, it is known that progesterone regulates the contractile activity of the junctional zone (JZ), so that it allows the conceptus to develop in a safe and controlled fashion underneath the myometrium (15). Progesterone acts partially by binding to its type A receptor (PR A) (16, 17), which in turn downregulates type B receptor (PR B), both being isoforms of the same transcriptional factor (18). The combined action of the two receptors in both stromal and glandular epithelial cells results in a modulated decidualization-like response of the endometrium in preparation for implantation. However, progesterone is not the only inducer of decidualization; in vitro progesterone only-induced differentiation of stromal cells would take up to 8-10 days under normal circumstances (19, 20). Rather, it is actually the cAMP pathway activation the main process that initiates decidualization (21, 22). In fact, many of the factors produced and secreted after ovulation like prostaglandins or gonadotropins, among others, stimulate cAMP in the stromal cells of the endometrium (22). Interestingly, cAMP levels detected from biopsies during the secretory phase are higher than during the proliferative phase (23, 24). After stimulation of cAMP, during the first stages of pregnancy, progesterone stimulates the expression of a group of interrelated genes in the endometrium, preparing the uterus for the implantation (25). Hormone profile at the beginning of gestation It has been exposed that during the luteal phase, the key acting hormone is progesterone, produced by the CL, the remnants of the Graafian follicle after ovulation. So, by the time the oocyte reaches the uterus, the endometrium is already prepared for implantation. However, if fertilization has not occurred, decidualization results useless and the physiological and endocrine profiles must be reversed. Thus, the transition from luteal to follicular phase starts around 2-3 days before the end of the menstrual cycle, with the so-called intercycle rise of FSH. This characteristic increase in FSH or FSH window is responsible for the development and eventual selection of one of the follicles in the first half of the cycle. However, after the rise of FSH, oestradiol (and inhibin B) concentration increases again, inhibiting FSH secretion, and so FSH levels are maintained (26). In humans, fertilization usually takes place in the distal third of the Fallopian tube. From there, the fertilized oocyte (now zygote) travels along the oviduct while undergoing a series of rapid divisions (cleavage), until it reaches the blastocyst stage. The blastocyst must now hatch from the hardened zona pellucida to be able to implant in the endometrium, a process that normally takes place between day 8 and 10 after ovulation (27). Not only maternal hormones regulate the process of decidualization, but the embryo itself also contributes to preparing the right microenvironment for implantation. As early as the 8-cell stage, the embryo produces hCG (human chorionic gonadotropin) (28, 29), its first active regulator of implantation. From here on, hCG secretion by the syncytiotrophoblast increases progressively after implantation, reaching detectable levels in maternal blood by day 10 of gestation (which can be normally used for pregnancy tests) and peaking around the 10th week (30). hCG has been known for decades and its current use is quite extended in reproductive medicine. In fact, it is employed to stimulate final follicle maturation before oocyte collection for IVF cycles (31), which along the external administration of progesterone for luteal phase support, helps to avoid potential problems of ovarian hyperstimulation syndrome (OHSS) derived from the use of LH. After fertilization, embryonic/placental production of hCG seems to be related with maintaining the role of the CL in secreting progesterone to sustain pregnancy (32). The need to rescue the CL from luteolysis is related to the luteoplacental shift, which occurs around the 6th week of gestation (33). This transition coincides with the decline in both progesterone and hCG serum levels detected following the 4th week and recovery thereafter (34). A crucial balance of hCG and progesterone must be reached between the CL and the syncytiotrophoblast to avoid luteal insufficiency, one of the main reasons for implantation failure during the first trimester (35). IMMUNOLOGICAL CHANGES DURING THE WOI The proliferative phase of the human endometrial cycle is characterized by the active growth of different cell types, including stromal, epithelial and vascular cells (36). On the other hand, the secretory phase supposes an increase of the glandular secretory activity and a significant increase in the number of immune cells. The major cell types in this phase are T cells, uterine NK (uNK) cells and macrophages (37). In the secretory phase, the endometrium is prepared for nidation of a conceptus, so it undergoes a series of changes during which the immune system plays an important role. Mainly, the aim of this role is to create an environment of maternal tolerance towards the conceptus, and at the same time protection against infections within the uterus (37). Immunological changes from proliferative to secretory phase T cells, essential components of the immune system and originally formed from stem cells in the bone marrow, are known to help protect the body from infections. They comprise around 45% of leukocytes in the proliferative endometrium and their number stays constant throughout the secretory phase (38). However, their relative number decreases because of a large increase in uNK cells during this phase and early pregnancy (37). During implantation, uNK cells comprise 70-80% of the total leukocyte population (39). The NK cells are a type of leukocyte or white blood cell that acts in the immune system as a first line of defense against foreign invaders. They are cytotoxic cells (40), whose function in the endometrium is to act as a source of important cytokines, including leukemia inhibitory factor (LIF), tumor necrosis factor (TNF)-α, interferon (IFN)-γ, granulocyte macrophage-colony stimulating factor (GM-CSF), and IL-10 (41). Cytokines are proteins that regulate the function of the cells that originate them on other cell types. They are primarily produced by activated lymphocytes and macrophages, and they are the agents responsible for intercellular communication, inducing the activation of specific membrane receptors, cell proliferation and differentiation, chemotaxis, growth and modulation of immunoglobulin secretion (40). Gene expression in the WOI Several genes related to the immune response are specifically regulated during the WOI. Worth to mention, among others: decay accelerating factor, indoleamine 2, 3 dioxygenase (IDO), interleukin (IL) 15, IL-15Rα, interferon regulatory factor (IRF)-1, lymphotactin, natural killer-associated transcript-2 (NKAT2) and granulysin NKG5. Some of these genes' functions are: to promote uNK cell proliferation, chemotaxis, to inhibit NK cell cytolytic activity, to inhibit cell growth and to inhibit the classical complement pathway. The functional diversity these genes represent likely serves the goal of preparing the endometrium for implantation, thereby adapting the maternal system to accommodate the immunologically ‘foreign’ fetus (42). Lobo et al. demonstrated by semi-quantitative RT-PCR (Fig. 4) and Northern analysis the upregulation of these genes in secretory versus proliferative phase (37). The mRNA of all these genes, while expressed in proliferative endometrium, demonstrated increased expression in the secretory phase. However, and taking into account the fact that uNK cells increase in number during the secretory phase, it is likely that the apparent upregulation of the above-mentioned genes is actually reflective of an increase in cell number rather than a true increase in gene expression (37). 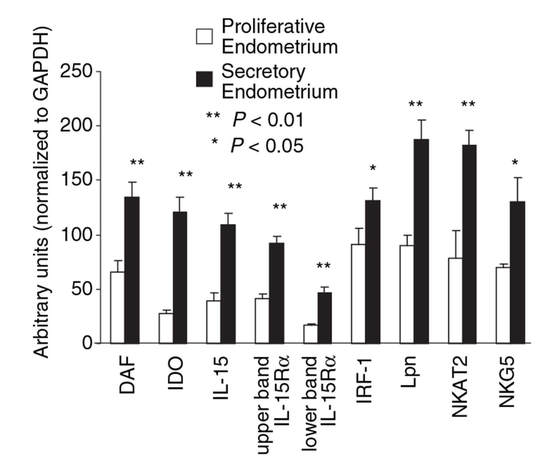 Fig.4. Graph bar showing semi-quantitative analysis of the expression of several genes, comparing proliferative (light bars) and secretory (dark bars) human endometrium. Gene expression analyzed by RT-PCR is normalized to GAPDH. There is a significant upregulation of all genes shown in the secretory phase (*P < 0.05, **P < 0.01). Modified from Lobo et al. (37). Genes that regulate an increase in uNK cell number IL-15 is a cytokine localized in the endometrium during the secretory phase of the human cycle. Its main function is to induce proliferation of uNK cells (43, 46). Some studies have demonstrated that IL-15 expression is upregulated in secretory versus proliferative phase (37, 44, 45). Additionally, Lobo et al. demonstrated the upregulation of IL-15Rα subunit from proliferative to secretory endometrium. This subunit belongs to the receptor of IL-15, and it confers high-affinity binding of cytokine to its receptor (37). IRF-1 is a transcription factor that regulates expression of IL-15 (45). IRF-1 expression has also been shown to increase during the secretory phase of the endometrium (37, 47). Genes that inhibit NK cell cytolytic activity Natural killer cells are classically considered to be cytolytic to non-self cells. Therefore, one would expect a decrease in their numbers from implantation through pregnancy to allow the fetus to be safely carried to term. Instead, there is a dramatic increase in NK cells during implantation. The answer to this paradox lies in the fact that uNK cells have effects that extend beyond their killer function, and that cytokines secreted by uNK cells are essential for implantation. In fact, NK cell-deficient mice exhibit decidual cell degeneration and endothelial cell distortion or displacement from supporting cells (48). It is believed that, at the maternal-fetal interface, human leukocyte antigen (HLA) class I molecules expressed on trophoblast cells interact with inhibitory receptors on uNK cells to protect the trophoblast from being cytolysed by uNK cells. An example of one of these inhibitory receptors is NKAT2, which shows an upregulated expression in the secretory phase (37). Genes that inhibit cell growth The uterine epithelium provides a physical barrier to infection. One mechanism for inhibiting such infections is through the secretion of endometrial peptides with bactericidal properties, which becomes especially evident at the beginning of implantation (49). NKG5 is an isoform of granulysin that exhibits antimicrobial activity against a huge spectrum of pathogens (50). The expression of this gene is especially high in NK cells surrounding the glandular epithelium in secretory endometrium (37). IDO is another example of genes that inhibit cell growth in the endometrium. It is an enzyme from the tryptophan catabolic pathway that depletes tryptophan in local tissue environments, thus suppressing proliferation of cells in the vicinity. IDO may not only inhibit T-cell proliferation, but it could also be bactericidal by this mechanism (51). An upregulation of this gene in secretory versus proliferative endometrium has been demonstrated (38). MORPHOLOGICAL CHANGES DURING IMPLANTATION WINDOW In the evaluation of women fertility, morphological studies are one of the most common procedures. These studies began with Noyes’ work, which describes the particular features of the endometrial histology, correlating it with the specific days of the menstrual cycle (52). In fact, these studies are still important and also improved and complemented thanks to the recent advances in technology. Endometrial thickness and pattern At present, there exists a data conflict between the various studies focused on endometrial thickness measurements by ultrasonography. Several papers agree in that, during an IVF cycle, the endometrium should be ≥ 7mm on the day of human chorionic gonadotropin (hCG) administration and ≥ 8mm thick on the embryo transfer day. Likewise, thickness < 6 mm has been reported to adversely affect implantation rate (53). Nevertheless, controversy arose when some studies documented no association between implantation rates and endometrial thickness. These articles claim that the important factor is the endometrial pattern, which changes throughout the entire menstrual cycle (54). Regarding endometrial pattern, the structure of a triple line is correlated with a receptive endometrium. Ultrasonography as a tool to study the endometrial pattern has a high sensitivity (79-100%), but also an elevated percentage of false positives (57-91%) (53). However, it is possible to achieve pregnancy with a “non triple-line” pattern, although at a low frequency (55). Uterine glands In the receptive endometrium, a complex secretory environment is established in order to active the blastocyst and to create both proper uterine receptivity and stromal decidualization. The structures in charge of creating this environment are the uterine glands. Once ovulation takes place, P4 levels increase and the endometrium switches into a secretory phase. The endometrium is divided into two layers, the upper one formed by loose stromal and glandular tissue in its majority, with dynamic structure and function during the cycle. On the contrary, the lower layer is structurally stable and it is formed by spiral glands and a dense stroma. During the time in which the endometrium is receptive, glycogen vesicles are disposed below the nucleus of the glandular epithelial cells, and their content is transported by microfilaments to the apical region, where glycogen is actively secreted (56). Uterine blood flow During endometrial receptivity the arterioles acquire spiral form, and there is also a similar growth of the subepithelial capillary plexus, both at basal and functional endometrium. Regarding spiral arteries, it has been reported that when glomerular capillary length reaches a certain limit, a new capillary loop is generated in order not to increase the resistance to blood flow. Additionally, during the early and mid secretory phase the vessel length, which has achieved its maximum density during the proliferative phase, experiments an increase in the number of vessel junctions (57). Recent studies have found that the existence of correct blood flow in the border area between the endometrium and myometrium plays an important role in endometrial receptivity. Uterine artery blood flow can be studied through the impedance, a term that includes the pulsatility index (PI) and the resistance index (RI) (58). Several research lines suggest that high blood flow resistance is associated with pregnant failures, whereas lower PI is a good indicator for pregnancy (Fig. 5). Applebaum et al (59). define the vascularity zones as follows:
It has been shown that pregnancy rates increase if the vessels reach the zone of the endometrium and the subendometrial halo. This fact demonstrates the relation between endometrial perfusion and endometrial growth, since the endometrium becomes thicker because of the presence of vessels. On the contrary, the absence of blood in the endometrial and subendometrial areas correlates with low probability of pregnancy, in which case uterine resistance is higher (60). Ultrasonography is also used in the study of uterine blood flow due to its non-invasiveness, real-time monitoring and repeatability. hCG administration day is normally the day for ultrasonography to be performed, because it is at this moment when highest sensitivity and specificity are achieved, although it can also be performed the day of embryo transfer (60). Uterine junctional zone The JZ is not only a distinct structure from the myometrium and endometrium, but it also presents functional differences (Fig. 6). There is supporting evidence for an important role of the JZ in the implantation process. On one hand, the movements of this layer allow the sperm to move rapidly to the dominant ovarian follicle. On the other hand, these contractions must decrease once fertilization has occurred. Otherwise, implantation might result impaired, reason why it is better to perform the embryo transfer during another cycle or, at least, at day 5-6 (58,61). Human endometrial transcriptomics Although morphological evaluation is very useful, more advantages are needed in order to study the window of implantation. Actually, transcriptomics can be used to analyze the expression of certain genes involved in the implantation process (62). Nowadays, two existing products that can be found in the market for studying receptivity using transcriptomics are “ER Map” and “Test ERA”. These products can be used in those cases in which the uterus is apparently normal, the endometrium presents the proper thickness and yet recurrent implantation failure occurs (3 or more in case of young women and 2 or more in elderly women), especially if the embryos transferred were of good quality (63,64). CONCLUSIONS The high versatility of the endometrial structure responds to the function of housing the conceptus for implantation at the right time of the cycle. It is essential that all the endometrial features adapt to the needs of both the embryo and the mother. In order to cover its nutritional requirements and, at the same time, to regulate this extraordinary changes of the endometrium, blood supply must be increased, and so there is a significant process of angiogenesis that involves restructure of the endometrium itself. The combination of all changes needed to support early embryo implantation and further development is known as decidualization. This is triggered and controlled mainly by the action of a series of hormones, mainly progesterone from the mother and hCG from the conceptus. Production and dynamics of both hormones are in turn controlled and regulated by a network of molecular interactions, involving hormones, enzymes and other factors acting in delicate balance. This allows for adaptation to the actual and potential situations, with the goal of being always prepared during the WOI, should conception happen. The menstrual cycle exhibits extreme complexity, given the high amount of factors involved in its regulation. Even though the aspects described in the present text (hormone profiles, gene expression and physiological transformation) have been studied for decades, further research is needed to fully understand the roles of other molecular factors. Unravelling complete interaction networks of these players may uncover potential roles in implantation, thus helping find new solutions to diverse causes for infertility. REFERENCES 1. Gilbert SF, Barresi MJF. Sex Determination and Gametogenesis - The biochemistry of oocyte maturation. Developmental Biology. 11th ed: Oxford University Press; 2016 - http://11e.devbio.com/wt0610.html 2. Krstić RV. Human microscopic anatomy. An atlas for students of medicine and biology. 2nd ed. Berlin-Heidelberg: Springer-Verlag; 1994. 3. Navot D, Scott RT, Droesch K, Veeck LL, Liu HC, Rosenwaks Z. The window of embryo transfer and the efficiency of human conception in vitro. Fertil Steril. 1991;55:114-8. 4. Lessey BA, Castelbaum AJ, Sawin SW, Sun J. Integrins as markers of uterine receptivity in women with primary unexplained infertility. Fertil Steril. 1995;65:535-42. 5. Strowitzki T, Germeyer A, Popovici R, von Wolff M. The human endometrium as a fertility-determining factor. Hum Reproduction Update. 2006;12(5):617-30. 6. Knobil E, Neill JD. Knobil and Neill's Physiology of Reproduction. 4th ed: Academic Press; 2015 01 Apr 2015. 2684 p. 7. Johnson MH. Essential Reproduction: Wiley-Blackwell; 2013. 8. Dafopoulos K, Kotsovassilis CG, Milingos S, Kallitsaris A, Galazios G, Zintzaras E, et al. Changes in pituitary sensitivity to GnRH in estrogen-treated post-menopausal women: evidence that gonadotrophin surge attenuating factor plays a physiological role. Hum Reproduction. 2004;19:1985-92. 9. Kobayashi M, Nakano R, Ooshima A. Immunohistochemical localization of pituitary gonadotrophins and gonadal steroids confirms the 'two-cell, two-gonadotrophin' hypothesis of steroidogenesis in the human ovary. J Endocrinol. 1990;126(3):483-8. 10. Messinis IE. Ovarian feedback, mechanism of action and possible clinical implications. Hum Reproduction Update. 2006;12:557-71. 11. Chauchereau A, Savouret JF, Milgrom E. Control of biosynthesis and post-transcriptional modification of the progesterone receptor. Biol Reprod. 1992;46(174-177). 12. Gellersen B, Brosens IA, Brosens JJ. Decidualization of the human endometrium: mechanisms, functions, and clinical perspectives. Semin Reprod Med. 2007;25(6):445-53. 13. Weitlauf HM. Biology of implantation. In: Knobil E, Neill JD, editors. The physiology of reproduction. New York: Raven Press; 1994. p. 391-440. 14. Croy BA, van den Heuvel MJ, Borzychowski AM, Tayade C. Uterine natural killer cells: a specialized differentiation regulated by ovarian hormones. Immunol Rev. 2006;214:161-85. 15. Brosens JJ, de Souza NM, Barker FG. Uterine junctional zone: function and disease. Lancet. 1995;346:558-60. 16. Wang H, Critchley HO, Kelly RW, Shen D, Baird DT. Progesterone receptor subtype B is differentially regulated in human endometrial stroma. Mol Hum Reprod. 1998;4:407-12. 17. Bergeron C. Morphological changes and protein secretion induced by progesterone in the endometrium during the luteal phase in preparation for nidation. Hum Reprod. 2000;15((Suppl 1)):119-28. 18. Tsai MJ, O'Malley BW. Molecular mechanisms of action of steroid/thyroid receptor superfamily members. Annu Rev Biochem. 1994;63:451-86. 19. Brar AK, Frank GR, Kessler CA, Cedars MI, Handwerger S. Progesterone-dependent decidualization of the human endometrium is mediated by cAMP. Endocrine. 1997;6:301-7. 20. Brosens JJ, Hayashi N, White JO. Progesterone receptor regulates decidual prolactin expression in differentiating human endometrial stromal cells. Endocrinology. 1999;140:4809-20. 21. Telgmann R, Maronde E, Taskén K, Gellersen B. Activated protein kinase A is required for differentiation-dependent transcription of the decidual prolactin gene in human endometrial stromal cells. Endocrinology. 1997;138:929-37. 22. Gellersen B, Brosens J. Cyclic AMP and progesterone receptor cross-talk in human endometrium: a decidualizing affair. J Endocrinol. 2003;178:357-72. 23. Tanaka N, Miyazaki K, Tashiro H, Mizutani H, Okamura H. Changes in adenylyl cyclase activity in human endometrium during the menstrual cycle and in human decidua during pregnancy. J Reprod Fert. 1993;97:33-9. 24. Tang B, Gurpide E. Direct effect of gonadotropins on decidualization of human endometrial stromal cells. J Steroid Biochem Mol Biol. 1993;47:115-21. 25. Bagchi IC, Cheon YP, Li Q, Bagchi MK. Progesterone receptor-regulated gene networks in implantation. Front Biosci. 2003;8:s852-s61. 26. Welt CK, Martin KA, Taylor AE, Lambert-Messerlian GM, Crowley Jr WF, Smith JA, et al. Frequency modulation of follicle-stimulating hormone (FSH) during the luteal-follicular transition: evidence for FSH control of inhibin B in normal women. J Clin Endocrinol Metab. 1997;82:2645-52. 27. Wilcox AJ, Baird DD, Weinberg CR. Time of implantation of the conceptus and loss of pregnancy. N Engl J Med. 1999;340(23):1796-9. 28. Bonduelle ML, Dodd R, Liebaers I, Van Steirtteghem A, Williamson R, Akhurst R. Chorionic gonadotrophin-β mrna, a trophoblast marker, is expressed in human 8-cell embryos derived from tripronucleate zygotes. Hum Reprod. 1988;3:909-14. 29. Lopata A, Hay DL. The potential of early human embryos to form blastocysts, hatch from their zona and secrete HCG in culture. Hum Reprod. 1989;4(8 Suppl):87-94. 30. Makrigiannakis A, Vrekoussis T, Zournakis E, Kalantaridou SN, Jeschke U. The Role of HCG in Impantation: A Mini-Review of Molecular and Clinical Evidence. Int J Mol Sci. 2017;18(6):pii: E1305. 31. Keay SD, Vatish M, Karteris E, Hillhouse EW, Randeva HS. The role of hCG in reproductive medicine. BJOG. 2004;111(11):1218-28. 32. Penzias AS. Luteal phase support. Fertil Steril. 2002;77:318-23. 33. Järvelä IY, Ruokonen A, Tekay A. Effect of rising hCG levels on the human corpus luteum during early pregnancy. Hum Reprod. 2008;23(12):2775-81. 34. Yoshimi T, Strott CA, Marshall JR, Lipsett MB. Corpus luteum function in early pregnancy. J Clin Endocrinol Metab. 1969;29:225-30. 35. Shah D, Nagarajan N. Luteal insufficiency in first trimester. Indian J Endocrinol Metab. 2013;17(1):44-9. 36. Talbi S, Hamilton AE, Vo KC, Tulac S, Overgaard MT, Dosiou C, et al. Molecular phenotyping of human endometrium distinguishes menstrual cycle phases and underlying biological processes in normo-ovulatory women. Endocrinol. 2006; 147(3): 1097-1121. 37. Lobo SC, Huang STJ, Germeyer A, Dosiou C, Vo KC, Tulac S, et al. The immune environment in human endometrium during the window of implantation. Am J Reprod Immunol. 2004; 52(4): 244-251. 38. Vassiliadou N, Bulmer JN. Quantitative analysis of T lymphocyte subsets in pregnant and nonpregnant human endometrium. Biol Reprod. 1996; 55(5): 1017-1022. 39. Pace D, Morrison L, Bulmer JN. Proliferative activity in endometrial stromal granulocytes throughout menstrual cycle and early pregnancy. Journal of clinical pathology. 1989; 42(1): 35-39. 40. Biron CA, Nguyen KB, Pien GC, Cousens LP, Salazar-Mather TP. Natural killer cells in antiviral defense: function and regulation by innate cytokines. Ann Rev Immunol. 1999; 17(1): 189-220. 41. Lash GE, Robson SC, Bulmer JN. Functional role of uterine natural killer (uNK) cells in human early pregnancy decidua. Placenta. 2010; 31: S87-S92. 42. Kao LC, Tulac S, Lobo SA, Imani B, Yang JP, Germeyer A, et al. Global gene profiling in human endometrium during the window of implantation. Endocrinol. 2002; 143(6): 2119-2138. 43. Verma S, Hiby SE, Loke YW, King A. Human decidual natural killer cells express the receptor for and respond to the cytokine interleukin 15. Biol Reprod. 2000; 62(4): 959-968. 44. Kitaya K, Yasuda J, Yagi I, Tada Y, Fushiki S, Honjo H. IL-15 expression at human endometrium and decidua. Biol Reprod. 2000; 63(3): 683-687. 45. Dunn CL, Critchley HO, Kelly RW. IL-15 regulation in human endometrial stromal cells. The Journal of Clinical Endocrinology & Metabolism. 2002; 87(4): 1898-1901. 46. Ashkar AA, Black GP, Wei Q, He H, Liang L, Head JR, et al. Assessment of requirements for IL-15 and IFN regulatory factors in uterine NK cell differentiation and function during pregnancy. The Journal of Immunology. 2003; 171(6): 2937-2944. 47. Kitaya K, Yasuda J, Fushiki S, Honjo H. Localization of interferon regulatory factor-1 in human endometrium throughout the menstrual cycle. Fertil Steril. 2001; 75(5): 992-996. 48. Greenwood JD, Minhas K, Di Santo JP, Makita M, Kiso Y, Croy BA. Ultrastructural studies of implantation sites from mice deficient in uterine natural killer cells. Placenta. 2000; 21(7): 693-702. 49. King AE, Critchley HO, Kelly RW. Presence of secretory leukocyte protease inhibitor in human endometrium and first trimester decidua suggests an antibacterial protective role. Molecular Hum Reprod. 2000; 6(2): 191-196. 50. Ernst WA, Thoma-Uszynski S, Teitelbaum R, Ko C, Hanson DA, Clayberger C, et al. Granulysin, a T cell product, kills bacteria by altering membrane permeability. J Immunol. 2000; 165(12): 7102-7108. 51. Sedlmayr P, Blaschitz A, Wintersteiger R, Semlitsch M, Hammer A, MacKenzie CR, et al. Localization of indoleamine 2, 3-dioxygenase in human female reproductive organs and the placenta. Molec Hum Reprod. 2002; 8(4): 385-391. 52. Noyes RW, Hertig AT, Rock J. Dating the endometrial biopsy. Fertil Steril. 1950; 1:3-25. 53. Friedler S, Schenker JG, Herman A, Lewin A. The role of ultrasonography in the evaluation of endometrial receptivity following assisted reproductive treatments: a critical review. Hum Reprod Update. 1996;2(4):323–35. 54. Gingold JA, Lee JA, Rodriguez-Purata J, Whitehouse M, Sandler B, Grunfeld L, et al. Endometrial Pattern but not Endometrial Thickness Impacts Implantation Rates in Euploid Embryo Transfers. Fertil Steril. 2015;104(3):620–628.e5. 55. Racowsky C, Jackson KV, Cekleniak NA, Fox JH, Hornstein MD, Ginsburg ES. The number of eight-cell embryos is a key determinant for selecting day 3 or day 5 transfer. Fertil Steril. 2000;73(3):558–64. 56. Tapia AA. Endometrial Receptivity to Embryo Implantation: Molecular Cues from Functional Genomics. In: Violin Pereira LA, editor. Embryology - Updates and Highlights on Classic Topics: CC BY 3.0 license; 2012. 57. Gambino LS, Wreford NG, Bertram JF, Dockery P, Lederman F, Rogers PAW. Angiogenesis occurs by vessel elongation in proliferative phase human endometrium. Hum Reproduction. 2002; 17(5): 1199-206. 58. Abdallah Y, Naji O, Saso S, Pexsters A, Stalder C, Sur S, et al. Ultrasound assessment of the peri-implantation uterus: a review. Ultrasound Obstet Gynecol. 2012;39(6):612–9. 59. Sardana D, Upadhyay AJ, Deepika K, Pranesh GT, Rao KA. Correlation of subendometrial-endometrial blood flow assessment by two-dimensional power Doppler with pregnancy outcome in frozen-thawed embryo transfer cycles. J Hum Reprod Sci. 2014;7(2):130–5. 60. Wang L, Qiao J, Li R, Zhen X, Liu Z. Role of endometrial blood flow assessment with color Doppler energy in predicting pregnancy outcome of IVF-ET cycles. Reprod Biol Endocrinol RBE. 2010;8:122. 61. Lesny P, Killick SR. The junctional zone of the uterus and its contractions. BJOG Int J Obstet Gynaecol. 2004;111(11):1182–9. 62. Díaz-Gimeno P, Ruíz-Alonso M, Blesa D, Simón C. Transcriptomics of the human endometrium. Int J Dev Biol. 2014;58(2–4):127–37. 63. Endometrial Receptivity Map. iGLS: http://www.igls.net/services/service-endometrial-receptivity-map/ 64. Test de receptividad endometrial | ERA. Igenomix: http://www.igenomix.com/es/tests/test-receptividad-endometrial-era/ Authors: Javier del Río, Noemi Díaz and Belén Gómez INTRODUCTION What is cryopreservation? The first successful in vitro fertilization (IVF) treatment was in 1978. Since that, there have been a remarkable number of advances in assisted reproductive technologies (ART). Initially, all available embryos were transferred in IVF treatments owing to its low success rate. However, improvements on clinical and laboratory aspects led not only to increased pregnancy rates, but also to increased risk of multiple pregnancies. To prevent this, fewer embryos are transferred and leftover embryos are cryopreserved for potential future cycles use (1). The first pregnancy resulting from transferring a thawed cryopreserved human embryo was reported in 1983 in Australia (2), and the first live birth following embryo cryopreservation was reported in 1984 in The Netherlands (3). Subsequently, the need for an effective cryopreservation program arose from rapid development and improvements of assisted reproductive technology protocols (1). Cryopreservation is a method that requires cells and embryos to be exposed to non-physiological ultra-low temperatures (from -20°C to -196°C) (Fig.2). It aims to achieve “cryogenic suspension of life” through multiple steps, although this puts the elements at risk of damage or “cryoinjury” during temperature changes and phase transitions. These damages could be chilling injury or ice crystal formation, for instance, as a result of the water exchange between the intra- and extracellular compartments, consequence of dramatic changes in osmotic potential (osmotic shock). Therefore, vitrification requires the use of cryoprotectants to avoid the formation of ice crystals in the cells. Two types of cryoprotectants are necessary: permeating and non-permeating. Mixing both at different relative concentrations reduces intracellular ice formation by removing water from inside the cell. Additionally, it creates an osmotic gradient that helps restrict water movement across the cell membrane, thereby preventing osmotic shock (4). There are two typical methods used for cryopreservation: slow freezing and rapid freezing to achieve vitrification. Vitrification is a term used to describe the transformation of a solution into glass by a dramatic increase in viscosity. This method requires to minimize the time for the sample to be exposed to temperature ranges associated with chilling injury and ice crystal formation. As slow freezing, vitrification causes cell dehydration using cryoprotectants. However, unlike that, there is no attempt to maintain equilibrium on both sides of cell membrane (4). The time frame required to reach ultralow temperatures by vitrification is very brief, almost instantaneous. But, the main concern is the need for using high concentrations of cryoprotectant solutions. These might lead to osmotic shock and it could be toxic to cells, affecting embryo survival. Nevertheless, it is possible to limit toxicity by mixing different cryoprotectants, thereby decreasing their relative concentration and the exposure time of embryos to the solution (5). How efficient is the vitrification? This technique seems to be more attractive than slow freezing because it does not require expensive equipment. It uses small amount of liquid nitrogen and it is a simpler technique to perform once the embryologist has gained enough experience in it (6). A recent research performed by Viladimoiv et al. suggests advantages arising from the freezing and thawing process; the authors hypothesize a theory about “cryo-treatment of the embryo”. According to these authors, as a result of freezing or thawing of the embryos there is a decrease in reactive oxygen species levels, in the rate of mitochondrial DNA mutation and cells detoxification is carried out. Also, the authors describe another mechanism involved in restoring the mitochondrial activity (“jumping effect”) which is part of the physiological process of implantation. However, current available data cannot confirm the hypothesis yet (7). Advantages and disadvantages of fresh and frozen cycles Nowadays, fresh embryo transfers (ET) are the most common choice in IVF cycles (8). Nevertheless, in the last years, controlled ovarian stimulation has increased the uncertainty on the possible adverse effects of the ovarian hyperstimulation syndrome (OHSS), and also on possible deleterious effects on the endometrium and implications in obstetric and perinatal results (9). In spite of this, recent developments in cryopreservation of oocytes and embryos have led to substantial improvement in IVF outcomes. This resulted in a significant increase in the number of cycles with frozen embryo transfer (FET), which subsequently led to the enhancement of live births rate (10). What are the advantages of a frozen cycle? Ovarian hyperstimulation syndrome The first strong argument for FET strategy is the prevention of OHSS, that results from an increase in vascular permeability (11,8). OHSS is a medical condition affecting the ovaries of some women who take fertility medication to stimulate oocyte growth. OHSS arguably remains a major cause of morbidity in IVF treatment (10). During a fresh cycle, a woman has to undergo hormonal treatment to regulate her menstrual period, to stimulate the development of multiple oocytes (superovulation), and to encourage their maturation (11, 12). However, in a frozen cycle (FC) the patient does not have to go through ovarian stimulation or egg retrieval depending on their circumstances (13). Many people find that FETs are less stressful than fresh cycles because they do not have to worry about oocytes production or whether there will be viable embryos, since those procedures have already been done (9). Deleterious effects on the embryo The optimization of vitrification protocols has reduced the deleterious effects that this process may produce in embryos. Also, it have been observed similar survival and embryo development in FCs compared to fresh cycles (10). Moreover, best quality embryos, morphologywise, can be stored and transferred in a future cycle in better conditions. These data have allowed for an increment of success rates and the confidence of sanitary personnel and patients over FCs (5). Endometrial receptivity The implantation process, one of the crucial steps in the success of ART, requires a reciprocal interaction between the embryo and the endometrium during a small period of time called window of implantation. This interaction involves the embryo, along with its inherent molecular program of cell growth and differentiation, as well as differentiation of endometrial cells into an adequate uterine receptivity (11). Some patients may find easier to turn to FCs, since dealing with the whole process of medication during a normal cycle for ovarian stimulation may result psychologically and emotionally overwhelming. In this regard, FC may also provide a better outcome (3). The importance of an adequate endometrial environment in ART is highlighted in those patients who resort to oocyte donation, where there must be a synchronization between donor and recipient in fresh cycles. Those cases that require an improvement in endometrial receptivity to stimulate implantation of these donor oocytes seem to obtain better results in frozen cycles or in the next fresh cycle (8). Multiplet pregnancies are one of the major safety concerns of IVF due to the increased risk of neonatal and maternal complications. To achieve good results, to would be ideal to select the optimal single embryo to be transferred. Elective single embryo transfer (eSET) is the most effective way to reduce those risky pregnancies (14). How can cryopreservation damage embryos? Upon analyzing some ART studies and results, embryos are able to adapt and develop in a large range of culture media, showing different gene expression models in different environments. Cryopreservation causes stress in embryos and it is known as “hormesis”(5) (Fig.3). However, if the conditions are too unfavorable or toxic, mitochondrial activity is suppressed below the threshold necessary for the development of the embryo, so that implantation in the endometrium will be affected (5). Results of embryo transfer in fresh cycles vs. frozen cycles The main current objective of IVF professionals is to improve pregnancy rates in both fresh and frozen-thawed cycles. It is clear that embryo and endometrial receptivity are important factors to promote pregnancy rate. Recently, many researches showed FET can enhance the embryo utilization rate and improve the success rate in contrast to other research lines (15). In Roque et al. systematic meta‐analysis for 633 cycles in women aged 27-33 years old showed that FET resulted in a statistically significant increase in the ongoing pregnancy rate and clinical pregnancy compared with the fresh transfer group (8). Interestingly, the fresh group showed a higher miscarriage rate, but no statistical difference was found when compared with the frozen group. According to these data, it seems that the results of IVF-ICSI cycles can be improved by performing the FET especially in patients with normal or high follicular response. This advantage could be explained thanks to a more physiological preparation of endometrium. Several studies have also shown good results with cryopreservation of all embryos and subsequent FET in those patients most susceptible to develop OHSS (8, 16-19). In contrast, Shavit et al. found lower rates of clinical pregnancy and live births in the vitrified-warmed blastocyst group. The difference in implantation and pregnancy rates could be attributed to a higher proportion of good-quality embryos in the fresh blastocysts transfer group. They suggest that in fresh cycles highest quality blastocyst is selected for transfer and the rest are usually vitrified. Thus, vitrified-warmed blastocysts may have slightly poorer grade after warming and prior to transfer (20). In addition, it is necessary to take into account those cycles with frozen oocytes. Braga et al. found that warmed oocytes transferred in endometrial prepared cycles yield better clinical outcomes than fresh ETs. Indeed, they found that fertilization rate, embryo quality, and developmental competence was decreased in embryos derived from vitrified oocytes (12). Conversely, previous studies have suggested that the results of oocyte vitrification followed by ICSI are not inferior with regard to fertilization, embryo developmental competence, pregnancy rates, and live birth (21, 22, 23). An interesting point found in Braga et al. research is that even with lower embryo developmental quality, warmed oocytes transferred in endometrial prepared cycles resulted in higher pregnancy and implantation rates compared with transfer in fresh cycles. This finding strongly suggests that controlled ovarian stimulation impacts endometrial receptivity, which may be a possible cause of implantation failure after ovarian stimulation (12). Indeed, some studies have suggested that pregnancy rate is inversely related to serum progesterone levels on the day of hCG administration (24-27). It has been demonstrated that elevated progesterone levels on hCG trigger day negatively influence the pregnancy, regardless of the oocyte quality. Raised concentrations of progesterone in the late follicular phase are likely to influence the secretory changes of the endometrium, leading to an asynchrony between embryo and endometrial dialogue, which may result in reduced implantation rate (12). Another issue to consider is the obstetric and perinatal outcomes of frozen-thawed cycles. Maheshwari et al. quantified in a meta-analysis the obstetric and perinatal risks for singleton pregnancies after FET and compared it with those after fresh embryo transfer (28). They indicated better perinatal outcomes in singleton pregnancies after the transfer of frozen‐thawed embryos when compared to fresh IVF embryos. This could be explained by antepartum hemorrhage, very preterm birth (delivery at <32 weeks), preterm delivery (delivery at <37 weeks), small for gestational age, low birth weight (birth weight <2500 g), and perinatal mortality significantly lower in women who received frozen embryos than those transferred with fresh embryos (29, 28). It is important to note that most studies comparing perinatal outcome of singleton births conceived after fresh and cryopreserved ETs included both single and multiple ETs. Therefore, part of the adverse perinatal outcome may be attributed to the vanishing twin phenomenon, which occurs in up to 10% of multiple ETs resulting in a singleton live birth (20). What can we conclude? Elective embryo cryopreservation followed by single FET has attracted increasing attention and has been regarded as a potential innovation of IVF treatment. Choosing the well-selected embryo could further increase the chance of live birth of a eSET, which is of high clinical significance. However, there are great gaps in the literature about the risk/benefit ratio of this strategy, which includes multiple steps of treatment (30). The good outcomes in FC might be associated with having a well‐balanced embryo‐endometrium interaction in FC, and also with lacking controlled ovarian hyperstimulation, which may adversely affect endometrial receptivity during fresh IVF cycles. In addition, when hormone replacement cycles were applied in FETs, estrogen and progesterone were given in physiological doses to mimic natural cycles, while supraphysiological doses of gonadotropins were given in fresh cycles (31). On the other hand, other authors find fresh cycles as the best choice, especially in patients who resort to oocyte donation. In fact, it seems that there is a higher proportion of good-quality embryos in fresh blastocysts compared to vitrified-warmed blastocysts, which may have slightly poorer grade after warming and prior to transfer. (8, 20). In conclusion, each case must be individualized in relation to clinical characteristics of the patients and to oocyte, seminal and embryo quality. By doing so, results will be optimized in each cycle and the chances of achieving a live birth will be highly improved. REFERENCES:
1. Wong KM, Mastenbroek S, Repping S. Cryopreservation of human embryos and its contribution to in vitro fertilization success rates. Fertil Steril. 2014;102(1):19-26. 2. Trounson A, Mohr L. Human pregnancy following cryopreservation, thawing and transfer of an eight-cell embryo. Nature. 1983;305(5936):707-9. 3. Zeilmaker GH, Alberda AT, van Gent I, Rijkmans CM, Drogendijk AC. Two pregnancies following transfer of intact frozen-thawed embryos. Fertil Steril. 1984; 42(2):293-6. 4. Sparks AE. Human embryo cryopreservation-methods, timing, and other considerations for optimizing an embryo cryopreservation program. Semin Reprod Med. 2015;33(2):128-44. 5. Konc J, Kanyó K, Kriston R, Somoskői B, Cseh S. Cryopreservation of embryos and oocytes in human assisted reproduction. Biomed Res Int. 2014;2014:307268. 6. Loutradi KE, Kolibianakis EM, Venetis CA, Papanikolaou EG, Pados G, Bontis I, et al. Cryopreservation of human embryos by vitrification or slow freezing: a systematic review and meta-analysis. Fertil Steril. 2008;90(1):186-93. 7. Vladimirov IK, Tacheva D, Diez A. Theory about the Embryo Cryo-Treatment. Reprod Med Biol. 2017;16:118–125. 8. Roque M, Lattes K, Serra S, Solá I, Geber S, Carreras R, Checa MA. Fresh embryo transfer versus frozen embryo transfer in in vitro fertilization cycles: a systematic review and meta-analysis. Fertil Steril. 2013;99(1):156-62. 9. Gurbuz AS, Gode F, Ozcimen N, Isik AZ.Gonadotrophin-releasing hormone agonist trigger and freeze-all strategy does not prevent severe ovarian hyperstimulation syndrome: a report of three cases. Reprod Biomed Online 2014;29:541-544. 10. Lattes K, Prat M, Robles A, Carreras R, Brassesco M, Checa MA. Ciclos de criopreservación y vitrificación de ovocitos y embriones: indicaciones y transferencia diferida. Guía 21 de Práctica Clínica de la SEF y de la SEGO. 11. Lessey BA. Endometrial receptivity and the window of implantation. Baillieres Best Pract Res Clin Obstet Gynaecol. 2000;14(5):775-88. 12. Braga D, Setti A, Figueira R, Azevedo M, Iaconelli A, Lo Turco E et al. Freeze-all, oocyte vitrification, or fresh embryo transfer? Lessons from an egg-sharing donation program. Fertil Steril. 2016;106(3):615-622. 13. Shapiro BS, Daneshmand ST, Garner FC, Aguirre M, Hudson C. Clinical rationale for cryopreservation of entire embryo cohorts in lieu of fresh transfer. Fertil Steril. 2014;102:3-9. 14. Tobias T, Sharara FI, Franasiak JM, Heiser PW, Pinckney-Clark E. Promoting the use of elective single embryo transfer in clinical practice. Fertil Res Pract. 2016;2(1):1-9. 15. Shen C, Shu D, Zhao X, Gao Y. Comparison of clinical outcomes between fresh embryo transfers and frozen-thawed embryo transfers. Iran J Reprod Med. 2014. Jun;12(6):409–14. 16. Griesinger G, von Otte S, Schroer A, Ludwig AK, Diedrich K, Al-Hasani S, et al. Elective cryopreservation of all pronuclear oocytes after GnRH agonist triggering of final oocyte maturation in patients at risk of developing OHSS: a prospective, observational proof-of-concept study. Hum Reprod. 2007;22(5):1348-1352. 17. D'Angelo A. Ovarian hyperstimulation syndrome prevention strategies: cryopreservation of all embryos. Semin Reprod Med. 2010;28(6):513-518. 18. Griesinger G, Schultz L, Bauer T, Broessner A, Frambach T, Kissler S. Ovarian hyperstimulation síndrome prevention by gonadotropin-releasing hormone agonist triggering of final oocyte maturation in a gonadotropin-releasing hormone antagonist protocol in combination with ‘‘freeze-all’’ strategy: a prospective multicentric study. Fertil Steril. 2011;95(6):2029-2033. 19. Devroey P, Polyzos NP, Blockeel C. An OHSS-Free Clinic by segmentation of IVF treatment. Hum Reprod. 2011;26(10):2593-2597. 20. Shavit T, Oron G, Weon-Young S, Holzer H, Tulandi T. Vitrified-warmed single-embryo transfers may be associated with increased maternal complications compared with fresh single-embryo transfers. Reprod Biomed Online. 2017;35(1):94-102. 21. Trokoudes KM, Pavlides C, Zhang X. Comparison outcome of fresh and vitri- fied donor oocytes in an egg-sharing donation program. Fertil Steril. 2011; 95:1996-2000. 22. Herrero L, Pareja S, Aragones M, Cobo A, Bronet F, Garcia-Velasco JA. Oocyte versus embryo vitrification for delayed embryo transfer: an observational study. Reprod Biomed Online. 2014;29:567-72. 23. Rienzi L, Romano S, Albricci L, Maggiulli R, Capalbo A, Baroni E, et al. Embryo development of fresh ‘versus’ vitrified metaphase II oocytes after ICSI: a prospective randomized sibling-oocyte study. Hum Reprod. 2010;25:66-73. 24. Xu, B., Li, Z., Zhang, H., Jin, L., Li, Y., Ai, J. et al, Serum progesterone level effects on the outcome of in vitro fertilization in patients with different ovarian response: an analysis of more than 10,000 cycles. Fertil Steril. 2012;97 (1321-7.e1-4). 25. Wu, Z., Li, R., Ma, Y., Deng, B., Zhang, X., Meng, Y. et al, Effect of HCG-day serum progesterone and oestradiol concentrations on pregnancy outcomes in GnRH agonist cycles. Reprod Biomed Online. 2012;24:511–520. 26. Bosch, E., Labarta, E., Crespo, J., Simon, C., Remohi, J., Jenkins, J. et al, Circulating progesterone levels and ongoing pregnancy rates in controlled ovarian stimulation cycles for in vitro fertilization: analysis of over 4000 cycles. Hum Reprod. 2010;25:2092–2100. 27. Hamdine, O., Macklon, N.S., Eijkemans, M.J., Laven, J.S., Cohlen, B.J., Verhoeff, A. et al, Elevated early follicular progesterone levels and in vitro fertilization outcomes: a prospective intervention study and meta-analysis. Fertil Steril. 2014;102:448–454.e1. 28. Maheshwari A, Pandey S, Shetty A, Hamilton M, Bhattacharya S. Obstetric and perinatal outcomes in singleton pregnancies resulting from the transfer of frozen thawed versus fresh embryos generated through in vitro fertilization treatment: a systematic review and meta-analysis. Fertil Steril. 2012;98:368–77.e1. 29. Qiao J, Zhang L, Yan L, Zhi X, Yan J. Female Fertility: Is it Safe to "Freeze?". Chin Med J (Engl). 2015;128(3):390. 30. Wei D, Sun Y, Liu J, Liang X, Zhu Y et al. Live birth after fresh versus frozen single blastocyst transfer (Frefro-blastocyst): study protocol for a randomized controlled trial. Trials 2017; 18(253): 1-7. 31. Zhang L, Yan LY, Zhi X, Yan J, Qiao J. Female Fertility: Is it Safe to “Freeze?” Chin Med J. 2015;128 (3):390-7. Authors: Shuyana Deba and Roberto de la Fuente 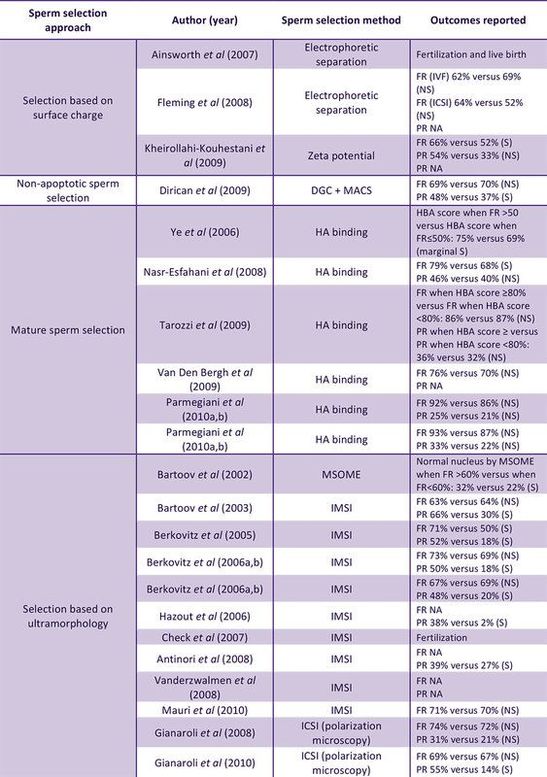 Figure 2. Collection of studies that relate the effects of advanced methods of sperm selection and clinical outcomes (FR=fertilization rate; PR=pregnancy rate). Modified from [11]. *Please consult the original source for references therein. Figure 2. Collection of studies that relate the effects of advanced methods of sperm selection and clinical outcomes (FR=fertilization rate; PR=pregnancy rate). Modified from [11]. *Please consult the original source for references therein. INTRODUCTION In assisted laboratories, semen samples are usually obtained after masturbation and the ejaculate is collected into a container. However, semen recovered from non-spermicidal condoms can be of higher quality because of the time producing the sample influences in the amount of seminal emission before ejaculation (2). Moreover, according to the World Health Organization criteria, laboratory manual for the examination and processing of human semen, the results of laboratory measurements of semen quality will depend on: completed collection of the sample, products of the accessory glands (that will dilute the concentration of epididymal spermatozoa), time past since last sexual activity, abstinence period and the size of the testis. All these factors, among others, will have an influence on the total sperm count per ejaculate (2). Contrary to what is found in natural fertilization after ejaculation, there are no actual barriers to enhance semen capacitation in vitro. As a consequence, in assisted reproduction laboratories sperm must be separated from the seminal plasma within one hour after ejaculation. The purpose of semen processing is to increase the concentration of high quality spermatozoa, and the method choice will depend on the nature of the sperm, that will in turn determine which reproductive procedure will be performed (2). Reports over the last two decades have made emphasis on the need for the optimal method for sperm selection to compile as many as possible of the following conditions: non-toxicity, ease to perform, inexpensiveness, suitability for high-throughput sample processing, accuracy in selecting the best subpopulation of sperm and ability to discard other cell types and seminal plasma as well as other substances that may harm the sperm (cryoprotectants, bioactive complements, motility enhancers) (3). Unfortunately, no known method to date successfully meet all the above, and so the selection of the most appropriate method normally depends on the specific procedure to follow. SPERM PREPARATION “Conventional” techniques like the nowadays common washing swim-up (WSU) and differential density gradient centrifugation (DDGC) procedures are normally used for a variety of assisted reproductive procedures, although it is clear that not all of them will have comparable efficiency in selecting high quality sperm. Some techniques are better indicated for certain conditions. For instance, high concentrations of highly motile sperm from normospermic men will show a good performance after WSU, and this will be suitable for regular IVF. On the contrary, severe oligospermic or asthenospermic individuals will require semen selection by using other techniques. The introduction of ICSI in the regular clinical practice enhanced the fertilization rates in the 90’s (4), but did not significantly increase live birth rates because of failures in early development, mostly due to the high incidence of structural chromosomal aberrations (5). This is a direct consequence of the inability of ICSI to specifically detect good quality spermatozoa. In the following paragraphs we discuss the appropriateness of different techniques for distinct assisted reproduction treatments, namely: in vitro fertilization (IVF), intrauterine insemination (IUI) and intracytoplasmic sperm injection (ICSI). Sperm preparation techniques for IVF In the last few years several studies have discussed about the best technique for sperm capacitation in vitro. Recently, Volpes and collaborators compared the effects of four methods used for sperm preparation, namely, direct swim-up, pellet swim-up, density gradient and density gradient followed by swim-up (6). The authors evaluated levels of DNA fragmentation using the sperm chromatin dispersion test for samples meeting the following criteria: minimum volume of 2 mL, minimum sperm concentration of 10 million/mL and a minimum motility of 35%. The study showed lower DNA fragmentation after pellet swim-up and after density gradient followed by swim-up. However, the study highlighted the limitation that clinical outcomes from IVF/ICSI procedures were not correlated with DNA fragmentation in the sperm (6). A different study compared sperm processing by WSU and DDGC in normospermic individuals, analyzing sperm motility, concentration, and morphology recovery rates (7). Conclusions were that, on the one hand, DGC was appropriate for males with low sperm concentration, since it yielded higher sperm concentration than the WSU technique. On the other hand, the latter facilitated a more efficient morphology-based sperm selection. Nevertheless, the main limitation of the study was the small number of patients and the fact that all of them were normospermic (7). In 2016, Yamanaka and coauthors attempted to determine the efficiency of combining both DGC and WSU techniques in reducing the number of sperm with abnormal nuclear morphology under the microscope (8). Results showed that combination of the two approaches was better than one alone; both DNA fragmentation levels and sperm motility were improved compared to results after just DGC. Data also demonstrated that the combination of both techniques was efficient in enriching the sample with sperm with normal head and flagellum morphology (8). Intrauterine Insemination (IUI) A study by Karamahmutoglu on the most effective sperm preparation technique for IUI compared WSU vs. DGC (9). Even though data showed higher IUI success rates after having performed the DGC approach, no significant difference was found in the "mild male factor" subfertile group (with sperm count in the range of 5-15 million/mL). Moreover, other factors were affecting fecundity success rate, such as female age, number of cycles and type of infertility (9). These observations on the efficacy of DGC and WSU methods for IUI have recently been confirmed in a similar study by Butt and Chohan (10). On the particularities of ICSI Sperm selection for ICSI is commonly carried out by the embryologist’s own judgement based on morphological criteria. This results in inconsistent decision-making and often selection of poor quality sperm, since semen samples are considered morphologically normal with just 4% of normal-looking spermatozoa (2). Therefore functional, physiological and molecular traits of spermatozoa cannot be evaluated by ICSI, and so unnoticed DNA abnormalities (even specific causes of sterility) might be passed on to the offspring by the selected spermatozoon. Thus it is easy to understand why the embryology and reproduction community has been trying to develop new strategies to successfully select the best spermatozoa regarding phenotype, functional characteristics and genetic and molecular integrity. Advanced methods for sperm preparation There are currently three main groups of methods to facilitate selection of high quality sperm, based on morphology, electrical charge and sperm surface maturity and organization (11, 12):
A simpler and cheaper technique uses the electrokinetic potential of the sperm (19); the electric potential difference of the sperm membrane decreases with capacitation, which is used to pipette washed sperm into positively charged tubes, so that negatively charged (mature) sperm can be retrieved afterwards. However, the total sperm recovered by using this technique is low, which represents an important limitation (11).
PS externalization to the outer sperm membrane is a typical apoptotic feature, which allows the cell to bind to magnetic beads conjugated with Annexin-V. This made it possible to develop a magnetic-activated cell sorting system (MACS) (25); the sperm suspension is incubated with the microbeads so that those apoptotic spermatozoa will bind to the beads, which will be subsequently retained within the MACS column in a magnet. Non-apoptotic sperm will then flow freely to be collected. Even though this technique enriches the sample in healthy sperm, it does not discard leukocytes or germ cells, and thus it must be combined with DDGC (26). A variation of the approach known as Annexin V glass wool (annexin V-GW) eliminates potential side effects of free magnetic beads (27), but still needs to be combined with repeated DGC cycles, which is actually not appropriate for oligospermic individuals (11). CONCLUSIONS The approach followed for semen preparation and selection for every patient/couple needs to be chosen upon a series of factors that mainly refer to the cause of infertility. The wide range of situations found regarding this topic makes it necessary to adopt a specific strategy every time. This will ultimately define the treatment to be applied and the techniques to be carried out subsequently. Recent advances have developed new and better ways to detect good quality spermatozoa, minimizing DNA fragmentation and optimizing the rates of good morphology or motility, for instance. However, it is important to remember that there exists no particular strategy that always relates to the optimal clinical outcome. On the contrary, each situation must be considered in the light of the patient’s needs and characteristics, and so the technique for sperm preparation must be chosen accordingly. REFERENCES
Authors: Inés Abad, Roberto de la Fuente and Sara Sanz INTRODUCTION Fertility treatments are more and more common in our days, reason why it is important to perform these procedures accurately resembling in vivo conditions. Additionally, male factor may oftentimes be underrated, and yet it is 50% of the treatment. The following text aims to establish an updated comparison between in vivo and in vitro semen preparation methods. In the first part a general description of the processes of maturation and capacitation of sperm are presented. MATURATION Where does sperm maturation take place? Once spermatogenesis is completed in the seminiferous epithelium, immature spermatozoa migrate towards the epididymis, the organ in which sperm maturation and storage take place. The epididymis is usually divided into three different parts: caput (head), corpus (body) and cauda (tail) (2). Typical changes in sperm during maturation 1. Acquisition of progressive motility. Even though immature sperm have functional movement machinery, motility of these cells begins in the caput segment. Whereas beating intensity is similar throughout the whole epididymis, flagellar amplitude is modified within this path. This is due to changes on the sperm surface, such as acquisition of new proteins and molecular changes involving inactivation of Ser/Thr phosphatases (3, 4). 2. Migration of the cytoplasmic droplet (CD). This droplet migrates from the neck towards the annulus of the mammalian spermatozoa (in humans, the CD is more proximal, located at the neck as opposed to the distal position of the annulus). The role of this droplet is to regulate ion homeostasis. It contains K+, Cl- and water channels, which have been suggested to work in regulation of sperm volume during the different regions of the epididymis. It also accumulates Ca2+, which has a biphasic role controlling phosphorylation pathways in sperm cells. In immature spermatozoa, it has been hypothesized that high Ca2+ levels found in the CD maintain low levels of tyrosine phosphorylation (5). 3. Changes in sperm protein and lipid profile. - Protein and lipid content Even though changes in these profiles are not well understood, there are three complementary mechanisms that participate in completing maturation:
Perhaps the most important among these changes is the significant reduction in cholesterol content of the sperm membrane. This reduction involves a decrease in the cholesterol/phospholipid ratio that facilitates protein trafficking from and onto the membrane and enhances its fluidity, which will eventually play a role in triggering capacitation and fertilisation (8). - Post-translational protein changes Additionally, certain post-translational modifications of proteins have also been hypothesised to occur during sperm maturation. This is the case for oxidation of thiol groups, which promotes the formation of disulphide bonds (S-S) and stabilises components of both the head and flagellum (9).  Fig.2. Schematic representation of the main items during sperm maturation. (A) Principal functional and morphological aspects in immature caput spermatozoa. (B) Molecular characteristics of immature spermatozoa. (C) Main morphofunctional traits of cauda mature spermatozoa and (D) their molecular features (4) (*) Ability to fertilize the egg will ultimately depend on completing capacitation. CAPACITATION What is the composition of the ejaculate? In mammals, semen is composed of two different phases:
Following ejaculation, semen is deposited to the anterior wall of the vagina, adjacent to the ectocervical tissues. From here on, for sperm to progress towards the egg through the oviduct or Fallopian tubes, semen must undergo liquefaction. This process usually takes about 20-30 minutes (11). Semen goes through the cervix and reaches the distal portion of the Fallopian tube, where sperm is stored and maintained by interacting with the endosalpingeal epithelium (12, 13). In 1951, Austin and Chang individually observed that a certain period of time in the female tract was required for sperm before fertilization could take place (14, 15). Later, observations in multiple mammalian species confirmed these first notions, and certain studies showed a delay of at least 2 hours before sperm entry into the egg. This supports the hypothesis of sperm maturation before becoming fertile after ejaculation (16). Nowadays, such process is known as capacitation, as opposed to maturation in the male tract explained above. Capacitation and fertility Some of the factors involved in sperm capacitation are steroid hormones such as oestrogens and progesterone, both produced by the follicle. These steroids play different roles: they act as chemoattractants, facilitate triggering of hyperactivation, regulate trafficking of cGMP or modulate the potential for completing acrosome reaction (17-19). Semen liquefaction following ejaculation is mainly modulated by prostate derived peptidase KLK3. In females, KLKs 5–8, 10–11, and 13–15 are expressed at very high levels in the cervix and vagina compared to other adult tissues (20, 21). Moreover, KLK1 and KLK3 transcripts are expressed at the highest level in human endometrium when circulating estradiol (E2) is elevated. These findings suggest that KLKs are expressed in the human reproductive tracts and that some of the KLKs in the uteri are regulated by E2. Abnormal E2 signalling in the female reproductive tract leads to semen liquefaction defects, associated with defective SEMG cleavage and sperm transport, which may result in some cases of infertility. It is known that mice lacking ESR1 (one of the oestrogen receptors) in the epithelial cells are infertile (22), partly due to a reduction in the number of sperm able to reach the oviduct (23). However, the effect of ESR1 loss in the epithelial cells on sperm transport in the uterus has not yet been investigated. Similarly, other potential research lines could investigate liquefaction defects caused by diminished KLK activity in females or regulation of KLKs by molecular signalling in the female tract. Once semen trespasses the cervix, sperm are known to achieve capacitation in an asynchronous fashion during the interaction with the epithelium, which results in a continuous flow of fertile spermatozoa towards the Graafian follicle (24). The ability to bind to the epithelium, in turn, may be indirectly related to the sperm DNA integrity, and so DNA fragmentation levels would be indicative of the fertility potential of the sperm (25). Elements involved in sperm capacitation Even though capacitation had traditionally been regarded as a two-step process, through which changes in the cell membrane would lead to the acrosome reaction (AR) (26), capacitation is currently considered as a continuous process that culminates in the AR. It would be difficult to describe all capacitation-related events separately because all of them are connected to each other in time. However, the most important changes in the sperm during the process are (27-37):
Sperm capacitation is a complex process with multitude of interconnected and highly regulated molecular pathways. One of the first events is the alteration of the permeabilization of the sperm plasma, so that the influx and intracellular concentration of certain ions are increased. The main molecules involved are probably Ca2+ and HCO3-; the net intake by the sperm cell triggers alkalinisation of the pH and the concomitant activation of the soluble adenylyl cyclase (sAC) (38, 39). As an immediate consequence, cAMP levels increase followed by activation of the protein kinase A (PKA) (40, 41). The rise of cAMP causes redistribution of certain phospholipids and proteins of the membrane, and so exposing cholesterol, which accumulates in lipid rafts (42). The organization of these rafts promotes the removal of cholesterol and its translocation to extracellular acceptors like albumin (43). Also, increased cAMP activates PKA, which in turn activates SRC kinase (44). Eventually, SRC kinase activity triggers tyrosine phosphorylation, which results and a wide range of proteins been modified and relocated in capacitating sperm. This has been described in several species, including humans (45). The end result of capacitation is the acrosome reaction (AR), the process by which the content of the acrosome is released to the extracellular environment. In natural conditions, this environment is actually the cummulus cells, whose connections will be broken by the chemical reactions of the acrosomal content, mainly proteases like acrosin and hyaluronidase, also exposed to the right membrane domains during lipid redistribution (46). It is not surprising that mutations affecting any of these processes will result in multiple causes for infertility (47). FUTURE PERSPECTIVES Findings like the one regarding post-ejaculated liquefaction, mutations on acrosome protease-encoding genes or other molecular mechanisms of sperm capacitation are crucial to progress in the field of reproductive medicine, and can lead to: (i) potential diagnostic tools for unexplained infertility cases, (ii) the development of a novel contraception technology to entrap sperm (48), (iii) or even revolutionary new methods for human sperm capacitation in the laboratory (49), which could significantly improve live birth rates for fertility treatments. In the following post... different methods for sperm selection in the laboratory will be explained, paying attention to advantages and disadvantages under different circumstances. The importance of different sperm features like DNA fragmentation or morphology will be discussed in relation to the best sperm selection method to achieve optimal clinical outcomes. REFERENCES:
Authors: Paula Brígido, Roberto de la Fuente and Javier Del Río Assisted reproduction technology (ART) can help fertile couples to achieve successful pregnancies. Sometimes, reproductive desires of these couples are affected by the presence of a genetic disease in either partner. In such cases, couples are at a reproductive risk and find themselves in the need of assistance that only ART can provide. Preimplantation genetic diagnosis (PGD) provides an alternative to prenatal diagnosis to detect the specific genetic condition or disease they suffer from, and allows them to avoid passing it on their offspring (2). It requires the analyses of the embryos generated by ART in the in vitro fertilization (IVF) laboratory, by means of accurate and sensitive methodologies such as embryo biopsy, genetics, single cell genomics and, of course, background on prenatal diagnosis and counselling from experts. Clinical application of PGD dates back to the late 60’s, when blastocysts of research animals could be sexed (3) (note that this was already possible ten years before Louis Brown, the first IVF baby, was born in the UK in 1978). At the beginning of the 90’s, early human embryos were sexed before implantation and the first genetic analyses were performed to avoid children inheriting Mendelian diseases. By the end of the century, other nowadays considered basic genetic methodologies were routinely used for preimplantation diagnosis and PGD was applied as a normal procedure to guarantee healthy babies (4). In the present post we aim to give an account of the importance of PGD and the current view of the main clinical approaches for its application. WHEN IS PGD INDICATED? Indications for PGD are multiple and emerge from different motivations. Firstly, the patient may have suffered from a number of terminations due to the embryo having inherited the genetic condition. It could also be motivated by the parents already having a child with a severe genetic disease. In this case they might be willing to avoid passing it on the next one or even looking for a suitable treatment, if possible. However, one of the parents (or both) may be worried about their family history, being aware of the presence of a specific genetic condition, regardless of the type of inheritance. If the parents are carriers of any genetic disease, either an autosomal-dominant disorder like Huntington disease or an autosomal-recessive one like cystic fibrosis, they are at reproductive risk because the resulting embryo may be affected (the probability depending on the specific disorder itself and the way it is inherited) (see [2] for details on inherited conditions). But there are even cases in which motivation is not based on biological but in ethical or religious reasons. Certain families might have serious concerns about going on for abortion of an affected embryo. In such cases, application of PGD may circumvent this kind of ethical conflicts. Applying PGD Broadly speaking, steps for PGD are as follows (2):
PGD vs. PGS Preimplantation genetic screening (PGS) is the general term for a compound of approaches that aim to evaluate the genetic content of the cell, in contrast to genetic tests whose goals are to determine whether an embryo is affected by a specific genetic condition (PGD). Originally termed PGD-AS (preimplantation genetic diagnosis for aneuploidy screening), PGS was developed to confirm the ploidy status of the embryo, searching for possible aneuploidies. Available data suggest that most of miscarriages occurred during the first trimester are a consequence of some sort of aneuploidies (5), and that mainly selected chromosomes were involved in these structural abnormalities (6). Thus, the main approach developed for PGS was the fluorescence in situ hybridization (FISH) for such chromosomes. Types of approaches for PGD in the laboratory Current technical methodologies for preimplantational genetic analyses mainly lie in one of the following:
WHEN TO PERFORM BIOPSY Typical biopsies for PGD (and PGS) are as described as follows:
DAY 3. CLEAVAGE STAGE BIOPSY There is a controversy regarding utility of this type of biopsy. In the cleavage stage biopsy, embryos are biopsied at day 3 when individual cells can be differentiated. This technique entails aspiration of one to two blastomeres to obtain the embryonic genetic material for PGD analysis (13). Following genetic diagnosis, embryo transfer may be performed on blastocyst stage. Embryos are usually selected for biopsy based on morphological criteria. Unfortunately, these do not predict the development potential of the embryo, and so it could fail to progress until blastocyst stage. This would compromise the advantages of using the day-3 approach (14). On the other hand, performing biopsy on the cleavage stage allows embryos to be cultured in vitro until they reach the blastocyst. This means they can be fresh transferred (15), whereas embryos biopsied on day 5 must be vitrified and transferred in a subsequent cycle. How many cells should be removed? The number of cells to be removed in the biopsy is still a controversial issue. Aspirating one cell reduces the cellular mass extracted but it can imply the presence of mosaicism. Conversely, aspirating two cells can reduce the risk of mosaicism, but removing such cellular mass could have consequences on the implantation rate (14). Reported data have shown a dramatic reduction of 39% in the implantation rate in cleavage stage biopsy (16). The authors related it with proportion of the embryo total cellular removed. Whereas around five cells pulled out of the embryo in the trophectoderm biopsy represent 2-3% of the total cell content (expanded blastocyst has 200-220 cells approximately), extraction of a single cell from an eight cell embryo supposes 13% of the total content (16). What do experts say? Cleavage stage biopsy produces different opinions among embryologists because of the presence of mosaicism and the possibility of self-correction of aneuploidies from cleavage to blastocyst stage (17). On the contrary, studies using array-comparative genomic hybridization (array-CGH) technology to analyse genetic abnormalities in day-3 blastomeres and confirming it in trophectoderm biopsy showed concordance between day 3 diagnosis and day 5 reanalysis; Treff and coauthors showed more reliable results for SNP-microarray (96% vs. 83%) and also a lower mosaicism degree (31%) for SNP-microarray samples in a study comparing array technology versus FISH technique (18). These data would support the suggestion of some authors, who proposed that the incidence of mosaicism may have been overestimated in previous studies due to technical inconsistency of the FISH technique (17, 18, 19). At present, this matter remains controversial. Regarding pregnancy rates, in both types of biopsies higher pregnancy rates are obtained comparing with the control group, in which no biopsy was performed (14, 19). To sum up: DAY 5. TROPHECTODERM BIOPSY The blastocyst stage is currently supposed to be an optimal time to perform biopsies for PGD/PGS. The combination of improved blastocyst culture, trophectoderm (TE) biopsy, refined cryopreservation techniques, and molecular assays, such as array comparative genomic hybridization that allows for 24-chromosome screening, have led to a renaissance of PGS. TE biopsy will not detect every circumstance in which the embryo is at risk of aneuploidy, but it will detect mosaicism more reliably than cleavage-stage biopsy (which cannot be relied on at all for this purpose) (20, 21). Moreover, when diagnosing monogenic disorders in single blastomere cells using PCR-based protocols, there is a high risk of PCR failure due to either no amplification (allele dropout) or preferential amplification of one of the alleles, potentially resulting in a reduced number of unaffected embryos available for transfer. Increasing the amount of starting DNA template should in principle increase the sensitivity and reliability of genetic diagnosis. Therefore, the biopsy of multiple trophectoderm cells from the blastocyst rather than a single cell from cleavage stage embryos should potentially lead to improved PGD outcome for patients (14). How many cells should be removed? Research to determine the appropriate number of biopsied TE cells in blastocyst biopsies are limited. The exact number of biopsied TE cells is hard to count visually because cells are small and usually remain as a clump. In most studies using comparative genome hybridization or single-nucleotide polymorphism array technology for genetic testing, biopsied TE cells were used for genome amplification and their number was impossible to know. Moreover, some studies showed that removing four to five cells leads to better results. Therefore, the biopsied cell number should be higher in the blastocysts with better TE quality than those with worse characteristics (22, 23). Can biopsies affect blastocyst development and its implantation? Whereas it remains possible that biopsy of cleavage-stage embryos can critically arrest further development through reduction of cell mass, the low miscarriage rates and high term birth rates in the present series, as well as data presently under analysis, suggest that this is not the case for TE biopsy. It can be speculated that the damage to blastocyst development potential caused by TE biopsy would be less for blastocysts with a greater number of TE cells (21, 22). Some experts assured that TE biopsy at the blastocyst stage had no meaningful impact on the developmental competence of the embryo as measured by implantation and delivery rates. This contrasts with the information above-mentioned on the significant reduction in the probability for an embryo to implant and progress up to delivery (16). When combined with TE biopsy and blastocyst vitrification, SNP microarray has resulted in high implantation and low miscarriage rates for some IVF patients (15, 16, 24). Are there any limitations? Owing to the limitations of genetic analysis, most of the biopsied blastocysts need to be cryopreserved by vitrification, and blastocysts with normal results would be transferred in the next frozen cycle. In addition, biopsy of numerous cells from blastocysts with grade B or C may cause damage to the embryo, leading to either its arrest or implantation failure. However, 1-5 cells may be the appropriate biopsied TE cell number to maintain the implantation potential (15, 22). Also, the personnel experience of different embryologists is an influencing factor in this technique. The number of biopsied cells in the blastocyst biopsy is hard to quantify and largely dependent on the experience of embryologist (22). To sum up: WHAT CAN WE CONCLUDE? The availability of new embryology and molecular techniques allow preimplantation genetic diagnosis laboratories to offer patients at genetic risk the transfer of developmentally competent embryos, unaffected by genetic disease. Cleavage stage biopsy allows for fresh embryo transfer after genetic diagnosis. However, there are reports of high levels of mosaicism when the biopsy is performed on day 3. Trophectoderm biopsy, in turn, provides sufficient material for an effective and more reliable diagnosis in embryos compared to those on cleavage stage. Moreover, it seems that it does not compromise embryo implantation and pregnancy rates in PGD cycles. The drawback for this option is the usual need for cryopreservation and transfer in a different cycle. The offer of PGD in fertility centres has increased over the last decade, primarily due to the progress on the application of diagnostic methods. The choice for either development stage relates to successful outcomes in the clinic, which mainly depend on technical challenges and timing of the developing embryo. For the embryologists, both day-3 and day-5 approaches are supported by evidence, but it will be essential to consider every single aspect of them to evaluate the best option for the laboratory. REFERENCES:
Authors: Shuyana Deba, Isabel Sánchez and Sara Sanz Every procedure carried out in an IVF laboratory, from ovarian puncture and semen capacitation to the embryo transfer, must be performed under specific safety conditions. These standards must be followed to avoid a decrease in gamete/embryo viability (2). From the in vitro culture, gametes and embryos are exposed to diverse artificial situations that do not take place in nature. In vivo, both fertilization and embryo development in the female reproductive tract occur in the complete absence of light. In this environment, other conditions include oxygen (O2) concentration of 2-8%, pH 7.2-7.4, 37⁰C and gradients of diverse nutrients to which the embryo is exposed (3). Changes in temperature, CO2 and O2 pressure, light exposure or volatile organic compounds may adversely affect embryo quality. Also, manipulation of embryos by embryologists is as important as air quality and culture conditions. Each human being is covered by about 1012 bacteria (4), which could contaminate embryo cultures if embryologists do not use the necessary clothing, such as laboratory cap, footwear and mask. Therefore, a daily quality control at different levels should be carried out to obtain good results in IVF cycles. We are going to analyze the effect of some of the elements that can affect germ cells and embryos. LIGHT EFFECT In vivo, mammalian germ cells and embryos are not exposed to light, which might explain why they do not exhibit any protection mechanism against this factor (5,6). In addition, sperm do not have the capacity to repair DNA, unlike oocytes and embryos, which do present some mechanisms for DNA repair (7). Light variables to be considered are intensity, duration and wavelength. It seems clear that photooxidation increases along with light intensity and duration. However, what type of wavelength would be the most harmful for embryos and germ cells? Energy increases when wavelength is shorter (8). Accordingly, artificial cool white fluorescent light has been demonstrated as the most stressful in mouse embryos. Incandescent light, in turn, seems to be less harmful, and the best outcomes are achieved when warm white fluorescent light is used (7). How can light affect the quality of these cells? Indirect effect: Culture and oil photooxidation can affect embryo development (8). In this case, modified components will damage the lipid membranes. Also, if HEPES- or riboflavin-containing media is exposed to light, it results in the formation of hydrogen peroxide, a highly cytotoxic substance (9). Additionally, light can heat up both the plasticware and the oil, resulting in more toxic and damaging components (8,9). Direct effect: Light can potentially compromise the quality of gametes and embryos, by activating stress-related genes or by ionisation, which may also damage the DNA. This phenomenon would cause DNA fragmentation and mutation, as well as an increase in the apoptotic index and change in the number of mitochondria levels (10). How can we avoid this effect? (7) 1) Reducing the exposure time. 2) Using warm white fluorescent light in the lab and green filters on microscopes. 3) Adding antioxidants in the media in order to mitigate damages from ROS. 4) Avoiding riboflavin, which is responsible of the phototoxicity in the media. VOLATILE PARTICLES EFFECT Since the 1990s, IVF laboratory indoor air quality has taken a high relevance. Thence, focused on creating an optimal environment, laboratories have become clean rooms where filtration of particles is performed by using high-efficiency particle arresting (HEPA) filters, and successful chemical air filtration is achieved by removing volatile organic compounds (VOCs) with solid-phase filtration (e.g., potassium permanganate-impregnated, carbon filters) (11). Focusing on VOCs (hydrocarbon-based compounds that are emitted by industries, cleaning products, computers, and microscopes among others), several studies have demonstrated their harmful effect on embryos, initially reported by Boone laboratory on mouse embryo development (11). Moreover, VOCs have been shown to increase DNA fragmentation in human sperm, and they can also have detrimental effects on pregnancy rates (12). Recently, a retrospective study by Munch et al. concluded that, without solid carbon filtration, fertilization, cleavage, and blastocyst conversion rates declined in fresh IVF cycles. Even more, results were found to be even worse in ICSI cycles, probably due to the lack of protective barrier provided by the cumulus cells (13). However, the authors did not observe the same results when embryos had been cryopreserved in an environment with carbon solid filtration but thawed in a laboratory deprived of such systems. The absence of significant changes in cleavage and blastocyst conversion rates, as well as in the proportion of good quality blastocyst developed after thawing suggests that embryos are affected in the peri-fertilization period (13). Also, products like cosmetics emit VOCs, especially perfumes, colognes, and deodorants. They are highly toxic to embryo development in vitro, primarily due to evaporation of their solvent bases (14,15). After analyzing the results of studies determining the toxicity of VOCs, ideal levels should be below 0.2 ppm but preferably zero (12). Personnel must understand the principles of air quality control, including the function of airflows and airlocks, hygiene, dress code and the use of cleaning agents (16). pH EFFECT pH level depends on bicarbonate concentration of culture media and the CO2 concentration of the incubator. However, other factors like altitude and composition of culture media could affect the pH level, too (17,18). Embryos are able to develop over a range of media pH, considering that they possess an intracellular mechanism to regulate its internal level (17,18). However, it is important to control pH variations because they affect development (17). To control pH level outside the incubator some culture media contain buffers like HEPES or MOPS, but long exposure of embryos to these buffers is not recommended (17). Thawed denuded oocytes and embryos are specially sensitive to pH variations because they do not have an inner system to regulate pH (17). So, an increase in the pH of the medium can affect the physiology and development of oocytes and embryos. Thus, acidification of the medium can even affect the fetal weight and size (18). CO2 EFFECT As previously mentioned, CO2 is necessary to control pH level of culture media (17,18). The importance of CO2 was demonstrated in 1985, in a study carried out on hamsters (19). The authors cultured hamster embryos in different CO2 concentrations (5% and 10%). They found a higher rate of blastocysts in those cultured at 10% compared to 5%, which demonstrated differences in embryonic development. This results showed that CO2 level is an important factor for embryo culture (19). The capacity of CO2 to get through cell membranes allows for regulation of the inner pH levels in blastomeres. In other studies, it has been shown that the required CO2 concentration to achieve the optimal pH varies in different species. For instance, the required CO2 level in rats is 7.5%, whereas for humans it is 6.5% (19). O2 EFFECT Some studies have compared different values of O2 concentration in the incubator and they show that a low level (5-6%) improves results when compared to an ambient level (21%). It has been shown that low O2 levels increase implantation, pregnancy and live birth rates (17,20). It seems that a low O2 level reduces ROS in the culture and the presence of volatile particles in the air, although the exact mechanism of action is still unknown (17). TEMPERATURE EFFECT Standard temperature generally used in IVF laboratories is 37⁰C (17,18). However, optimal temperature is unknown because in the female reproductive tract it could be slightly lower, about 36⁰C. On average, temperature of the Fallopian tube is about 1.5⁰C less, whereas the follicular liquid temperature can reach 2-3⁰C lower than core body temperature (17). It is important to control and prevent temperature variations because it can affect meiotic spindle stability and alter embryonic metabolism. It has been shown that an increase of 2⁰C during 20 minutes potentially alters the integrity of the meiotic spindle, which cannot be completely repaired when temperature is set back to 37⁰C. As a consequence of this increase in temperature, embryos express some stress-response genes that compromise development (18). Interestingly, a small decrease in temperature does not have any effect on oocytes, whereas a large difference can be severely harmful for the meiotic spindle (18). CULTURE MEDIA EFFECT Nowadays, there exist two kinds of culture media: one-step media and sequential media (with different compositions for days 0-3 and 3-6) (17,21). All culture media are similar in composition; they contain energy substrates like glucose, pyruvate or lactate, and both organic and inorganic salts, which must be balanced accordingly. Culture media also contain amino acids in different proportions. The exact composition of amino acids in culture media is unknown. One of the most important problems related to the presence of amino acids is the ammonium generated as a product of metabolism. Ammonium has negative effects on embryo and fetal development. To avoid this problem, some culture media contain glutamine, which reduces ammonium production (17,21,22). Also, culture media can be supplemented with macromolecules and other components like HSA, α and β globulins, growth factors, vitamins, lipids, nucleotides, cytokines and hormones (17,22). What can we conclude? There are many parameters that should be kept in mind in order to maintain the optimal conditions for both gamete and embryo development in an IVF laboratory. In vitro, cells and embryos are exposed to different stress situations that must be minimized. Therefore, a routine control at different levels needs to be performed, so that the environment in the laboratory is adapted to resemble the reproductive tract and the intrauterine conditions. REFERENCES:
Authors: Paula Brígido, Shuyana Deba, Javier Del Río and Isabel Sánchez Implantation is one of the key steps in human reproduction, and hatching of the blastocyst is a critical point in the sequence of physiological events that lead to implantation (2). It has been estimated that only 15-20% of embryo transfers culminate in implantation, and as a consequence clinical pregnancy and live birth rates are quite low (3, 4). The potential of an embryo to implant is related to its own nature, the quality of gametes and the endometrial receptivity. But failure in blastocyst hatching is also an important factor that prevents implantation (2, 3, 4). With the aim of solving this problem and in order to improve both implantation and pregnancy rates after Assisted Reproductive Technology (ART) procedures, scientists developed assisted hatching (AH) (2, 3, 4). AH consists of an artificial alteration of the zona pellucida (ZP) either by slimming or breakage. This technique was first described on 1988 by Cohen et al., who reported the first pregnancy after AH (4), and successive works have since shown its efficiency (2, 3). Because AH does not seem to present clear advantages to all patients, it should not be applied, in principle, to all of them as a rule (3). Some studies have shown that AH has effectively improved ART outcomes in patients with bad prognosis, like those with a history of 2+ implantation failures, bad embryo quality, aged patients (38+), patient with frozen embryos or those who have oocytes with a thick ZP (2, 3, 4, 6). ART have some actual consequences on gametes and embryos, one of them being hardening of the ZP by the use of certain culture media or by cryopreservation. These and other negative effects may hamper blastocyst hatching, which might be solved through AH. Overall, this approach might be useful considering the early implantation window in women treated with exogenous gonadotrophin stimulation, compared to a natural cycle. Additionally, benefits from AH include the possibility of ZP breakage improving the embryo-endometrium communication (5). In order to help the embryo hatch through the ZP before implantation, a variety of techniques over the years have been developed (7). As a commonality for all of them, it is important to minimize the time of the embryo being outside the incubator and to optimize the methodologies to reduce both pH and temperature variations (3). MECHANICAL ASSISTED HATCHING (PARTIAL ZONA DISSECTION) This method does not require any new equipment, and it can be easily performed by an embryologist handling a micromanipulator (8). During this procedure, the embryo is held by the suction exerted by the holding pipette, while the microneedle is passed through the zona pellucida at the largest visible portion of the perivitelline space and introduced in a tangential trajectory to the embryo. Then, in order to tear the ZP apart, the embryo is released from the holding pipette and rubbed against it (7). However, one of the main disadvantages of this technique is the difficulty of creating a hole of significant size without abruptly damaging the embryo. Moreover, it is not a fast procedure, which means embryos need to remain outside the incubator for a long time, and so this may affect development of the transferred embryos (7, 9). ACID TYRODE'S ASSISTED HATCHING For this procedure, first the embryo is stabilized with the holding pipette. Second, a pipette containing acid Tyrode is approached to the embryo, oriented at the 3 o`clock position, next to an area of empty perivitelline space. Finally, the acidic solution is gently expelled over a small area (~30 µm Ø) and immediately washed away (7). This technique allows the formation of a hole of significant size; nevertheless, the exposure to acid is problematic, since this compound may be potentially embryotoxic. Furthermore, this technique requires practice and expertise from the operator (7, 10). LASER-ASSISTED HATCHING Laser-assisted hatching (LAH) is designed for easy positioning of the embryos, focus and shooting. It can be executed with a single click of the mouse controller. The diameter of the drilled holes vary between 5 and 10 µm according to how many shots are performed and the irradiation time (7). From the technical point of view, this method is easier, better controlled and more precise. Without physical contact with other embryos, the procedure can be completed faster than other methods. So, time of the embryos out of the incubator is shorter than for other techniques (9). Even though the equipment may result expensive, the laser-assisted technique seems to represent the lowest potential risk for the embryo, and it is relatively simple to perform with consistency between operators (3). EFFICACY OF AH Although it has been performed for more than 20 years, up to date results are still inconclusive. Ma and co-authors performed one of the first trials to determine the overall effect of this technique. They concluded that implantation rates could be enhanced by performing ICSI along with AH, but differences were not statistically significant (11). Two recent meta-analyses evaluating potential benefits of this technique have reported significant heterogeneity among results (12, 13), suggesting that effects of AH may differ depending on specific patient features (14). Most researchers support the hypothesis that this technique improves clinical pregnancy rates in patients with previous failed IVF cycles or poor prognosis. However, there is insufficient evidence to affirm that AH improves live-birth rates in these populations, and so it remains uncertain whether AH is beneficial to other patients (6, 14, 16, 17). IS THERE AN INCREASE IN MULTIPLE GESTATION RATE? Even though certain reports associate artificial manipulation of the zona pellucida with multiple pregnancies, there is actually insufficient evidence to support an increased risk of monozygotic twinning after AH. In fact, the overall rate of monozygotic twin pregnancy in IVF with AH is less than 1% (6, 16). MISCARRIAGE AND CONGENITAL MALFORMATIONS RATES It has been reported that this technique may enhance implantation of abnormal embryos. Thus, the lower live birth rate observed, which is related to the high number of pregnancies ending in early miscarriage, may be due to chromosomal abnormalities of the embryos (17). However, Ma et al. performed cytogenetic tests on miscarriaged embryos and umbilical cord blood from newborn infants, where they found a similar incidence of major congenital malformations in ICSI-born patients compared to the general population (11), thus ruling out any direct relationship of AH and miscarriage. EFFECTS OF ASSISTED HATCHING ON FROZEN CYCLES Data show that cryopreservation may induce zona hardening as well as advanced female age and in vitro culture conditions (18). The answer to this problem could be AH, but results obtained on implantation and pregnancy rates after AH for frozen embryo transfer cycles are controversial, as well as for fresh IVF cycles (19). These discrepancies may be attributed to the type of AH, the extent of ZP microdissection, the number of patients and criteria for their selection, or even the quality and stage of embryos selected for AH performance. In an earlier study, Primi et al. (2004) were unable to show any specific advantage of LAH in cryopreserved embryos. In this study, no embryo selection was reported, so this could explain why the implantation rates observed were lower than those achieved when embryos were selected (20). Ng et al. (2005) also found a negative effect of LAH on frozen-thawed embryos in their randomised study (implantation rates: 9.0% vs. 12.5%; pregnancy rates: 6.8% vs. 15%). Although a subgroup analysis showed a higher implantation rate, differences were not statistically significant when LAH was performed on embryos with zona thickness of 1.6 mm (21). In contrast, other studies using similar techniques of LAH were able to show improvement in implantation and pregnancy rates in the LAH group. Such was the case in a study by Balaban and co-authors (2006), in which their data revealed that implantation rates (20.1% vs. 9.9%) and pregnancy rates (40.9% vs. 27.3%) were significantly higher in the group in which embryos had been subjected to LAH before transfer, as compared to those from the control group (22). Valojerdi et al. (2008) in turn showed that LAH increased significantly both implantation and pregnancy rates in embryo cryopreservation cycles (23). These conclusions resemble those by Kanyo and co-authors (2016), who compared clinical pregnancy rates after using LAH technique on day-3 frozen-thawed embryos, and found a higher pregnancy rate after applying the LAH procedure (24). CONCLUSIONS Reports on patient populations are found that show benefits from AH, which include patients whose embryos present a thick zona, with elevated FSH, over 38 years old and cryopreserved cycles. It seems that the laser-based method is the most used among professionals and the most effective, although differences between are found between studies. However, no significant data show any increase in clinical pregnancy rates that could be translated into an encouragement of this technique to be routinely performed. It should also be noted that some studies do not include specific important confounding factors, such as patient socioeconomic status, parity, duration of infertility, number of repeated failed cycles, embryo quality and/or smoking and alcohol intake, which might cause residual variance in the obtained results. Additional limitations of the study include the lack of information on the type of assisted hatching (mechanical, chemical, or laser), which may have changed over time or between clinics. To learn more about this topic, visit our ‘VIDEOS’ section, where you will find videos about mechanical, chemical and laser hatching. REFERENCES
Authors: Shuyana Deba, Javier Del Río, Isabel Sánchez and Sara Sanz Fertilization is a sequence of coordinated events that results in the metabolic activation of the ootid (nearly mature oocyte) and triggers cleavage of the zygote (2). Nowadays, in assisted reproduction laboratories cleavage can be evaluated in vitro and in real time. Once in vitro fertilization (IVF) has been accomplished, early development of the embryo can be recorded by using time-lapse systems (TLP) (3). This approach makes it possible to evaluate morphology, including dynamic parameters, based on the uninterrupted culture of the embryo, which also allows for a better embryo selection, thus rising pregnancy rates (4). Even so, there are still clinics all over the world that select embryos for transfer using light microscopy, which means the use of a conventional incubation method (5). CRITERIA FOLLOWED FOR EMBRYO CLASSIFICATION It is known that an international consensus is needed in the way embryos are assessed and described. The following standardized criteria is from Alpha Scientists in Reproductive Medicine and ESHRE Special Interest Group of Embryology, 2011 and includes ‘minimum standards’ for oocyte and embryo morphology scoring (6): the current expected observation for embryo development is 4 cells on day 2 and 8 cells on day 3 after fertilization (day 0). Moreover, embryos with <10% fragmentation, stage-specific cell size and not multinucleated are considered of good quality (6). According to this consensus, scoring for day 4 (morula stage) regards as good embryos those that enter into a fourth round of cleavage, which implies evidences of compaction that virtually involve the whole volume of the embryo (6). Finally, on day 5 blastocysts are to be observed expanded with: a prominent inner cell mass (ICM) consisting of many cells, compacted and tightly adhered together; and a trophectoderm (TE), forming a cohesive epithelium (6). Nevertheless, these parameters do not restrict laboratories to annotate further observations in order to select the best embryo for transfer (6). BEST DAY TO PERFORM EMBRYO TRANSFER One of the most important aspects that influence the success of ART is embryo transfer from the culture medium to the uterus (7). This has been a controversial subject that still generates quite some doubts. Morphological evaluation of embryos is sometimes a subjective process, and it provides limited information on the possible genetic abnormalities that embryos may have (8). Currently, there exists a great controversy on the optimal moment to carry out embryo transfer.
IN WHICH CASE DOES THIS TRANSFER USUALLY TAKE PLACE? Day 2 transfer is usually indicated in cases of poorly responding patients. Indeed, it is also indicated when the sperm, oocyte and/or embryos are also of low quality and/or number (9, 10, 11). WHAT DO EXPERTS SAY? Several retrospective studies have compared embryo transfer on day 2, day 3 and day 5 after oocyte recovery, all of which presented conflicting results. A study performed by Mahdavi et al. among poor responder patients revealed no clinical differences between day-2 and -3 embryo transfer (10). However, this study found that pregnancy rates per oocyte retrieval and embryo transfer were significantly higher in the day-2 embryo transfer group compared to day 3 group. It is worth mentioning that other investigators did not find significant differences in pregnancy outcomes when they compared embryo transfer on day 2 and day 3 (11, 12). Additional results from other studies have revealed higher clinical and ongoing pregnancy rates after embryo transfer on day 2 than on day 3 in poor responders. This suggests that the occurrence of miscarriage can be reduced by restricting embryo culture to only 2 days, which could also provide an alternative for managing poorly responding patients (11). That is the reason why embryo transfer on day 2 is still performed in many IVF centres; there is an actual risk of compromising the viability of embryos by prolonged in vitro culture in sub-optimal conditions, with an increased risk of obtaining no blastocysts to replace on day 5 (9, 13, 14). Even though there seems to be a large number of benefits for these patients, certain disadvantages that may potentially occur must also be taken into account, as it can be seen below (Table 1).
PATIENTS IN WHICH TRANSFER D+3 SHOULD BE PERFORMED There exists no criterion to select patients who should be transferred on D+3. Traditionally, embryo transfer has been performed on cleavage stage, so the chosen day was D+3 of embryo development (7). Generally speaking, embryo transfer was carried out on this day in all patients, until a culture medium was developed that allowed to keep embryos in the laboratory for 5-6 days, and with the exception of the cases previously mentioned (11). SCIENTIFIC LITERATURE TO SUPPORT D+3 AS THE BEST DAY FOR EMBRYO TRANSFER Many studies show contradictory results on whether it is better to transfer on D+2 or D+3. However, there are no significant differences as for implantation, clinical pregnancy or live birth rates when comparing transfer on these days. A study by Modares et al. (15) with patients under 40 years old showed a slight improvement in these results when transferring on D+3, although differences were not significant. The authors also showed embryo quality to be worse when the transfer was performed on D+3 than on D+2. Thus, implantation rate has been observed to be higher in D+3, because extending embryo culture for one day allows to discard those embryos that stop their development from D+2 to D+3 (16, 17). Furthermore, it is necessary to consider that there are other external factors that affect embryo development and, consequently, the selection of the best day to transfer. Quinn et al. (18) determined that one of these factors is culture media. Thus, in sub-optimal lab conditions, it would be interesting to transfer on D+2 rather than D+3, in order to spend the shorter time possible in the media. Regarding D+5 transfer, some studies have shown higher implantation rates in embryos transferred on the blastocyst stage compared to those transferred on D+3 (cleavage stage). However, it is necessary to consider that only 25% of embryos reach the blastocyst stage (15); this implies that the number of embryos transferred and vitrified in a cycle is lower than for D+2 and D+3. As a consequence, when considering cumulative pregnancy rates no significant differences are found between transferring on cleavage stage and blastocyst (7). Again, benefits for the patients must be considered along with potential disadvantages (Table 2).
It has been observed that transfer on blastocyst stage helps to improve pregnancy rates and reduce the risk of a multiple pregnancy. Why? One reason might be that there is no method to determine whether embryos that initially seem to be of good quality are likely to develop up to blastocyst (19). WHO ARE THE IDEAL PATIENTS? 1. Those with a large number of embryos (20). 2. Those whose day-3 embryos are of good quality (20). 3. Those in which day-1 embryos exhibit pro-nuclei and present a grading profile (20). 4. Young women with good ovarian response (21). 5. Those whose embryos display an early cleavage (22). POTENTIAL BENEFITS OF BLASTOCYST-STAGE TRANSFER vs. CLEAVAGE-STAGE TRANSFER First of all, the new culture media allow us to perform longer incubations in the laboratory, after which the best embryos can be selected with higher accuracy and with lower risk of aneuploidies (23). Moreover, there will exist a better synchronization between the embryo and the mother. Additionally, uterine contractility decreases during the luteal phase (24, 25). The size of these blastocysts is bigger, so some studies have found fewer cases of ectopic pregnancies in comparison to transfers on day 3 (26). A parallel comparison of benefits vs. disadvantages for this procedure can be seen in below (Table 3). IS IT BETTER TO TRANSFER ON DAY 5 OR ON DAY 6?
The optimal time for embryo transfer depends on a variety of factors, such as the embryo growth speed. However some studies have revealed both implantation and pregnancy rates to be more successful when embryos are transferred on day 5 compared to day 6. This is due to the fact that viability of embryos expanded on day 5 is higher than for those expanded on day 6 (30). In conclusion, it seems difficult to define the most appropriate day for embryo transfer to be applied for each patient. Therefore, every single case must be individually analyzed. In addition, several factors should be taken into account when deciding on the day for embryo transfer, such as maternal age, sperm and oocyte quality or physiological response of the woman and/or the available embryos. By doing so, a good decision can always be made in order to improve implantation and pregnancy rates. REFERENCES
|
Entries
March 2019
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media