|
Authors: Shuyana Deba and Roberto de la Fuente 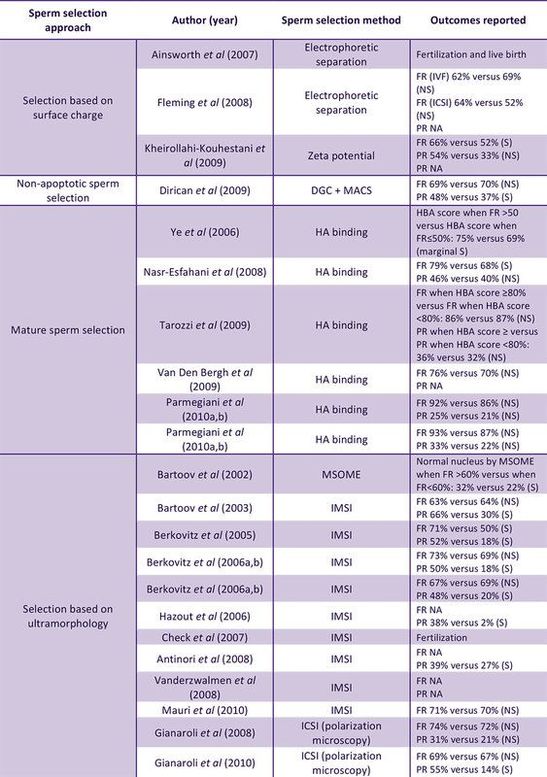 Figure 2. Collection of studies that relate the effects of advanced methods of sperm selection and clinical outcomes (FR=fertilization rate; PR=pregnancy rate). Modified from [11]. *Please consult the original source for references therein. Figure 2. Collection of studies that relate the effects of advanced methods of sperm selection and clinical outcomes (FR=fertilization rate; PR=pregnancy rate). Modified from [11]. *Please consult the original source for references therein. INTRODUCTION In assisted laboratories, semen samples are usually obtained after masturbation and the ejaculate is collected into a container. However, semen recovered from non-spermicidal condoms can be of higher quality because of the time producing the sample influences in the amount of seminal emission before ejaculation (2). Moreover, according to the World Health Organization criteria, laboratory manual for the examination and processing of human semen, the results of laboratory measurements of semen quality will depend on: completed collection of the sample, products of the accessory glands (that will dilute the concentration of epididymal spermatozoa), time past since last sexual activity, abstinence period and the size of the testis. All these factors, among others, will have an influence on the total sperm count per ejaculate (2). Contrary to what is found in natural fertilization after ejaculation, there are no actual barriers to enhance semen capacitation in vitro. As a consequence, in assisted reproduction laboratories sperm must be separated from the seminal plasma within one hour after ejaculation. The purpose of semen processing is to increase the concentration of high quality spermatozoa, and the method choice will depend on the nature of the sperm, that will in turn determine which reproductive procedure will be performed (2). Reports over the last two decades have made emphasis on the need for the optimal method for sperm selection to compile as many as possible of the following conditions: non-toxicity, ease to perform, inexpensiveness, suitability for high-throughput sample processing, accuracy in selecting the best subpopulation of sperm and ability to discard other cell types and seminal plasma as well as other substances that may harm the sperm (cryoprotectants, bioactive complements, motility enhancers) (3). Unfortunately, no known method to date successfully meet all the above, and so the selection of the most appropriate method normally depends on the specific procedure to follow. SPERM PREPARATION “Conventional” techniques like the nowadays common washing swim-up (WSU) and differential density gradient centrifugation (DDGC) procedures are normally used for a variety of assisted reproductive procedures, although it is clear that not all of them will have comparable efficiency in selecting high quality sperm. Some techniques are better indicated for certain conditions. For instance, high concentrations of highly motile sperm from normospermic men will show a good performance after WSU, and this will be suitable for regular IVF. On the contrary, severe oligospermic or asthenospermic individuals will require semen selection by using other techniques. The introduction of ICSI in the regular clinical practice enhanced the fertilization rates in the 90’s (4), but did not significantly increase live birth rates because of failures in early development, mostly due to the high incidence of structural chromosomal aberrations (5). This is a direct consequence of the inability of ICSI to specifically detect good quality spermatozoa. In the following paragraphs we discuss the appropriateness of different techniques for distinct assisted reproduction treatments, namely: in vitro fertilization (IVF), intrauterine insemination (IUI) and intracytoplasmic sperm injection (ICSI). Sperm preparation techniques for IVF In the last few years several studies have discussed about the best technique for sperm capacitation in vitro. Recently, Volpes and collaborators compared the effects of four methods used for sperm preparation, namely, direct swim-up, pellet swim-up, density gradient and density gradient followed by swim-up (6). The authors evaluated levels of DNA fragmentation using the sperm chromatin dispersion test for samples meeting the following criteria: minimum volume of 2 mL, minimum sperm concentration of 10 million/mL and a minimum motility of 35%. The study showed lower DNA fragmentation after pellet swim-up and after density gradient followed by swim-up. However, the study highlighted the limitation that clinical outcomes from IVF/ICSI procedures were not correlated with DNA fragmentation in the sperm (6). A different study compared sperm processing by WSU and DDGC in normospermic individuals, analyzing sperm motility, concentration, and morphology recovery rates (7). Conclusions were that, on the one hand, DGC was appropriate for males with low sperm concentration, since it yielded higher sperm concentration than the WSU technique. On the other hand, the latter facilitated a more efficient morphology-based sperm selection. Nevertheless, the main limitation of the study was the small number of patients and the fact that all of them were normospermic (7). In 2016, Yamanaka and coauthors attempted to determine the efficiency of combining both DGC and WSU techniques in reducing the number of sperm with abnormal nuclear morphology under the microscope (8). Results showed that combination of the two approaches was better than one alone; both DNA fragmentation levels and sperm motility were improved compared to results after just DGC. Data also demonstrated that the combination of both techniques was efficient in enriching the sample with sperm with normal head and flagellum morphology (8). Intrauterine Insemination (IUI) A study by Karamahmutoglu on the most effective sperm preparation technique for IUI compared WSU vs. DGC (9). Even though data showed higher IUI success rates after having performed the DGC approach, no significant difference was found in the "mild male factor" subfertile group (with sperm count in the range of 5-15 million/mL). Moreover, other factors were affecting fecundity success rate, such as female age, number of cycles and type of infertility (9). These observations on the efficacy of DGC and WSU methods for IUI have recently been confirmed in a similar study by Butt and Chohan (10). On the particularities of ICSI Sperm selection for ICSI is commonly carried out by the embryologist’s own judgement based on morphological criteria. This results in inconsistent decision-making and often selection of poor quality sperm, since semen samples are considered morphologically normal with just 4% of normal-looking spermatozoa (2). Therefore functional, physiological and molecular traits of spermatozoa cannot be evaluated by ICSI, and so unnoticed DNA abnormalities (even specific causes of sterility) might be passed on to the offspring by the selected spermatozoon. Thus it is easy to understand why the embryology and reproduction community has been trying to develop new strategies to successfully select the best spermatozoa regarding phenotype, functional characteristics and genetic and molecular integrity. Advanced methods for sperm preparation There are currently three main groups of methods to facilitate selection of high quality sperm, based on morphology, electrical charge and sperm surface maturity and organization (11, 12):
A simpler and cheaper technique uses the electrokinetic potential of the sperm (19); the electric potential difference of the sperm membrane decreases with capacitation, which is used to pipette washed sperm into positively charged tubes, so that negatively charged (mature) sperm can be retrieved afterwards. However, the total sperm recovered by using this technique is low, which represents an important limitation (11).
PS externalization to the outer sperm membrane is a typical apoptotic feature, which allows the cell to bind to magnetic beads conjugated with Annexin-V. This made it possible to develop a magnetic-activated cell sorting system (MACS) (25); the sperm suspension is incubated with the microbeads so that those apoptotic spermatozoa will bind to the beads, which will be subsequently retained within the MACS column in a magnet. Non-apoptotic sperm will then flow freely to be collected. Even though this technique enriches the sample in healthy sperm, it does not discard leukocytes or germ cells, and thus it must be combined with DDGC (26). A variation of the approach known as Annexin V glass wool (annexin V-GW) eliminates potential side effects of free magnetic beads (27), but still needs to be combined with repeated DGC cycles, which is actually not appropriate for oligospermic individuals (11). CONCLUSIONS The approach followed for semen preparation and selection for every patient/couple needs to be chosen upon a series of factors that mainly refer to the cause of infertility. The wide range of situations found regarding this topic makes it necessary to adopt a specific strategy every time. This will ultimately define the treatment to be applied and the techniques to be carried out subsequently. Recent advances have developed new and better ways to detect good quality spermatozoa, minimizing DNA fragmentation and optimizing the rates of good morphology or motility, for instance. However, it is important to remember that there exists no particular strategy that always relates to the optimal clinical outcome. On the contrary, each situation must be considered in the light of the patient’s needs and characteristics, and so the technique for sperm preparation must be chosen accordingly. REFERENCES
2 Comments
Mark Owsman
7/16/2017 18:31:52
Good job!
Reply
Roberto
7/17/2017 03:52:32
Thank you, Mark!
Reply
Leave a Reply. |
Entries
March 2019
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media