|
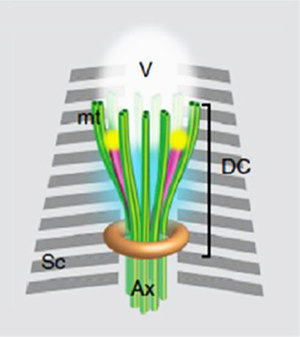
Author: Andrea Núñez “The sperm centrosome contains, in addition to the known centriole, an atypical centriole undiscovered to date” Biologically speaking, human development begins after the joining of a sperm and an egg. Around 100 years ago, the German biologist Theodor Boveri brilliantly summarised the unique contributions of each gamete, stating: “The ripe egg possesses all of the elements necessary for development save an active division-center. The sperm, on the other hand, possesses such a center, but lacks the protoplasmic substratum in which to operate. In this respect the egg and the sperm are complementary structures; their union in syngamy thus restores to each the missing element necessary to further development. Accepting this it follows that the nuclei of the embryo are derived equally from the two parents; the central bodies [centrosomes] are purely of paternal origin; and to this it might be added that the general cytoplasm of the embryo seems to be almost wholly of maternal origin” (1). Since the human oocyte lacks centrioles, it seems that the embryo centrioles are exclusively paternally inherited. 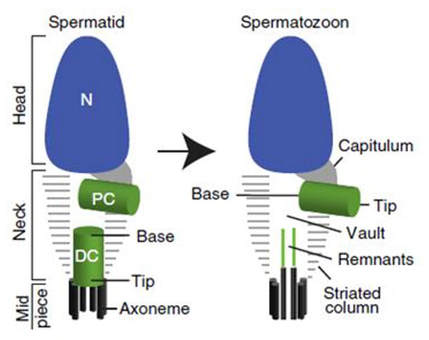 Fig. 1: According to the current dogma, mature sperm cells display a single centriole. The spermatid neck contains a proximal centriole (PC) and a distal centriole (DC). The ejaculated spermatozoon shows a proximal centriole and an empty space, the vault, which marks the location of the degenerated distal centriole. Modified from (2). The current prevailing dogma claims that, during spermatogenesis, the sperm centrosome is remodelled through a process called centrosome reduction, leaving the mature sperm cell with a single centriole left. Throughout this process, the centrioles of immature spermatozoa and their surrounding pericentriolar material (PCM) are modified, resulting in a vault in place of the distal centriole, along with residual microtubules and proteins (Fig. 1). To date, no functions have been associated with those remnants. Accordingly, mature sperm have a single functional centriole that is provided to the oocyte, therefore the zygote inherits only one centriole. The recently-formed embryo needs two centrosomes to give rise to four centrioles, two for each daughter cell. Since new centrioles form by duplication of pre-existing ones, where does the zygote obtain its second centriole? A research group at University of Toledo (Ohio, USA) might have found the answer. In a recent study published in Nature Communications (2), they aim to overturn the old dogma by proposing mature sperm actually carries two centrioles. The authors have demonstrated that the sperm centrosome contains, in addition to the known centriole, a surrounding matrix of pericentriolar material and an atypical centriole undiscovered to date. Using cutting-edge microscopy techniques, it was found that the distal centriole microtubules do not degenerate during spermatogenesis as previously thought. Instead, these are remodelled during spermiogenesis into an atypical centriole. In mature sperm, the distal centriole is found attached to the base of the axoneme, but its microtubules splay outwards. Those splayed microtubules surround previously undescribed rods of centriole luminal proteins, forming a novel atypical structure (Fig. 2). Human sperm cells have been widely studied since 1677, when Antonie van Leeuwenhoek was captivated by his animalcules (3), becoming the first person to see living sperm cells. Thus, it is surprising that this elusive structure has not been discovered until today. Likely, the second centriole was previously overlooked because it is completely different from the known centriole, in terms of protein composition and structure. It may also have been disrupted by the classical chemical fixation methods used for transmission electron microscopy. Unlike previous studies, Fishman et al (2018) used super-resolution microscopy, electron microscopy with high-pressure freezing and correlative light and electron microscopy (2), which allowed them to see proteins at the highest resolution. Despite the structural differences of this newly discovered centriole, results show that it may function similarly and along with the known proximal centriole. Researchers simulated a fertilisation environment without creating embryos, using a cell-free in vitro system that exposed demembranated sperm to egg extracts from Xenopus laevis. This system demonstrated that the human distal centriole is capable of recruiting PCM protein and γ-tubulin, suggesting its competency. To further investigate the action of this structure, researchers used bovine sperm as a model. Bovine sperm has compositional similarities with human sperm, as well as an increased size that allows for the use of certain microscopy techniques that cannot be used on human material. An additional feature that makes it a suitable model is that sperm cells from the bovine species were also thought to contain only the proximal centrosome. However, following the bovine sperm distal centriole into the zygote, the authors found that a new daughter centriole was formed and localised to the spindle pole during mitosis, all while maintaining its attachment to the axoneme. Abnormalities in the formation and function of the distal centriole may be involved in currently-known idiopathic forms of male infertility. As a consequence, a better understanding of this newfound structure may help not only to learn more about early human embryonic development, but also to develop new therapies for related infertility issues. In fact, Dr. Avidor-Reiss and his team are currently planning to take this research to the next level, working with urology colleagues to understand the clinical implications of this atypical centriole. REFERENCES 1. Wilson EB. The Cell in Development and Heredity. 3d ed. New York: Macmillan; 1925. 2. Fishman EL, Jo K, Nguyen QPH, Kong D, Royfman R, Cekic AR, et al. Author Correction: A novel atypical sperm centriole is functional during human fertilization. Nat Commun. 2018;9(1):2800. 3. van Leeuwenhoek A, Dobell C. Antonie Van Leeuwenhoek and His "Little Animals". A collection of the writings of the father of protozoology and bacteriology. New York, Dover Publications Inc.; 1960.
0 Comments
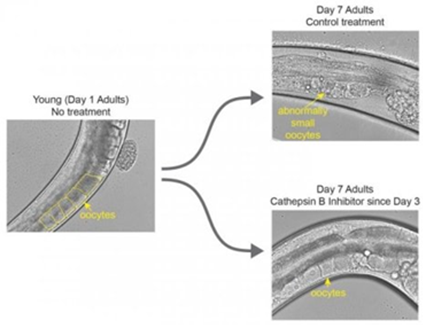
Author: Iris Martínez "In humans, this could translate into a 3- to 6-year extension of female fertility" One of the challenges of modern society is being able to synchronize our own socio-economic context with the biological clock in order to find the most appropriate moment to become parents. Reproductive medicine professionals have been warning about the importance of women awareness about the acute decrease of fertility right before their mid-30s. Should they be fully aware, it would be possible for them to better plan their motherhood by preserving oocytes at young age. However, there are news on this issue: research by Coleen Murphy’s team at Princeton University has reported a protein to be potentially able to extend eggs’ life. This is a key protein present in old and poor-quality oocytes from Caenorhabditis elegans, cathepsin B. In their work, recently published in Current Biology, the authors successfully achieved to extend egg viability longer than their regular life-span, as confirmed by two independent experiments. First, they knocked down cathepsin B-coding genes. The end result was an extension of fertility of the individuals by 10% in time. In humans, this could translate into a 3- to 6-year extension of female fertility. The second approach was to apply a cathepsin B inhibitor at a moment of the worm´s life comparable to the early 30s of a woman. WHY C. elegans? Murphy is professor of Molecular Biology and Director of Paul F. Glenn Laboratories for Aging Research at Princeton. She is specialised in C. elegans, a nematode commonly used as an animal model for a wide variety of studies, including longevity. These tiny animals have several advantages for researchers: not only they present a short life cycle of three weeks, but this species exhibits similar fertility decline towards mid-life, while their oocytes also experiment decrease in quality with age. In addition to this, many of their genes are conserved in humans and, more interestingly, some of them are involved in the regulation of the aging process. One of those genes, with a conserved role in regulation of longevity -and fairly studied by Murphy´s group- is the insulin-like growth factor-1 (onwards, IIS) and its pathway. Even though IIS´s transcriptional targets had been previously characterized, it was not known whether the same mechanisms had any influence on oocyte aging. CATHEPSIN B AND OOCYTE QUALITY EXTENSION When Murphy and colleagues investigated the issue of oocyte aging, they focused on genes and proteins present in higher proportion in healthy and young eggs than in aged ones. But research yielded better results when trying the opposite; this is, by looking at less common proteins in lower-quality oocytes. By doing so, they found a group of proteins absent from high-quality oocytes but abundant in those degraded by age: cathepsin B proteases. In particular, they found cathepsin B protease activity levels in oocytes to be dramatically increased around day 7 of adulthood, which corresponds to mid-life in worms. On the contrary, such levels were low in young adult high-quality oocytes. Cathepsin B knock down If cathepsin B proteases were responsible of oocyte aging and associated to deleterious activity, loss of cathepsin B proteases should lead to decreased oocyte quality damage with aging. Therefore, researchers knocked down individual cathepsin-B genes in adult worms by using iRNA. Surprisingly, those worms had their reproductive life-span increased by 10% compared to wild-type individuals. In addition, aged oocytes exhibited improved morphology. These results point to a deleterious role of cathepsin-B gene in reproductive aging regulation. Cathepsin B inhibitor Once cathepsin B proteases raised as oocyte-aging effectors, Murphy’s team explored pharmacological treatment of aging animals to find out whether the quality of aged oocytes had improved. By treating wild-type adult worms with a potent cell-permeable cathepsin B inhibitor (MDL-28170), the authors showed slowed down decline of oocyte quality over age. Evidences were reduced frequency of cavities between oocytes, misshapen oocytes and abnormally small oocytes in day-5 worms exposed to the inhibitor compared to the control (Figure 1). It is important to highlight that pharmacological inhibition of age-dependent damage in oocyte quality was achieved even when the treatment was applied around mid-life. It suggests that age-related reproductive decline could be pharmacologically treated through mid-life intervention. Figure 1. Image on the left shows a worm at the beginning of its reproductive window with healthy unfertilised eggs, at day 1 of adulthood. The upper-right image represents control cases, in which worms did not receive the treatment by day 7 of their adulthood. Here, eggs are abnormally small and deformed. The lower-right image displays a worm treated with the cathepsin B inhibitor; these individuals show healthy eggs on day 7 (1). WIDER IMPLICATIONS
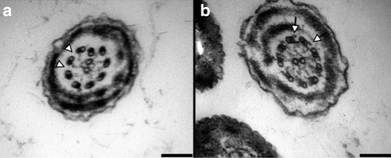
In an interview to Princeton University press, Murphy showed excited about the doors opened by this study. What she likes the most is the possibility of worms to be useful in mammalian research, even though most people think “worms are so different from mammals”. Dr. Murphy and colleagues have set new horizons on interventions to slow down the loss of reproductive aging, although cathepsin B inhibitor is nowhere near ready for testing in humans. However, in Dr. Murphy’s words: “ (...) if you could do something mid-reproduction to improve the rest of reproduction- for me, that’s a game changer”. REFERENCE 1. Templeman N, Luo S, Kaletsky R, Shi C, Ashraf J, Keyes W et al. Insulin Signaling Regulates Oocyte Quality Maintenance with Age via Cathepsin B Activity. Current Biology. 2018;28(5):753-760.e4. Author: Sara Gómez "LAVA is a non-invasive technique that affects neither the sperm membrane nor DNA integrity". 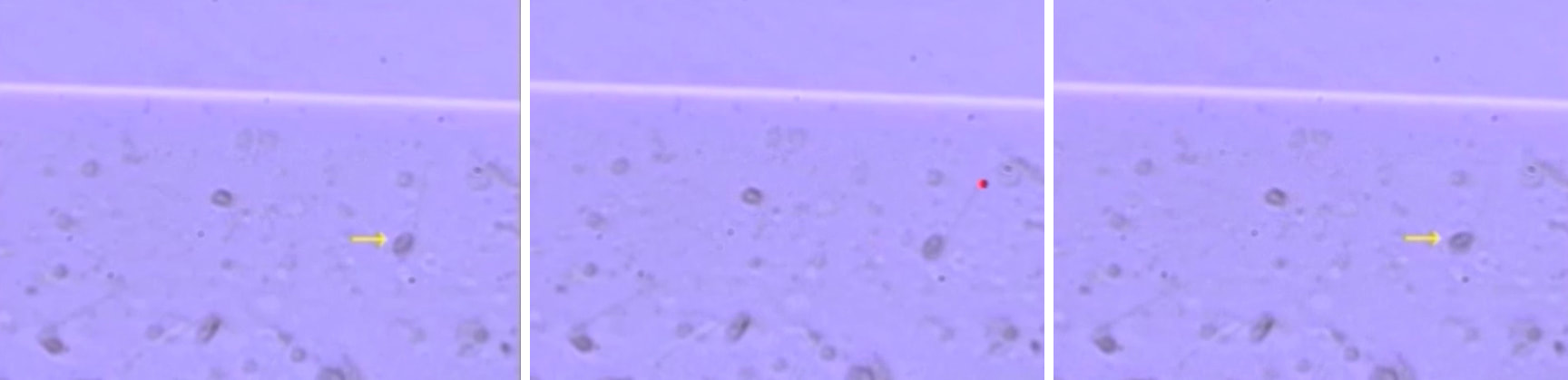 Fig 1: Immotile spermatozoa (yellow arrow) that had responded to the laser shot (red) by either a curling reaction of the tail or sudden head displacement were considered to be likely viable, and thus retrieved for injection into the oocyte. Watch the next video for more information: https://download-tls-cdn.edge-cdn.net/videodb/5501/videodb_5501_77706_10917412_hp.mp4 (1). The ability of sperm to pass through both the uterus and the Fallopian tube and fertilise an egg depends on sperm motility and progression. The flagellum confers motility on the sperm but, sometimes, spermatozoa present an abnormal axoneme ultrastructure and so they cannot swim (asthenozoospermia). The axoneme, the “scaffolding” of the flagellum, is composed of 9+2 microtubules pairs and provided with dynein arms, which conforms the major motor protein and provides flagella with movement. Certain autosomal recessive genetic disorders affect the structure (hence function) of specific motor proteins and, therefore, they result in the impaired action of cilia and flagella. Kartagener syndrome (KS) is caused by different mutations in various genes that encode proteins necessary for ciliary structure and function. Not only these patients suffer from respiratory tract diseases, but also their sperm exhibit an abnormal structure of the axoneme that makes it impossible for the flagellum to beat. Consequently, these patients are infertile. Motility in a sperm sample is one of the markers for viability when it comes to sperm selection in ART. Live and dead sperm are equally subjected to selection in KS patients since both types are immotile, and so the result may end up being unfertilised oocytes or poor quality embryos. In order to avoid such outcomes, there are currently several options to offer to these patients, like promotion of sperm motility, oocyte activation, testis biopsy, hypo-osmotic test, sperm tail flexibility test (STFT) or donor sperm. Nowadays, the most commonly used technique is probably the administration of pentoxifylline, a drug demonstrated to enhance sperm motility (2). However, there are cases in which the patient is resistant to pentoxifylline, and so any effort to improve sperm motility is in vain. What to do in such a situation? In a recent study led by Sinain Ozkavuku at Ankara University School of Medicine (Turkey) (1), a new solution known as LAVA (laser assisted viability assessment) has been proposed. Even though this technique has been known since 2004, when reported by Aktan et al (3), this is the first time it has been applied on a KS patient. LAVA consists in shooting the so-called “end-piece” of the sperm flagellum with a non-contact diode laser pulse of 350 µs with an outcome wavelength of 1480 nm. By doing so, the shot would cause an immediate tail curling or a sudden displacement of the head on a live spermatozoon, whereas dead sperm would remain non-reactive (Fig. 1). This study reports a couple that had gone through 9 years of infertility (no pregnancy). The male partner was diagnosed with Kartagener syndrome, had a history of oligoasthenoteratozoospermia with total immotile sperm and was pentoxifylline-resistant. Different approaches were carried out to confirm the syndrome: transmission electron microscopy (TEM) reported the absence of dynein arms on the axoneme at the ultrastructural level (Fig. 2), and next generation sequencing (NGS) analysis revealed different mutations, all related to KS cases. Moreover, viability test showed 54% of viable sperm (1). Usually, sperm selection for ICSI (intracytoplasmatic sperm injection) in KS or total asthenozoospermic patients is made at random. However, for this study the authors made use of LAVA to select live viable sperm in order to guarantee good-quality embryos and high implantation rates. The ICSI day, 22 mature oocytes were microinjected, resulting in four blastocysts on day 5 and 6 (which indicates a proper timing for embryo development). One 4AB and one 2AA blastocysts (=good-quality embryos) were transferred, but one of them subsequently divided into two embryos, and so three babies were born. Does LAVA represent the next approach in the future for this type of patients? Even though it is certainly possible, this solution is limited to IVF centres that count on laser equipment, and to be used mostly with high-viability sperm samples. Nevertheless, in their study the authors concluded that LAVA allowed for a practical and effective selection of viable spermatozoa, and that negative impact on embryo development or implantation was never observed. Moreover, LAVA is a non-invasive technique that affects neither the sperm membrane nor DNA integrity (4). Despite LAVA's limitations in the clinic as a practical approach, it may increase the chances of having a child for patients suffering from Kartagener syndrome or total sperm immotility. This would certainly bring new hopes to couples who, until recently, had few or no options to become parents. REFERENCES 1. Ozkavukcu S, Celik-Ozenci C, Konuk E, Atabekoglu C. Live birth after Laser Assisted Viability Assessment (LAVA) to detect pentoxifylline resistant ejaculated immotile spermatozoa during ICSI in a couple with male Kartagener’s syndrome. Reproductive Biol Endocrinol. 2018; 16(1):10. 2. Shen MR, Chiang PH, Yang RC, Hong CY, Chen SS. Pentoxifylline stimulates human sperm motility both in vitro and after oral therapy. Br J Clin Pharmacol. 1991; 31(6):711–4. 3. Aktan TM, Montag M, Duman S, Gorkemli H, Rink K, Yurdakul T. Use of a laser to detect viable but immotile spermatozoa. Andrologia. 2004; 36(6):366–9. 4. Ortega C, Verheyen G, Raick D, Camus M, Devroey P, Tournaye H. Absolute asthenozoospermia and ICSI: what are the options? Hum Reprod Update. 2011; 17(5):684–92. Author: Javier del Río “This could eventually help in developing new fertility preservation treatments”. Currently, there are many research lines focused on the improvement of assisted reproduction technologies (ART) and the development of new ones that guarantee new reproductive options for patients. Among the most recent investigated topics there are some worth to mention, like 3D-printed ovaries, stem cells-derived gametes or the use of CRISPR-Cas9 technology for embryos. All these promising approaches have opened new paths of research in ART, too. Recently, a paper has been published reporting functional human eggs grown in the laboratory for the first time. The study was carried out by McLaughlin and colleagues from University of Edinburgh (UK), whose research focuses on the mechanisms of follicle development. As part of the procedure, ovarian samples were collected from 10 women, aged between 25 and 39, who were undergoing elective caesarean section. Tissue pieces that showed no damage or abundant stromal tissue were dissected in the laboratory. A total of 87 fragments with follicles of a mean diameter of 40 μm were cultured for 8 days at 37°C in humidified air with 5% CO2 and renewing half the media every two days. Secondly, the growing follicles (presumably secondary follicles) were mechanically dissected and cultured individually for another 8 days in the same incubation conditions. In the following step, those follicles with visible antral cavities were isolated, from which 48 oocyte-granulosa cell complexes were removed. These were then placed on a membrane with growth-supporting proteins for the next 4 days. A total of 32 complexes containing oocytes >100 μm in diameter showed significant oocyte growth and were selected for in vitro maturation. After 24 hours, only 9 emitted polar bodies and presented cumulus cell expansion. In their paper, the authors point out the larger size of these polar bodies compared to those from in vivo grown oocytes, suggesting the possibility that oocytes may have lost a valuable part of cytoplasm. Finally, the team confirmed the proper organization of the polar bodies and the presence of a meiotic spindle in all 9 oocytes by confocal analysis, which supported the in vitro maturation process. Although complete oocyte growth and maturation had been previously achieved in mice, this is the first time researchers have developed human oocytes all the way from their earliest stages up to the point at which they would be released from the ovaries in vivo. The authors point out that these oocytes were of low quality, which explains their low maturity and the low probabilities of resuming the meiotic division. These results provide proof of concept in developing a complete in vitro growth system to support human oocyte maturation. However, there is a need to optimize each of the stages of development and to enhance our understanding of how culture systems affect the growth process, in particular the epigenetic status of any embryo derived from these eggs. Also, genetic analysis of the obtained oocytes is needed to confirm their healthy condition. Despite these issues, researchers believe that this could represent a new approach for women whose oocytes fail to fully develop in their body or who need to undergo any sort of harmful treatment such as chemotherapy or radiotherapy, which can damage oocytes and cause infertility. It would also be an alternative for girls suffering from cancer who have not yet hit puberty. Because these patients cannot produce mature oocytes, cryopreservation and reintroduction of ovarian tissue may represent a valid option, but they imply a potential risk of reinserting cancer cells, too. In addition, it may be useful for women who experiment premature fertility loss and for low responders, whose follicles do not respond to external stimulation. Potential applications of this technique would aim to women who opt for in vitro fertilization treatments and whose oocytes are too immature to be used. Furthermore, it will help to understand the mechanisms involved in the development of human oocytes. Definitely, there is a chance that the new technique could increase the total number of oocytes available for in vitro fertilization and a woman’s odds of conceiving. Much more work is needed though to make sure the technique is safe before it can be ascertained whether these oocytes remain normal during the process, and if they could actually lead to healthy live births once fertilized. REFERENCES
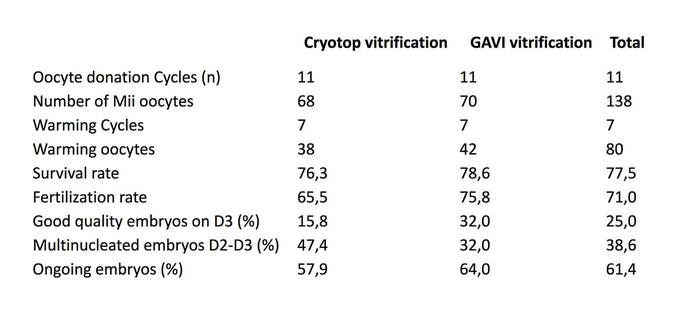
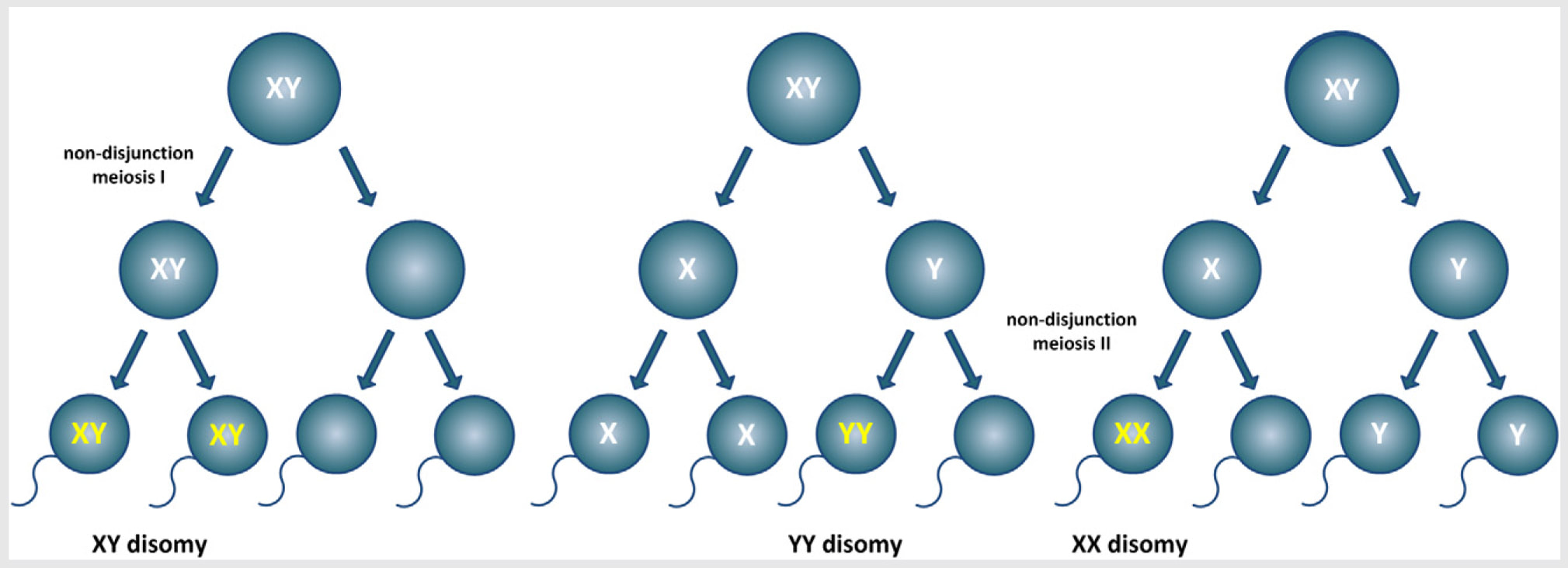
1. McLaughlin M, Albertini DF, Wallace WHB, Anderson R, Telfer E. Metaphase II oocytes from human unilaminar follicles grown in a multi-step culture system. Mol Hum Reprod. 2018;1-8. "The use of automated systems may result in higher quality oocytes". Author: Roberto de la Fuente Image by Johannes Plenio in Unsplash CRYOPRESERVATION AS A MEANS TO PRESERVE FERTILITY Human reproduction has benefited from the possibilities offered by cryopreservation as much as any other discipline, in particular with the latest use of vitrification as opposed to slow freezing techniques (1). The increased efficiency of different techniques has progressively led to a “freeze-all” strategy within last years, which supports an improvement in clinical outcomes. Current social trends imply a significant delay in the age of motherhood. There are a variety of factors to explain such shifts in demographic charts. However, not only social freezing has popularised the use of vitrification for female gametes; the reality is that research on oocytes, donor banks, or fertility preservation treatments in cases of cancer are possible thanks to cryopreservation. Considering the efficiency of vitrification over slow-rate freezing methods when applied to embryos and oocytes (1), it is understandable how technology and research combined succeed in providing new and improved techniques. A good example of this is the development of devices for automated vitrification like GaviTM (2). AUTOMATED OOCYTE VITRIFICATION The basics of vitrification are the combination of an ultra-rapid drop of temperature and the use of high concentrations of cryoprotectants, with the aim of achieving the total elimination of ice crystal formation, both in the inside and the outside of the cell. The volume of the medium in which the oocyte is immersed also needs to be minimized in order to favour the fastest temperature drop. Closed systems (in which the oocyte is not in direct contact with liquid nitrogen) need to make up for the recipient and its volume to avoid further interference in this vein. Prior to the ultra-fast cooling, the oocyte is exposed to different solutions that need to be adequately incorporated (and removed) to safely “equilibrate” the cell while avoiding toxic effects and osmotic shock (1, 3). To date, multiple open and closed systems to cryopreserve oocytes (and embryos) have been designed (4, 5), but all of them require a strict control of incubation times, steady hands and high expertise from the embryologist. However, automated systems allow treating every single cell in the exact same way, thus removing any external variable that may introduce artefactual variation between oocytes, and guaranteeing a consistent vitrification method. Potentially, procedures like this may represent a more efficient approach. EVIDENCES FOR A MORE EFFICIENT VITRIFICATION: AUTOMATED SYSTEMS Recently, the group of Dr. Montserrat Boada from Dexeus Mujer (Barcelona, Spain) has presented original results from an on-going investigation on the fertility potential of automated vitrified oocytes. According to the authors, there is strong evidence to support that the use of automated systems may result in higher quality oocytes than by performing the manual procedures currently standardised in clinics (6). Improved data may be explained by the elimination of inter-oocyte variation, achieved by treating every single cell in exactly the same way; oocytes are first loaded in batches onto specific pods in the device, which automatically adds and removes the cryoprotectant and equilibration solutions. Additionally, the specific design guarantees minimization of the volume, any possible interference with the oocytes is eliminated, and pods are sealed to protect from direct contact with liquid nitrogen (7, 8). These and other features of the system represent a great advance as a whole, given the results achieved by the investigators (2, 6). Oocytes that were automatically vitrified showed a slightly higher survival rate. This correlates with higher quality, even though the authors state that “(…) results were not significant due to the small number of oocytes utilized up to date (…)” (6). Nevertheless, fertilization rates were in fact notably higher than for manually vitrified oocytes when the experimental group was subsequently subjected to ICSI (6). Moreover, specific defects on oocytes were less frequent in those from automated vitrification (Table 1). Altogether, results shown are actually promising, and may point to the right path in the laboratory for future applications. The study has been presented at the recent Congress of the American Society for Reproductive Medicine, held in San Antonio, Texas (US), and at the IX Congress of the ASEBIR in Barcelona (Spain), in which Dr. Miquel Solé, first author of the published study, was awarded the Best Cryobiology Communication Prize. Nonetheless, news got even better; the work of this group has resulted in the first baby in the world born from a (semi-)automatically vitrified oocyte. The achievement took place last September in Italy, thanks to the work done at Dexeus Mujer in Barcelona [click here to read the press release from Dexeus Mujer (in Spanish)]. The baby was born through natural labour and was perfectly healthy. With these data and the proven clinical success, it seems reasonable to think that automation of vitrification arises as a significant progress in assisted reproduction. Nevertheless, further clinical and technological research is needed to keep improving this type of approaches, and only time will tell how much automated procedures can serve assisted reproduction. This article has been selected for publication in the Scientists in Reproductive Technologies (SIRT) Newsletter of The Fertility Society of Australia: DE LA FUENTE, R. (2017) Automated vitrification systems: the future of embryo/oocyte vitrification? Fertility Society of Australia - SIRT Newsletter 4(20): 23-24. REFERENCES 1. Edgar DH, Gook DA. A critical appraisal of cryopreservation (slow cooling versus vitrification) of human oocytes and embryos. Hum Reprod Update. 2012;18(5):536-54. 2. Roy TK, Brandi S, Tappe NM, Bradley CK, Vom E, Henderson C, et al. Embryo vitrification using a novel semi-automated closed system yields in vitro outcomes equivalent to the manual Cryotop method. Hum Reprod. 2014;29(11):2431-8. 3. De Munck N, Vajta G. Safety and efficiency of oocyte vitrification. Cryobiology. 2017. 10.1016/j.cryobiol.2017.07.009. 4. Vajta G, Rienzi L, Ubaldi FM. Open versus closed systems for vitrification of human oocytes and embryos. Reprod Biomed Online. 2015;30:325-33. 5. Momozawa K, Matsuzawa A, Tokunaga Y, Abe S, Koyanagi Y, Kurita M, et al. Efficient vitrification of mouse embryos using the Kitasato Vitrification System as a novel vitrification device. Reprod Biol Endocrinol. 2017;15(1):29. 6. Sole M, Polyzos N, Gonzalez Llagostera C, Carrasco B, Coroleu B, Veiga A, et al. Automatic vs manual vitrification of human oocytes. Preliminary results of the first randomised controlled trial using sibling oocytes. Fertil Steril. 2017;108(3 Supplement):e57. 7. https://www.professionalsinfertility.com/en/our-fertility-technology/gavi.html 8. https://www.geneabiomedx.com/Gavi Fibroblasts derived from trisomic mice can be reprogrammed into diploid iPSCs, differentiate into functional haploid sperm and generate normal fertile mice. Author: Roberto de la Fuente The common sex chromosome dosage in mammals is XX for females and XY for males. This implies that under normal circumstances each one of the X chromosomes in XX females comes from one of the parents, whereas in XY males the only X chromosome is always maternal, because the Y chromosome always comes from the father. Even though both X chromosomes in an XX individual behave as homologs, they may contain different variations of the same genes (alleles). Also, and because they come each from a different parent, they carry epigenetic information known as genetic imprinting, which allows to discriminate one from the other. During spermatogenesis (sperm formation in the testis), an essential step called meiosis takes place. It consists of two rounds of cell divisions without DNA replication in between, separating first homologs and then chromatids, each into a single spermatid (the precursor of the spermatozoon). As a result, gametes with half the number of chromosomes are formed (haploid, as opposed to diploid somatic cells), hence the separation of X and Y chromosomes into different cells. However, errors do occur, and so it happens that sex chromosomes do not always follow their normal behaviour. Instead, there are occasions in which both chromosomes may end up in the same spermatid (Fig. 1). If an XY-bearing spermatozoon fertilizes a normal X-bearing egg, the result is an XXY embryo. This sex chromosome trisomy (SCT) is one of the most common causes of infertility (in addition to other phenotypic traits), and it is known as Klinefelter syndrome. Similarly, other errors leading to nondisjunction of sex chromosomes can result in X0 females (Turner syndrome), XYY males (Jacobs syndrome) or even more complex combinations with multiple sex chromosomes (XXX, XXXX,...). Even though in most cases there are characteristic phenotypes associated to these chromosome abnormalities, mosaicism is also frequent (different cells containing different chromosome dosage), and so affected individuals may too have chromosomally normal children if their contributing gametes happen to be also genetically normal. In a recent study led by sex chromosome expert Dr. James Turner (Francis Crick Institute in London, UK) (2), SCT-infertility has been overcome in mice. Researchers bred XXY and XYY infertile mice and used them to generate a line of cultured fibroblasts (with the original SCT complement), and then reprogrammed these into induced pluripotent stem cells (iPSCs). iPSCs were then differentiated into primordial germ cell-like cells (PGCLC) and later on into functional sperm (Fig. 2). 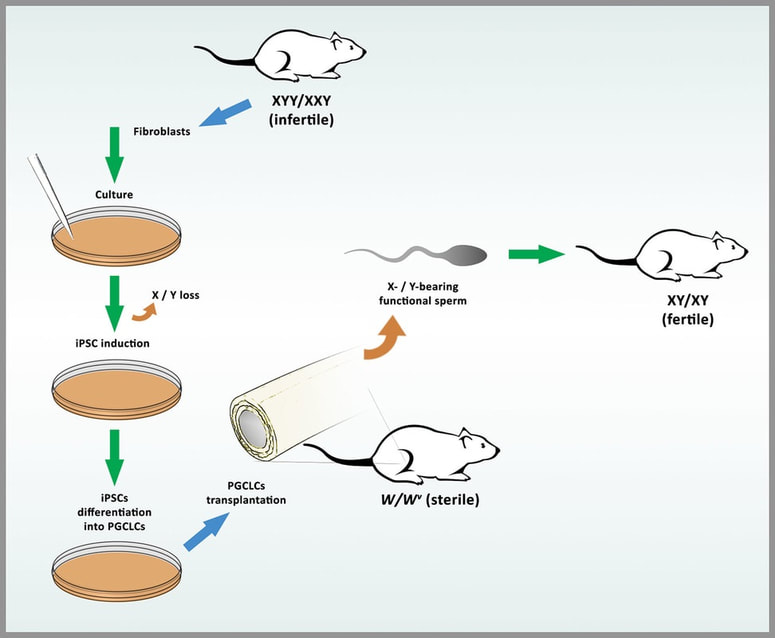 Fig. 2. Illustration of the process detailed in Hirota et al (2017). Fibroblasts from infertile, trisomic XXY and XYY mice are cultured and reprogrammed into iPSCs. During the process, the extra sex chromosome is lost. iPSCs are induced to differentiate into PGCLCs and then transplanted into germ cell-deficient testes from sterile W/Wv mice. Spermatogenesis is fully restored and functional sperm is formed. Normal sperm differentiated from XXY-/XYY-derived iPSCs is then injected into a normal X-bearing egg through ICSI, generating euploid, fertile offspring. Interestingly, both in XXY and XYY iPSCs lines, spontaneous loss of a sex chromosome was observed. In both cases, a high percentage of cells had lost the extra duplicated sex chromosome, leaving the remaining complement as a normal XY. Such loss was also observed in normal XX and XY control lines, yet in a significantly much lower rate. Following chromosome loss back to “normal”, XXY- and XYY-derived iPSC lines were differentiated into male germ cells (PGCLC), which were subsequently transplanted into testes from infertile mice lacking the germline. The result was the restoration of spermatogenesis for all cell lines used. Furthermore, functional sperm originated from these lines was successfully employed to fertilize eggs through ICSI, giving rise to genetically normal embryos and healthy, normal and fertile offspring. The authors also showed promising results in preliminary experiments with human cells, demonstrating that returning cells to their original normal chromosome complement is possible. However, they warn about all risks associated with induced pluripotent stem cell manipulation and transplantation, as well as legal and ethical issues on these matters. This said, it is undeniable that current technologies can help treat specific genetic conditions resulting in infertility, thus giving hope to patients who had found no alternative, so far. The future is becoming present, faster and faster every day. REFERENCES 1. Maiburg M, Repping S, Giltay J. The genetic origin of Klinefelter syndrome and its effect on spermatogenesis. Fertil Steril. 2012;98:253-60. 2. Hirota T, Ohta H, Powell BE, Mahadevaiah SK, Ojarikre OA, Saitou M, et al. Fertile offspring from sterile sex chromosome trisomic mice. Science. 2017;pii: eaam9046. Author: Roberto de la Fuente Genetic modifications are still seen with both excitement and reticence by general population. This poses a challenge, being scientists and media responsible for the information available to the public. Besides GMOs (genetically modified organisms) for industry or food, the last decade has witnessed a growing interest in genetic modifications and genome-editing techniques applicable to humans. So, it is important to note that people who are not specialists in the field have every right to be aware of the progress on these technologies and to have access to breaking-news on the subject.
The latest groundbreaking achievement has been just published online this week in Nature by an international collaboration of different groups (1). The paper reports the “correction” of a mutated gene in human embryos that leads to a severe heart disease in adults. This mutation consists in a four-nucleotide deletion in the MYBPC3 gene, which encodes a protein essential for the proper development and structure of the heart (cardiac myosin-binding protein C or cMyBP-C) that affects contraction and relaxation of the cardiac fibres. Individuals carrying this mutation suffer from hypertrophic cardiomyopathy or HCM, and it is the cause of unusual cardiac growth, arrhythmias and a high percentage of sudden death due to heart failure in otherwise healthy people. Because it is an autosomal dominant mutation, the disease is manifested with just one of the two alleles mutated. Additionally, it is normal that the individual has already fathered offspring when the disease manifests, which implies the mutation has been passed on to the next generation. This is an example of those diseases in which prevention is not yet possible, and the only viable treatments are symptomatic attenuation. Therefore, a current possible approach for preventing transmission of the mutation to the offspring is preimplantation genetic diagnosis (PGD), which allows for discarding affected embryos (theoretically 50% of them because of the dominant inheritance). CRISPR-BASED GENOME-EDITING Scientists found the goldmine for genetic modifications at the beginning of the 21st century with the development of the ZFN (zinc finger nucleases) that led to TALENs (transcription activator-like effector nucleases). These systems gave enormous expectations on the possibilities for editing the genome. A multitude of studies were carried out on these elements in different species, but one of the methodological obstacles was the need to design specific proteins that would recognize specific DNA sequences in each case, which resulted in promising but eventually relatively low efficient approaches in many cases. Moreover, the difficulty for the exonucleases to enter the cell was a problem to deal with and not always appropriately solved. The mechanism of action of the CRISPR-Cas9 system is based on the recognition of specific DNA sequences by a guide RNA, and their cleavage by the Cas9 nuclease. CRISPR stands for clustered regularly interspaced short palindromic repeats. These DNA sequences were originally discovered in the 90's by Dr. Francis Mojica (2), and later developed by other groups into the genetic tool that is nowadays giving so many surprises. The use of this technology allows for modification, repair or substitution of virtually any sequence within the genome. Cleavage sites of DNA by the nuclease are marked as DNA double-strand breaks (DSBs), which are repaired by specific cell mechanisms. One of them is usually the non-homologous end-joining (NHEJ), which essentially restores the DNA double strand. However, this method commonly induces mutations because there is no actual fidelity when it comes to DNA repair, and the result is the introduction of insertions and/or deletions (“indels”), so the DNA sequence is largely altered. From the point of view of therapeutic applications, this mechanism is clearly not an option if the aim is to “fix” the mutation and restore the gene to the original non-mutated form (“wildtype allele”). On the contrary, there is another DNA repair mechanism that the cell can also use, depending on the activation molecular pathway, and the result is the so-called homology-directed repair (HDR), which literally performs a “copy’n’paste” from a template. Thus, using a homologous DNA sequence from an exogenous sample, the mutated gene can be modified; the CRISPR-Cas9 system recognizes the target sequence, cuts it off and replaces it with a new “healthy” sequence using the provided sample as template. Unfortunately, the efficiency of HDR is not high yet, so therapeutic applications do not seem to be currently viable. WHAT DID THE AUTHORS ACTUALLY DO? In the paper, the authors used heterozygous zygotes in which the mutant allele had been contributed by the sperm, while the copy from the oocyte came from healthy donors. Interestingly, DNA breaks introduced in the MYBPC3 paternal gene were mostly repaired by using the oocyte DNA as a template (HDR), which suggested a preferential or specific mechanism of DNA repair in the germline. This may represent a milestone for future approaches in the gene-editing strategy for curing genetic diseases in embryos. Treated embryos later developed into blastocysts with a similar success rate to that from control non-treated embryos, and the overall efficiency rate of gene-editing from the mutated form to the original wildtype form was about 70%. These results seem to be a promising start, but they also mean real therapeutic applications are still far down the road. Also, other problems such as off-target hits (mutations induced by the CRISPR-Cas9 system in different DNA sites) or embryo mosaicism due to gene-editing are an obstacle for the purpose of clinical applications. Thereby, even if these effects are minimized, they still need to be completely eliminated before any similar approach can be brought into the clinic to cure a genetic disease. This is where we are now as for the scientific and physical reality. It is clear that extreme caution needs to be a must in the laboratory when performing these experiments. However, what about ethical concerns? Similarly to other common practices, it is necessary to step back for a minute and think twice whether gene-editing is a good idea, and if so, when and why. We talk here about genetic problem solving, improved gene therapy and definite solutions to currently lethal or sublethal conditions. Again, the idea is that, potentially, any gene could be eventually modified in order to turn a genetically affected individual into a perfectly healthy person. Diabetes, Huntington’s disease, cystic fibrosis, hypertrophic cardiomyopathy... any epidemiologically significant condition could be subjected to removal from a population. That is the current main goal of genome-editing. THE ETHICAL IMPLICATIONS Generally speaking, this sort of treatments and techniques must be performed at some embryo stage, and research on human embryos is forbidden in most countries (meaning forbidden in those countries that have an actual regulation on this issue; there are too many countries that lack the proper regulation to act on these matters, and so they could potentially perform certain kind of operations that might be doubtful from the ethical point of view). The current situation has been catching attention from the media since a group in China published their experiments on human embryos in 2015 (3). Based on the author's claims, research was perfectly legal and it had been approved by the corresponding Chinese authorities. Plus, the embryos used for the experiments were actually not viable (3PN embryos), so there was no issue about any potential future human being. The end point of the Chinese group was to demonstrate that this research was already possible, doable anywhere and by anyone, and so it must be regulated worldwide. Besides other experiments on human somatic line material, genetic modification of human embryos hit the media again in February 2016, when Dr. Kathy Niakan’s group from the Francis Crick Institute in London specifically asked the HFEA (Human Fertility and Embryology Association) for permission to conduct experiments on human embryos. Permission was granted, and so things are since running. What does this mean? For a large part of the scientific community, the Chinese opened the Pandora box. But many others think that paper proofed the inevitable, and the British group took the lead to demonstrate that genetic modification must be studied in humans in order to know exactly what the consequences and limitations are. On these grounds, in September 2015, the Hinxton Group (a compound of specialists in different fields and topics in experimental and social sciences, as well as philosophers and humanists) had previously met in Manchester and had released a statement in which they supported research on human embryos for genetic modification, and of course they also explained why. Essentially, in the text they referred to the needs of understanding the right tools and biological models for research, the consequences and the responsibilities, and they made emphasis on the need and urgency of letting the general population know about genetic modification, genome-editing technologies, reasons and consequences. In 2015, Dr. Jennifer Doudna, world expert on CRISPR systems from University of California, Berkeley, exposed her ideas on human embryo genome-editing (4). A summary of her main points would be as follows: 1) Safety. Need for ensuring efficiency with no off-target hits to reproduce experiments. 2) Communication. Essential to make scientific progress accessible to scientists, public and society to make them aware of the importance of the new and reach properly based opinion. 3) Guidelines. Collaboratively elaborate policies along with scientists and experts for standards and to establish what is ethically acceptable in research. 4) Regulation. Apply those guidelines to appropriately conduct research and look out for the compliance of the agreement. 5) Caution. Technology cannot be applied to routine practice yet (such as for assisted reproduction). WHAT TO EXPECT IN THE FUTURE The research published this week is not the only one on human embryo genetic modification. Different groups keep publishing evidences that show these techniques are working well enough to invest properly and dedicate substantial efforts to improve it and eliminate every single problem mentioned earlier (5). It is certain that many more publications are yet to come, and the latest paper in Nature is only part of the beginning. This publication has once again shaken the scientific community opinion, but favourable reactions have also been immediate. In fact, two important institutions such as the US National Academy of Science and National Academy of Medicine have released their own report as well, backing research on gene-editing of human embryos. Genetic engineering is since many years ago an exciting and still developing field with unimaginable possibilities. We need to step back out of the box, have a look at the big picture and think. Then, work on it, get better on it and become successful in (1) identifying reasons/circumstances to apply genetic modifications, (2) perform genome-editing without error or off-target hits, and (3) explaining why this is important to the general public. Given differences between countries, legislation, lifestyle, economic resources and technological progress, it is likely that the debate will remain open for a long time. But society must be aware of the progress of science, and people need to know the hows and whys of scientific research and how it can affect society. If you want to know more about CRISPR-based technology, do not miss the videos from our section. REFERENCES 1. Ma H, Marti-Gutierrez N, Park SW, Wu J, Lee Y, et al. Correction of a pathogeneic gene mutation in human embryos. Nature. 2017; doi:10.1038/nature23305 2. Mojica, FJM, Díez-Villaseñor C, Soria E, Juez G. Biological significance of a family of regularly spaced repeats in the genomes of Archaea, Bacteria and mitochondria. Mol Microbiol. 2000; 36:244-246 3. Liang P, Xu Y, Zhang X, Ding C, Huang R, et al. CRISPR/Cas9-mediated gene editing in human tripronuclear zygotes. Protein Cell. 2015; 6(5):363-372 4. Doudna J. Editing needs scrutiny. Nature. 2015; 528(7580):S6 5. Tang L, Zeng Y, Du H, Gong M, Peng J, Zhang B, et al. CRISPR/Cas9-mediated gene editing in human zygotes using Cas9 protein. Mol Genet Genomics. 2017; 292(3):525-33. Author: Javier Del Río Riego One of the scientific advances made in recent years is 3D printing of human organs. The creation of an organ from a suitable material often allows medical doctors to decide on the best way to perform a surgical procedure. In addition, it opens up new possibilities for the use of 3D printing technologies in organ transplant programs. Now, researchers from Northwestern University Feinberg School of Medicine and McCormick School of Engineering have developed prosthetic ovaries made of gelatin, which allowed mice to conceive and give birth to healthy offspring. The bioprosthetic ovaries are constructed of 3D-printed scaffolds that house immature eggs, and have been successful in boosting hormone production and restoring fertility in mice. The material used for building up these ovaries is a biological hydrogel made from decomposed collagen naturally found in animal ovaries. This material is degradable to allow cellular remodelling, contains cell adhesion sites and shows soft durable mechanical properties. Scientists built the ovaries by printing various patterns of overlapping gelatin filaments on glass slides. Modulating the distance between these filaments, as well as the advanced angle between layers, allowed for the generation of different pore sizes and geometries. The ovaries are referred to as "bioprosthetic" organs because they contain both living material (ovarian cells) and nonliving material (gelatin). They release hormones in the same way a normal ovary would, allowing the animal to go through its natural cycle, including ovulation. Also, this scaffolding needed to be made of organic materials, stiff enough to be handled during surgery and sufficiently porous to interact naturally with the tissues of the mouse body. In the study, after having placed the biosynthetic ovaries in the mouse, 40-50 follicles were carefully implanted into each scaffold. Vascularization in the ovary is critical because it provides oxygen and nutrients to the follicles, and because it allows follicle-produced hormones to circulate in the blood stream. Since gelatin is a natural material, the body can recognize the implant as a regular body part and allows blood vessels to grow into it. Although mice had been sterilized by the removal of both ovaries, the reproductive tract (including the oviduct), had been left intact to assess fertility through natural mating after the surgical introduction of the 3D-printed organs. The study culminated in the pairing of a dozen female mice, two mice with sham controls (bioprosthetic ovary without follicles) and seven mice with bioprosthetic ovaries were mated with males who had previously sired pups. Three bioprosthetic ovary recipients had litters of one or two pups each, while none of the sham controls had pups. In all cases, the mothers produced milk to feed them. FUTURE APPLICATIONS IN HUMANS Researchers hope in particular to help cancer survivors whose treatments had damaged their ovaries. The 3-D printed bioprosthetic scaffold can be repopulated using patient's previously extracted own follicles or donated samples from ovarian tissues. Another possible use would be helping women with impaired ovaries, so that symptoms of menopause could be alleviated. Rather than using synthetic hormones, which often entail unpleasant side effects, women could have an all-natural source of hormones implanted directly into her body. The team is now preparing a similar trial on pigs, which are closer to humans in size and biology. They believe that it might be a challenge to scale up the 3D-printed structure to the size needed for human use. Original publication: Laronda MM, Rutz AL, Xiao S, Whelan KA, Duncan FE, Roth EW et al. A bioprosthetic ovary created using 3D printed microporous scaffolds restores ovarian function in sterilized mice. Nat Commun. 2017; 8:15261. (1) Available from: http://www.npr.org/sections/health-shots/2017/05/20/528646323/scientists-one-step-closer-to-3-d-printed-ovaries-to-treat-infertility. [Cited 22 May 2017]. Authors: Shuyana Deba Rementeria, Javier Del Río Riego and Sara Sanz Juste Since 2015, the fertility-related company OvaScience from Canada has recently launched a new technique called AUGMENT, which has resulted in high controversy around the world. WHAT IS AUGMENT? It is a new IVF technique designed to improve women’s oocyte quality. How does it work? Through this technique, doctors have to biopsy tissue from the lining ovaries of a woman normally under general anaesthesia, the entire procedure being performed via laparoscopic surgery. Then, samples are transported to the OvaScience laboratory, where the mitochondria from egg precursor cells are isolated and subsequently transferred through ICSI into a mature egg, as well as the sperm. Status: Comercial Serving patients in a total of six countries: Canada, Spain, Japan, Panama, Turkey and United Arab Emirates. What do you think about it? Do you think it is a useful technique? Would you offer this approach at your fertility clinic? You can read more about OvaScience and their treatments in the following links. If you cannot find any paper, let us know and we will send it to you. In the scientific area: Fakih M. The AUGMENTSM Treatment: Physician Reported Outcomes of the Initial Global Patient Experience. Journal of Fertilization: In Vitro - IVF-Worldwide, Reproductive Medicine, Genetics & Stem Cell Biology. 2015;03(03). Couzin-Frankel J. Eggs unlimited. Science. 2015;350(6261):620-624. Imudia A, Wang N, Tanaka Y, White Y, Woods D, Tilly J. Comparative gene expression profiling of adult mouse ovary-derived oogonial stem cells supports a distinct cellular identity. Fertility and Sterility. 2013;100(5):1451-1458.e2. Woods D, Tilly J. Isolation, characterization and propagation of mitotically active germ cells from adult mouse and human ovaries. Nature Protocols. 2013;8(5):966-988. In the media: http://www.ovascience.com/ http://www.dailymail.co.uk/health/article-3414921/Turbocharged-IVF-ll-help-older-women-conceive-Cells-taken-parts-ovary-boost-poor-quality-eggs-leades-greater-chance-pregnancy.html http://time.com/3849127/baby-stem-cells-augment-ivf/ |
Entries
October 2018
Categories
All
Join our mailing list and keep yourself up to date!
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media