|
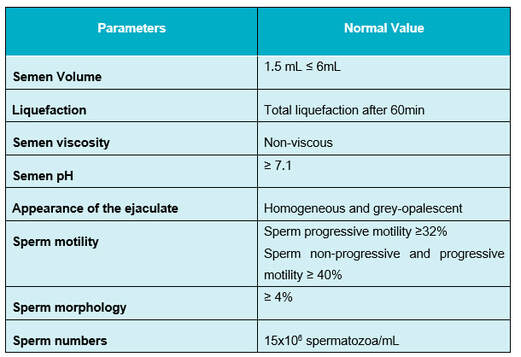
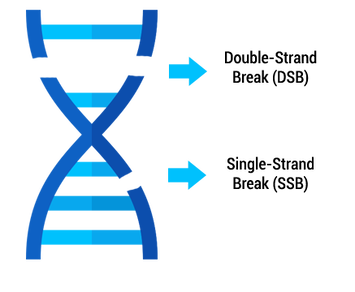
Author: Sara Gómez García “The objective of assisted reproductive technology is to bring home a healthy child, therefore, it is necessary to successfully carry out techniques such as sperm DNA fragmentation (SDF), when needed, in order to accomplish this goal” The goal of assisted reproductive technology (ART) must be to deliver a healthy baby and, thus, it is necessary to select the best gametes. In this scenario, male factor contributes to approximately 40-50% of patients pursuing ART (1). Semen analyses are needed to evaluate male infertility. However, about 15% of infertile men present sperm parameters regarded as normal (Table 1), and so these standard measures alone are insufficient in predicting male fertility potential. Therefore, additional tests become necessary (2). The genetic information of the newborn is the result of the conjunction of both oocyte and sperm DNA. This should be intact, as any damage on the DNA may affect embryonic development. Consequently, sperm DNA fragmentation (SDF) has been under the scope for some time with increasing interest. This factor has been related to reduced fertilisation and pregnancy rates, abnormal embryonic development and increased risks of miscarriage and of neonatal disease in the offspring (2, 4, 5, 6, 7). Furthermore, high SDF is associated to abnormalities in seminal parameters like concentration, morphology and motility, as well as genetic abnormalities in embryos. Nevertheless, it is estimated that around 25-40% of seminal samples with normal parameters present a high DNA fragmentation index (DFI) (2, 5). WHAT IS SPERM FRAGMENTATION? Deoxyribonucleic acid (DNA) is the complex molecule that contains the genetic information, and it is composed by two long joined chains. Small breaks between the chains are present in some sperm cells, which could produce DNA mutations. Different effects are obtained depending on the type of fragmentation present (8) (Figure 2):
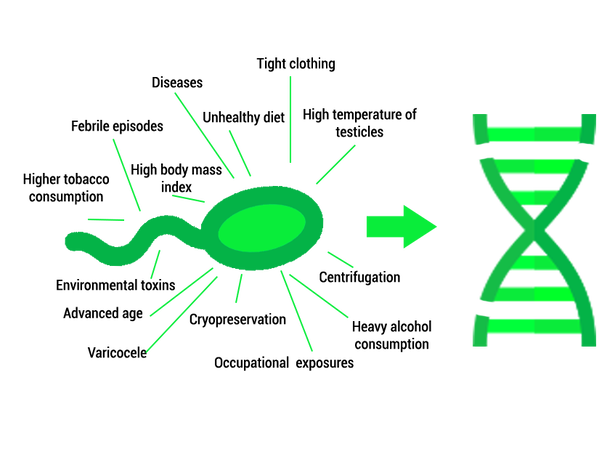
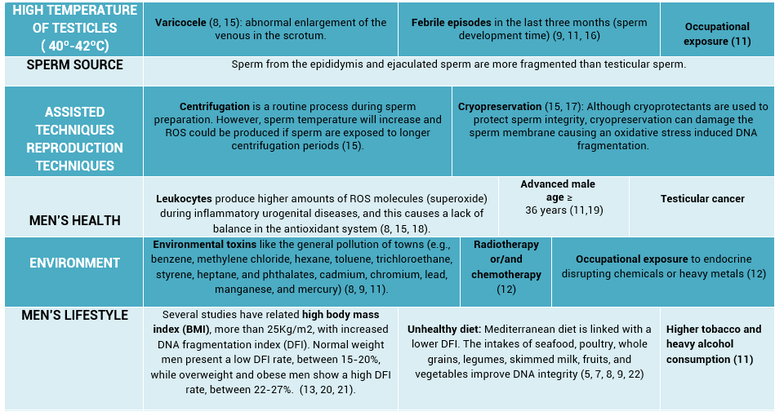
HOW DOES SDF OCCUR? There are mainly two ways in which DNA strand breaks could occur (9, 10): 1. Errors during spermatogenesis Under normal circumstances, DNA nicks are generated and they are repaired during sperm maturation. If repair is impaired, sperm DNA can remain fragmented. In addition, aberrations in sperm development could be produced, which could make sperm DNA more vulnerable (7, 8, 11). 2. Oxidative Stress Reactive Oxygen Species (ROS) are metabolites of oxygen and nitrogen, among others, and are highly reactive in that they readily oxidize other molecules. These metabolites are produced by sperm or external factors, and even though they play physiological roles in supporting sperm function, an imbalance in those levels may lead to DNA damage. Sperm contain antioxidant mechanisms to regulate this balance; however, an excess of ROS or defects in the antioxidant system can result in DNA damage. This process is associated with SSBs (8). Even though oocytes can repair sperm DNA damage to some extent, it remains unclear whether they are able to successfully repair DSBs (7, 11). In addition, this capacity depends on the oocytes’ age, quality and external factors. Aged oocytes exhibit an inefficient and imperfect repair mechanism, thus such oocytes are unable to repair sperm DNA fragmentation. WHAT ARE THE SOURCES OF SDF AND THE CURRENT INDICATIONS TO CARRY OUT SFD TEST? Several studies suggest that SDF could have a variety of origins and, therefore, sperm DNA could be subjected to a multitude of damaging events (2, 12) (Figure 3). However, it is unclear whether all these factors have a direct causal relationship with DFI, and research is ongoing to understand the aetiology behind (13, 14). Several factors could indicate the use of a sperm DNA fragmentation test. Some of them are related to men’s lifestyle, health, environment, sperm origin (testis, male tract or ejaculate), or assisted reproduction techniques (ART) (Table 2) (2, 7, 8, 9). The embryologist or the medical specialist may also decide to apply SDF tests on the basis of diagnosis or previously undertaken procedures, for example:
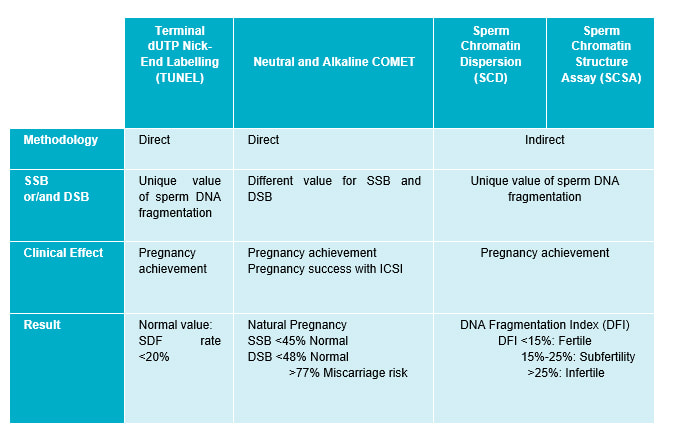
HOW IS SPERM DNA FRAGMENTATION EVALUATED? Sperm DNA fragmentation can be evaluated with different tests (Table 3) (7, 8):
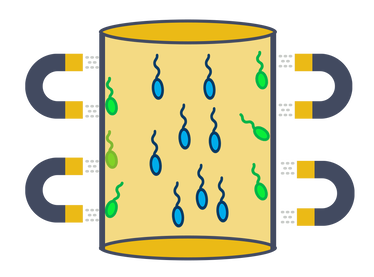
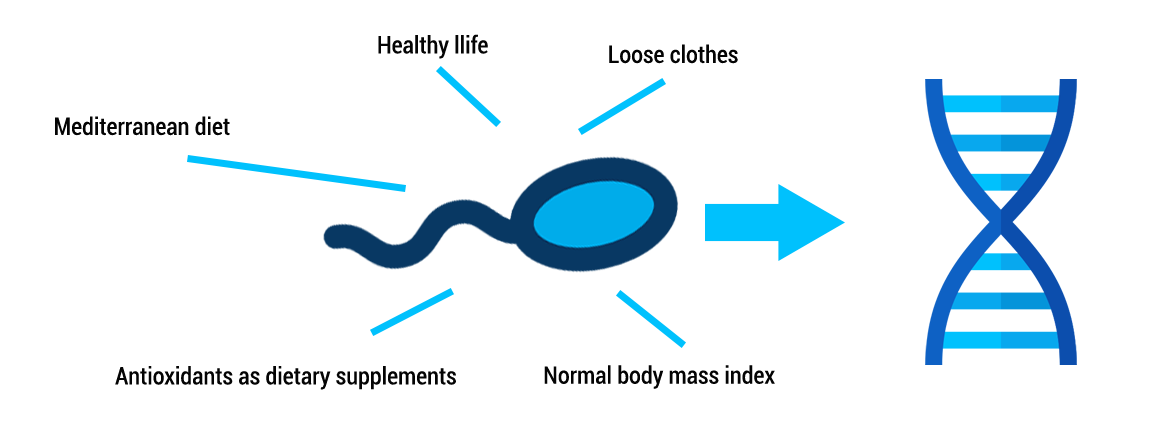
The alkaline comet assay has the highest sensitivity followed by the TUNEL, SCD and SCSA tests (5). In addition, TUNEL and Comet assay have shown the best predictability (23). WHAT ARE THE CURRENT TECHNIQUES TO ELIMINATE SPERMATOZOA WITH FRAGMENTED DNA? The most commonly used techniques to eliminate sperm cells with fragmented DNA are: Magnetic Activated Cell Sorting (MACS) This technique leads to the separation of dead sperms or those with fragmented DNA from those alive and without DNA fragmentation. The semen sample is mixed with a magnetic particle containing an antibody (Ab), which can detect dead DNA fragmented-sperm. Sperm are allowed through the column; those that bind to the Ab are held back, whereas healthy sperm are drawn through the column. So, the semen sample is improved and chances of pregnancy augmented (Figure 4) [to learn more about MACS, visit our previous post]. The FERTILE Chip This is a new technique that enables selection of spermatozoa with the best motility, morphology, lowest DNA fragmentation rate and lowest quantity of ROS. Increased pregnancy rates and decreased risk of miscarriage have been suggested as the end result for this technique (24). The FERTILE Chip is a microfluidic chip to whose end the semen sample is deposited. Healthy motile sperm make it to the other end of the channel, whereas poor sperm quality are retained in the inside (25). HOW TO AVOID SDF? Lifestyle modifications like stopping smoking, reducing alcohol intake and adopting a more balanced diet are the easiest way to improve SDF (Figure 5). Several studies have demonstrated that the use of antioxidants like vitamin C, vitamin E, vitamin B, 𝛃-carotene, isoflavones, (8, 22) etc as dietary supplements can reduce oxidative DNA fragmentation or prevent oxidative stress. However, such positive effect is not observed in all patients, and so further research is needed (8). Additionally, the over-use of antioxidants might also cause various diseases and infertility (2, 5, 12). Antioxidants added to culture media during sperm preparation have been demonstrated to reduce ROS levels produced by ART and so improve sperm parameters. Also, research on mice has shown that antioxidant inclusion during fertilisation has led to improved embryo development (26). Men with inflammatory diseases could be treated with medications to decrease the amount of ROS and, thus, to improve fertility outcomes. Patients suffering from varicocele can undergo varicocelectomy to decrease ROS levels (2). Recurrent ejaculation leads to the discard of older sperm cells with more SDF. Similarly, testicular sperm, directly extracted by surgery from the testis, exhibit lower SDF (2). CONCLUSIONS Female infertility has historically been more widely studied than male factor. Male reproductive potential is evaluated by seminal parameters, like motility, morphology, volume, total sperm count...etc. However, these measures are insufficient as they do not assess the quality of sperm DNA. Nowadays, male factor has become a more interesting focus of attention, and so it is subjected to more and deeper research, such as SDF evaluation. Nevertheless, further studies are needed in order to determine what factors are causing SDF and to identify the optimal antioxidants regime that could reduce the DFI. SDF must be studied in semen samples of ART patients due to its negative impact on embryo development and pregnancy success. The objective of ART is to bring home a healthy child, therefore, it is necessary to successfully carry out techniques such as SDF, when needed, in order to accomplish this goal. REFERENCES 1. Kumar N, Singh AK. Trends of male factor infertility, an important cause of infertility: A review of literature. J Hum Reprod Sci. 2015;8(4):191–6. 2.Cho C-L, Agarwal A. Role of sperm DNA fragmentation in male factor infertility: A systematic review. Arab Journal of Urology. 2018;16(1):21–34. 3.World Health Organization, Department of Reproductive Health and Research. WHO laboratory manual for the examination and processing of human semen. Fifth edition. 2010. 4.Rodríguez-Alonso B, López-Fernández C, Núñez-Calonge R, Caballero P, Guijarro-Ponce JA, Martínez M. Impacto del estrés oxidativo en la dinámica de fragmentación del ADN espermático - ScienceDirect. 2016. [3] ([3]):137-143. 5. Rex AS, Aagaard J, Fedder J. DNA fragmentation in spermatozoa: a historical review. Andrology. 2017; 5(4):622–30. 6. Asghar W, Velasco V, Kingsley JL, Shoukat MS, Shafiee H, Anchan RM, et al. Selection of functional human sperm with higher DNA integrity and fewer reactive oxygen species. Adv Healthc Mater. 2014;3(10):1671–9. 7. Dada R. Sperm DNA damage diagnostics: when and why. Transl Androl Urol. 2017;6(Suppl 4):S691–4. 8. Ribas-Maynou J, Benet J. Single and Double Strand Sperm DNA Damage: Different Reproductive Effects on Male Fertility. Genes. 2019;10(2):105. 9. Aitken RJ, Bronson R, Smith TB, De Iuliis GN. The source and significance of DNA damage in human spermatozoa; a commentary on diagnostic strategies and straw man fallacies. Mol Hum Reprod. 2013;19(8):475–85. 10. Ferreyra JG. High Levels of DNA Fragmentation Observed in an Infertile Population Attending a Fertility Center are Related to Advanced Paternal Age. Journal of Fertilization: In vitro. 2012;02(05). 11. García-Ferreyra J. Sperm DNA Fragmentation and Its Relation With Fertility. New Discoveries in Embryology Bin Wu, IntechOpen.2015. 12. Panner Selvam MK, Agarwal A. A systematic review on sperm DNA fragmentation in male factor infertility: Laboratory assessment. Arab Journal of Urology. 2018;16(1):65–76. 13. Bandel I, Bungum M, Richtoff J, Malm J, Axelsson J, Pedersen HS, et al. No association between body mass index and sperm DNA integrity. Hum Reprod. 2015;30(7):1704–13. 14. Winkle T, Rosenbusch B, Gagsteiger F, Paiss T, Zoller N. The correlation between male age, sperm quality and sperm DNA fragmentation in 320 men attending a fertility center. J Assist Reprod Genet. 2009;26(1):41–6. 15. Park Y-S, Lee S-H, Choi HW, Lee HS, Lee JS, Seo JT. Abnormal Human Sperm Parameters Contribute to Sperm DNA Fragmentation in Men with Varicocele. The World Journal of Men’s Health. 2018;36(3):239–47. 16. Agarwal A, Majzoub A. Role of Antioxidants in Assisted Reproductive Techniques. World J Mens Health. 2017;35(2):77–93. 17. Zini A, Finelli A, Phang D, Jarvi K. Influence of semen processing technique on human sperm DNA integrity. Urology. 2000;56(6):1081–4. 18. Agarwal A, Durairajanayagam D, du Plessis SS. Utility of antioxidants during assisted reproductive techniques: an evidence based review. Reprod Biol Endocrinol. 2014;12. 19. Zeqiraj A, Beadini S, Beadini N, Aliu H, Gashi Z, Elezaj S, et al. Male Infertility and Sperm DNA Fragmentation. Open Access Maced J Med Sci. 2018;6(8):1342–5. 20 Craig JR, Jenkins TG, Carrell DT, Hotaling JM. Obesity, male infertility, and the sperm epigenome. Fertil Steril. 2017;107(4):848–59. 21. Lu, Jin-Chun & Jing, Jun & Chen, Li & Ge, Yi-Feng & Feng, Rui-Xiang & Liang, Yuan-Jiao & Yao, Bing. Analysis of human sperm DNA fragmentation index (DFI) related factors: A report of 1010 subfertile men in China. Reproductive Biology and Endocrinology 16(1). 2018. 22. Nassan FL, Chavarro JE, Tanrikut C. Diet and men’s fertility: does diet affect sperm quality? Fertility and Sterility. 2018;110(4):570–7. 23. Cissen M, Wely M van, Scholten I, Mansell S, Bruin JP de, Mol BW, et al. Measuring Sperm DNA Fragmentation and Clinical Outcomes of Medically Assisted Reproduction: A Systematic Review and Meta-Analysis. PLoS ONE. 2016;11(11):e0165125. 24.Alagöz O, Özkara G, Koçer M, Ficicioglu C. Improving Pregnancy Rate in IVF Cycles by Preparing Sperm via Microfluidic Sperm Chips. ESHRE. 2017. 25. Quinn MM, Jalalian L, Ribeiro S, Ona K, Demirci U, Cedars MI, et al. Microfluidic sorting selects sperm for clinical use with reduced DNA damage compared to density gradient centrifugation with swim-up in split semen samples. Hum Reprod. 2018. 26. Truong T, Gardner DK. Antioxidants improve IVF outcome and subsequent embryo development in the mouse. Hum Reprod. 2017;32(12):2404–13.
2 Comments
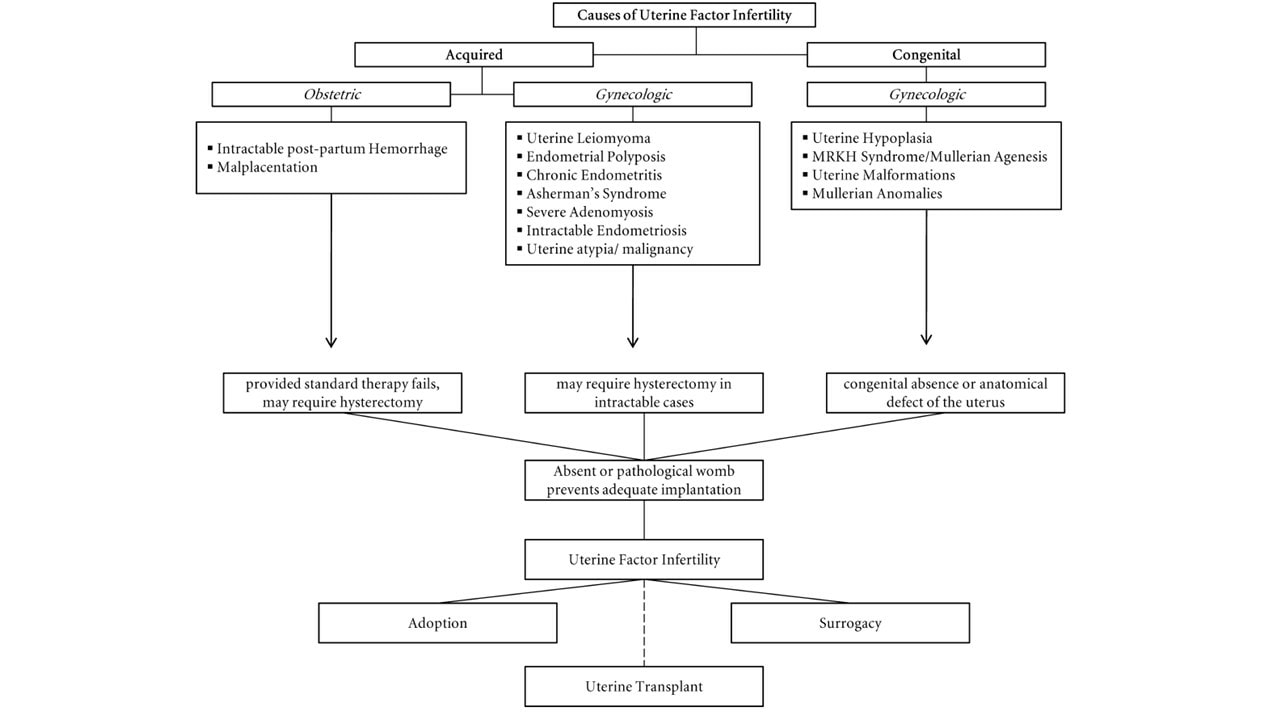
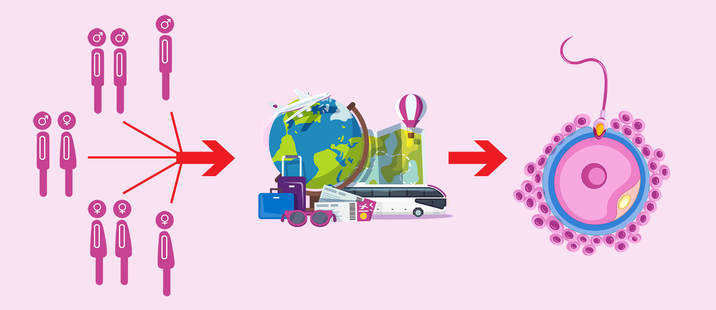
"Increasing efforts in making this procedure feasible and more widely available are expected, since there is a significant clinical demand" Author: Iris Martínez Rodero WHAT IS UTERUS TRANSPLANTATION? Uterus transplantation is a surgical technique in which a healthy and functional uterus is temporarily transplanted into a woman whose uterus is absent or dysfunctional (1, 2). Thereby, uterine transplant is a potential treatment for absolute uterine factor infertility (AUFI), where normal embryonic implantation cannot occur. AUFI causes up to 3-5% of female infertility cases worldwide, whose underlying conditions are (Figure 1) (1):
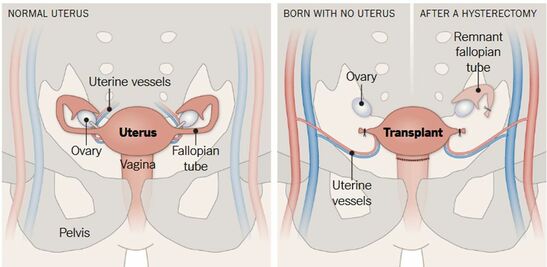
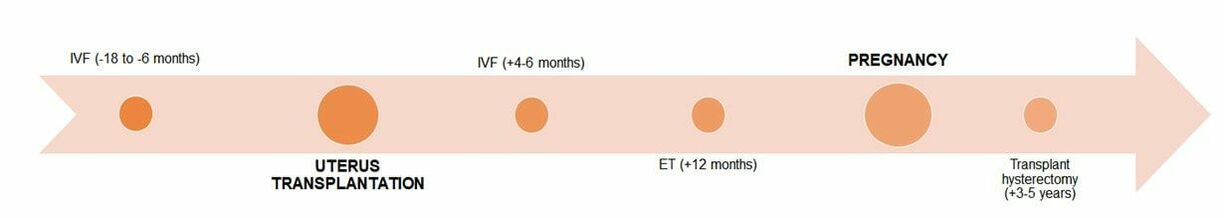
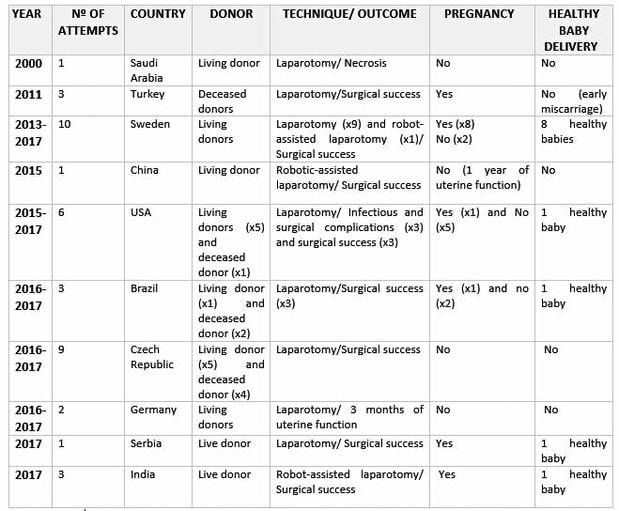
Figure 1. Causes of uterine factor infertility and the options for having offspring (1). Overview of the whole process For a uterine transplantation, both a recipient and a compatible donor are needed. The ideal approach is using a uterus from a deceased donor to avoid putting healthy women at risk (1, 2, 4). The surgery is more complicated than a standard hysterectomy (surgery where the uterus is removed), and for a living donor it entails an 11-hour long operation, with surgeons working near vital organs. It consists of the removal of the donor’s uterus, cervix, part of the vagina and other tissues necessary to attach the uterus to the recipient. It is a meticulous procedure, since tiny blood vessels need to be resected without injuring the donor. In the case of deceased donors, the organ can be removed faster since there is no risk of causing harm. Once extraction is completed, the organ can survive for at least six hours if maintained under the proper conditions (1, 4). The next step is the transplant surgery, which normally takes about five hours. In this procedure, an artery and a vein on either side of the uterus are connected to the recipient’s blood vessels, while there is no need to reconnect the nerves. Part of the donor’s vagina is stitched to the recipient’s (Figure 2). Then, the transplanted uterus is stabilized into the pelvis using supporting tissue removed from the donor (1, 4). Figure 2. Uterus transplant. Surgeons remove the uterus, cervix and part of the vagina from a donor (either living or deceased). Small uterine vessels are also preserved so they can carry blood to the organ. The uterus is then stitched to the recipient’s vagina and the small uterine vessels are connected to large blood vessels. If present, the recipient’s ovaries are moved away and remnant Fallopian tubes are not in contact with the transplanted uterus. One year after the surgery, the recipient may undergo embryo transfer procedures (4). Given that the recipient Fallopian tubes are not be connected to the transplanted uterus, a natural pregnancy will be impossible. Therefore, the embryo is conceived by in vitro fertilization, which can be performed either before or, preferably, after transplantation (1, 4). For a full recovery from surgery to be achieved and in order to adjust the right doses of immunosuppressant/anti-rejection medicine, normally a whole year is required (Figure 3). Past this time, the patient can try to conceive. As previously mentioned, the doctors will transfer one in vitro fertilized embryo at a time into the uterus, until pregnancy is achieved. Subsequently, in order to avoid the transplanted uterus undergoing the stress of delivering the baby, a caesarean section is performed before the due date (1, 4). After delivery the uterus can be kept, should the mother want to try to conceive again eventually. For safety reasons, the limit is set for two babies per transplanted uterus. In case the woman does not wish to conceive another child she may decide to have the uterus removed through hysterectomy, so she can stop taking the immunosuppressant medication (1). Figure 3. Uterine transplantation timeline (1) Complications of uterine transplantation Transplantation is a hugely complex intervention; not only does it involve potential complications of surgery as postoperative bleeding, possible infections or atrial fibrillation, but also immune rejection of the transplanted organ. To avoid an immune reaction, immunosuppressants need to be taken after transplantation. Since these weaken the immune system, infections and tissue recovery are even more complex to solve (1, 2, 4, 5). CHRONOLOGY OF UTERINE TRANSPLANTATION: FROM TRIALS ON ANIMALS TO CURRENT SUCCESSFUL ATTEMPTS In the early 50’s, the first trials of uterus transplantation were performed in various animal models, including primates (1, 2). What is considered to be the world’s first clinical uterus transplantation is a trial performed by Fagee and his team in Saudi Arabia in 2000. The surgery was proved technically possible, even though the uterus was removed 100 days later due to a necrosis episode (5). Nonetheless, more trials were carried out along the years, mainly in Turkey, where uteri from deceased donors or attained pregnancies were used, all of them ending in miscarriages (1). In 2014, the first delivery of a healthy baby from a donor uterus was achieved. This event, led by Mats Brännström from Gothenburg University in Sweden, opened the doors to the feasibility of uterus transplantation and prompted new areas of clinical and basic research (6). Nowadays, at least 12 healthy babies have been born from uterine transplantation (1). Until 2017, all babies had been born from uteri donated by living women. Several attempts of uterus transplantation from deceased donors had been surgically successful, but the first healthy baby was born on Dec 15, 2017 in Hospital das Clínicas, University of São Paulo, Brazil (7). A summary of both successful and unsuccessful uterus transplantation attempts reported in the literature is shown below (Table 1): Table 1. Uterine transplantation clinical trials from 2000 to 2018 (1,6,7,8) RISK-BENEFIT ANALYSIS Currently, women suffering from uterine infertility who wish to have offspring carrying their genetic background can turn to surrogacy combined with in vitro fertilization techniques to accomplish their desire. By “borrowing” or “renting” the womb of another woman, a baby conceived in vitro from the gametes of the infertile couple can be born (2). However, surrogacy not only does give rise to multiple ethical controversies, but it is also outlawed in many countries, such as Spain or Italy. Surrogacy is perceived as damaging the dignity of motherhood, while turning babies into a commodity, often resulting in negotiations and litigations. In this sense, some authors position uterus transplantation as a “more ethical” alternative to surrogacy (2, 4, 5). Aside from ethical concerns, uterine transplantation is often considered to be more feasible because it allows the recipient to experience gestation and labour, whereas surrogacy does not (2). It is worth noting that 97.5% of women who underwent a survey in the UK, where surrogacy is legal, preferred uterine transplantation over gestational surrogacy or adoption (9). Furthermore, a recent survey in Japan revealed that uterine transplant was two times more accepted than gestational surrogacy among among 3,892 women from the general population aged 25 to 39 (10). In Sweden, 2000 out of 3500 randomly selected women aged 30-39 preferred uterine transplant over surrogacy (11). Nevertheless, uterus transplant is still a very complex surgical procedure, which presents significant health threats and does not guarantee a successful, healthy pregnancy. Medical staff should therefore inform patients about the risks inherent to the procedure, particularly about the hazards of immunosuppressant treatment typical of a transplant. In this case, immunosuppressant drugs may nullify the purpose of the transplant itself, as they can result in injury to the foetus and even lead to its death (1, 2, 5). Moreover, women should be warned that, as there is no nerves reconnection, they will not perceive the fetus pressing against the inner walls neither will they feel the transplanted womb contractions (2). All the above mentioned, it is clear that a risk-benefit ratio of uterus transplantation should be carefully assessed and described in each case. ETHICAL REMARKS UterIne transplantation entails multiple medical, ethical and legal issues, involving both donors and recipients. That is why in 2012, Lefkowitz and colleagues from McGill University in Canada proposed “the Montreal Criteria” (Figure 4). This is a set of proposed criteria required for a woman to be considered a candidate for uterine transplant, and ethically and medically approved (2, 3). Figure 4. The Montreal Criteria (3).
CONSEQUENCES FOR DONORS, RECIPIENTS AND CHILDREN Possible implications for donors, recipients and children have not been yet studied. If uterus transplantation becomes a routine procedure, a multi-disciplinary team effort would be necessary to monitor uterus donors, recipients and babies (1). There exist a few results about long-term psychological strains in living donors (1, 12). In a observational study conducted by Brännström et al. in the 1-year follow up, the donors were asked if they had returned to predonation mental health, if they had any regrets and if they had returned to previous activities both professionally and privately (12). They concluded all donors returned to their predonation levels of physical health (12). Other 'side effects' were similar or comparable to minor medical and surgical complications, which were actually reversible and could be solved shortly after the surgery. Still, the absence of long-term complications should be confirmed (1, 5). Regarding recipients, an established program of follow-up should be defined, which would consist of medical exams and psychological assessments (1). Additionally, the study should continue to monitor the wellbeing of the recipients even after they have delivered their babies and had the uterus removed (1, 2). The surgery for the transplanted uterus removal would be planned after the delivery of the desired number of babies (1, 2, 5). To date, all children born after uterus transplantation have been reported to be healthy. However, they developed inside a womb that might have been taken from a deceased donor, and so it could have been exposed to anti-rejection drugs (5,6). Since possible side-effects might be detected at a later stage, a registry should be created in order to collect all possible information about their long-term health (1, 5). FUTURE PROSPECTS FOR UTERUS TRANSPLANT Tissue engineering and the creation of artificial organs is a promising field that would dramatically improve the current situation of transplants (13). Uterus transplant would benefit from that kind of advances, as second-party donors and immunosuppressive therapy would not be needed. Although a human bioartificial uterus is far from being ready for clinical application, there are some promising results in animals (14, 15, 16). CONCLUSIONS Women who suffer from uterine factor infertility need to rely on surrogacy to fulfill their wish to conceive their own offspring. Nevertheless, surrogacy is illegal in many countries, whereas uterus transplantation can be a sustainable and ethically acceptable alternative for patients who wish to achieve motherhood. From an experimental point of view, uterus transplantation has evolved rapidly from its origin in animal models to its current successful clinical application. However, this should still be regarded as an experimental procedure, and a risk-benefit analysis is due on a case-by-case basis to ensure it is applied in an ethical and appropriate manner. A detailed registry of surgical outcome and reported complications and side effects needs to be collected from clinical trials before uterus transplant becomes a routine procedure. Increasing efforts in making this procedure feasible and more widely available are expected, since there is a significant clinical demand. REFERENCES
“HIV+ serodiscordant couples should seek advice from fertility experts regarding the optimal route for safe conception” Author: Belén Gómez Giménez Human Immunodeficiency Virus (HIV) is a retrovirus that attacks and weakens the immune system (body's defense) against infections, diseases and some types of cancer. The most advanced stage of HIV infection is Acquired Immunodeficiency Syndrome (AIDS), which can take from 2 to 15 years to develop after infection (1). At least 36.9 million people have been estimated to be infected by HIV. In the past, patients were given short life expectancy but, due to the introduction of combined antiviral therapies, HIV is now classed as a chronic disease. Estimations from the World Health Organization (WHO) are that, between 2000 and 2017, the number of new infections and deaths were reduced by 36% and 38%, respectively. As a result, nowadays there are more HIV carriers who can enjoy a normal life, which allows them to pursue family planning. Nevertheless, certain risk of the virus transmission to both the offspring (vertical transmission) and the partner (horizontal transmission) is still to be considered (1). There is disagreement among experts on whether natural conception is safe in HIV+ couples or whether they should turn to assisted reproduction techniques (ART) instead. There is some evidence that virus transmission in seropositive couples with a highly active antiretroviral therapy (HAART), in combination with timed intercourse and pre-exposure prophylaxis for the HIV negative partner, is associated with minimal risk, thus allowing for natural conception. On the contrary, other studies recommend ART-based approaches, since these represent a safer choice for serodiscordant couples who have access to these modern methods (2). Female HIV-seropositive Non-coital ovulatory vaginal or intrauterine insemination (IUI) is a simple, safe method for infected individuals to get pregnant. In addition to this, patients should continue with HAART, which suppresses the viral load during the course of pregnancy and breast-feeding to minimize the risk of vertical transmission to the offspring (2). HIV-seropositive males Males can transmit the virus through seminal fluid and, as a result, partners and/or offspring may result infected. Semen induces changes in the female reproductive tract that promote conception and pregnancy. However, these alterations can also facilitate HIV activation, thus increasing the likelihood of the partner becoming infected with HIV (3). Human seminal plasma is a made up of a mix of fluid secretions, in which cell types other than sperm are found. These non-sperm cells have been recognized to be the agent behind viral transmission (4). Consequently, a method to overcome spreading of HIV known as “sperm washing” was developed, based on the principle of separating motile sperm from free HIV particles and infected cells (5). This method was first applied to sperm in 1992 (6). The sample protocol for working with HIV-seropositive semen samples in laboratories is summarized below (5, 7): In order to avoid viral contamination, it is necessary to work in isolated laboratories (biosafety cabin workstation, exclusive centrifuge) and set samples apart from non-infected samples when stored in nitrogen tanks to avoid cross-contamination. Otherwise, the same standard sperm preparation procedures are applied for infected and non-infected samples, as outlined as follows:
Depending on female characteristics such as age, reproductive tract malformations or endometriosis and male partner semen parameters, different methods of assisted reproduction may be applied. The simplest one is IUI, but in the presence of any limiting factors, either in vitro fertilization (IVF) or intracytoplasmic sperm injection (ICSI) is used. Differences between these procedures have been explained in our previous post. The main difference between non-infected and HIV+ sample preparation is that the latter requires the sperm washing procedure. This protocol entails two distinct preparation techniques (Figure 1): density gradient centrifugation (DGC) and swim up.
The consecutive use of these two techniques has achieved a notable reduction in the risk of transmission of the virus. Following sperm preparation, polymerase chain reaction (PCR) will be performed to analyse the genetic content of spermatozoa and screen for HIV. This technique, however, is limited by the fact that the analysed sperm only represent a small fraction of the overall sample, which can lead to a false negative result. CONCLUSIONS Semen washing is the best method to eliminate the presence of HIV in sperm and avoid transmitting the virus to the partner or offspring. Although there are studies that suggest that natural coitus can be a safe method if the patients are treated with anti-retroviral therapies, the general consensus is that this is not recommended if the affected couple has access to ART (2). Throughout ART treatment, pregnancy and during the first year after birth, diagnostic testing for both the mother and infant is recommended to ensure transmission has not occurred (5). Within assisted reproduction, there is no consensus on which method (IUI, IVF or ICSI) should be used in patients, and so this decision will be based on the HIV status of the couple (female positive, male positive or both), as well as their own fertility potential. Many studies have demonstrated that after semen wash, the risk of vertical and horizontal virus transmission is significantly reduced, showing that this is an effective procedure (10). Despite this, certain studies must be taken into account that have reported presence of the HIV-virus in spermatozoa following the procedure (11). Overall, it is critical that more studies are carried out to demonstrate these techniques are completely risk-free, and efforts should be made to unify the semen washing protocol between laboratories (10). HIV+ serodiscordant couples should always seek advice from fertility experts regarding the optimal route for safe conception. REFERENCES
1- WHO: HIV/AIDS [Internet]. Geneva: World Health Organization; c2018 [ cited 2018 August 31]. Available from: http://www.who.int/en/news-room/fact-sheets/detail/hiv-aids. 2- Zakarin Safier L, Sauer MV. Fertility care interventions should be provided as the first line options for HIV+ serodiscordant couples who desire children in settings with affordable access to care, regardless of their fertility status. J Int AIDS Soc. 2017;20(Suppl 1):21294. 3- Doncel GF, Anderson S, Zalenskaya I. Role of semen in modulating the female genital tract microenvironment--implications for HIV transmission. Am J Reprod Immunol. 2014;71(6):564-74. 4-Jindal SK, Rawlins RG, Muller CH, Drobnis EZ. Guidelines for risk reduction when handling gametes from infectious patients seeking assisted reproductive technologies. Reprod Biomed Online. 2016;33(2):121-30. 5-Medicine PCoASfR. Recommendations for reducing the risk of viral transmission during fertility treatment with the use of autologous gametes: a committee opinion. Fertil Steril. 2013;99(2):340-6. 6- Semprini AE, Levi-Setti P, Bozzo M, Ravizza M, Taglioretti A, Sulpizio P, et al. Insemination of HIV-negative women with processed semen of HIV-positive partners. Lancet. 1992;340(8831):1317-9. 7- Garrido N, Meseguer M, Bellver J, Remohí J, Simón C, Pellicer A. Report of the results of a 2 year programme of sperm wash and ICSI treatment for human immunodeficiency virus and hepatitis C virus serodiscordant couples. Hum Reprod. 2004;19(11):2581-6. 8- WHO laboratory manual for the examination and processing of human semen- 5th ed. In: Geneva: WHO Press, 2010. 9- Zamora MJ, Obradors A, Woodward B, Vernaeve V, Vassena R. Semen residual viral load and reproductive outcomes in HIV-infected men undergoing ICSI after extended semen preparation. Reprod Biomed Online. 2016;32(6):584-90. 10- Mencaglia L, Falcone P, Lentini GM, Consigli S, Pisoni M, Lofiego V, et al. ICSI for treatment of human immunodeficiency virus and hepatitis C virus-serodiscordant couples with infected male partner. Hum Reprod. 2005;20(8):2242-6. 11- Elizaveta E B, Bocharova E, Azova M. Spermatozoon as a Vehicle for Viruses. Int J cell Sci & mol biol. 2017; 2(4) : 555593. Author: Sara Gómez García “Gene expression in the embryo can be modulated by the uterus conditions or pregnant woman lifestyle” A couple seeking assisted reproduction treatment can choose either to use their own oocytes (eggs) or those from a third party (donor). In the latter case, oocytes will be inseminated with the partner’s semen or donor semen. INDICATIONS TO USE DONATED OOCYTES
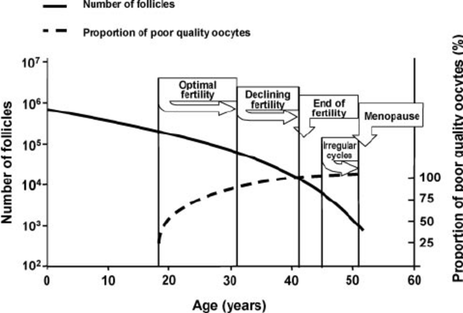
At birth, the number of oocytes of a baby girl is limited, and progressively reduced over the course of her life. Broadly speaking, oocyte quality decreases with age, which increases the number of miscarriages and the number of children born with some kind of genetic abnormality (2, 3) (Fig.1). When the patient cannot count on her own oocytes, one of the most suitable options may be looking for a donor. WHO CAN BECOME AN EGG DONOR? An egg donor is a woman of legal age who has decided to give her eggs away for other women. Donors can be anonymous or non-anonymous, depending on what laws dictate in a specific country:
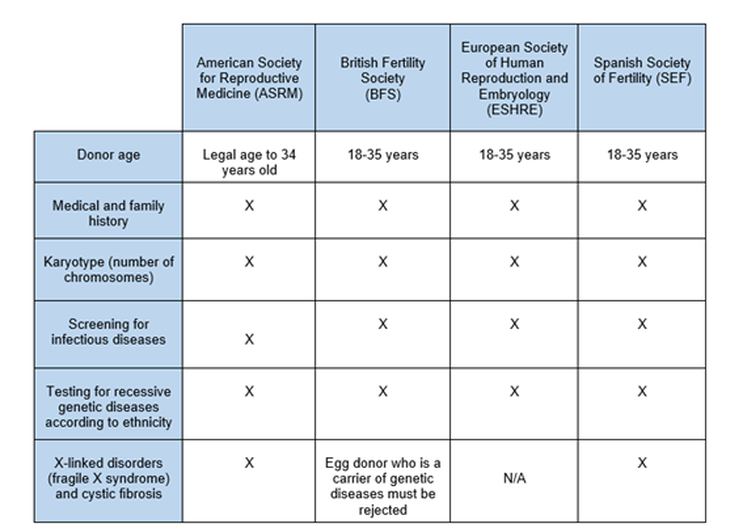
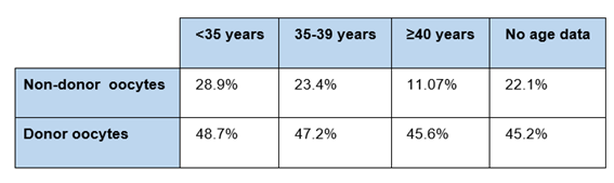
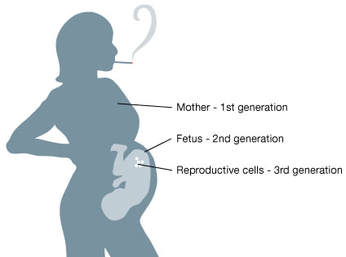
Egg donors must pass physical, genetic and psychological tests before even being able to commence the process. The assisted reproduction guidelines, stated by a variety of professional bodies, recommend different tests to evaluate the health status of oocyte donors (Table 1) (4, 5, 6, 7): WHERE IS LEGAL TO PERFORM AN EGG DONATION TREATMENT IN EUROPE? Even though egg donation is an procedure in expansion, it is not yet legal everywhere. In addition, treatments use anonymous and/or non-anonymous donors, as specified by the local legislation of each European country (Table 2). HOW COMMON IS IT TO USE DONOR EGGS? The first egg donation was performed thirty-five years ago (8). It is progressively becoming more common to use egg donors due to changes in lifestyle, with couples now waiting longer to have their first child. In 2013, 39,000 egg donation treatments were made in Europe out of a total of 500,000 IVF cycles, which corresponds to 7.8% of overall cycles. Furthermore, 50% of all them were performed in Spain (3). In 2015, 14,655 egg donation procedures were performed in Spain alone, which represents about 20% of all human assisted reproduction cycles in the country (9). In addition, data from 2014 showed 8.5% of all treatments in Spain were performed to foreign patients, 66% of them actually corresponding to egg donation procedures (3). It is possible that Spain carries out most of the donor oocyte cycles across Europe due to a combination of factors: less restrictive legislation, higher quality of techniques and excellent clinical outcomes. IS THIS PROCEDURE EFFECTIVE? Pregnancy rates with donor oocytes are higher than those achieved using the patient’s own oocytes in the different age ranges. This is likely due to the younger age of donors and the fact that uterine age does not affect results (Table 3). WILL MY CHILDREN SHOW ANY PHYSICAL RESEMBLANCE TO ME? The oocyte is fertilized by a sperm to form the embryo. Its DNA is, therefore, composed of oocyte DNA and sperm DNA. This mixture of DNA (genes) determines the physical characteristics of the embryo. However, there is another factor known as epigenetics that could modify such features. Epigenetics are changes in gene expression without changing the DNA sequence, which can be influenced by environmental conditions, age and a variety of factors. Regarding gene expression in the embryo, it can be influenced by the uterus conditions or lifestyle of the pregnant woman (diet, stress, drugs, toxins, hormones...). Also, not only the first generation (woman) is affected in terms of gene expression at the epigenetic level, but also the second and third generation may be affected, as well. Even the fourth generation could inherit an epigenetic effect despite not being directly exposed (Fig.2) (10). An example of these epigenetic changes is illustrated by a study performed in mice, in which the author showed that modulating the diet may modify coat color. Even though the investigated yellow and brown mice were genetically identical, the diet of the mother likely produced brown mice under a yellow genetic background (changes in gene expression) (11). In humans, studies have shown that adherence to a Mediterranean diet during pregnancy can have a protective effect against the development of metabolic diseases in the offspring (12, 13). Other research teams have investigated the effect of maternal cigarette smoking on children. This has been correlated to low birth weight, asthma, obesity, altered neurobehavior and cancer (14, 15). In conclusion, an ongoing communication is performed between mother and child (embryo) that could modulate gene expression in the embryo. Nevertheless, new research is necessary to fully understand this process. TAKE-HOME MESSAGE Egg donors can be anonymous and/or non-anonymous, and it is regulated by different legislations in different countries. Egg donation is becoming a more common process, pushed forward by the high pregnancy rates obtained in several age ranges without effects related to uterus age. Interestingly and as mentioned above, despite not being the genetic mother, gene expression in the embryo can be modulated by the uterine conditions or pregnant woman lifestyle. And remember... “Being a mother is an attitude, not a biological relation” Robert A. Heinlein REFERENCES
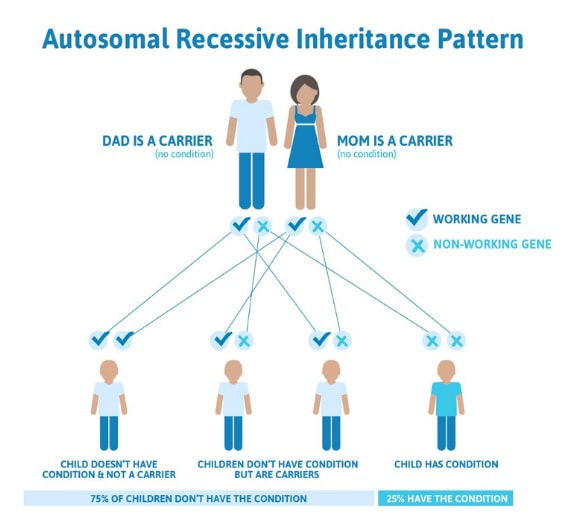
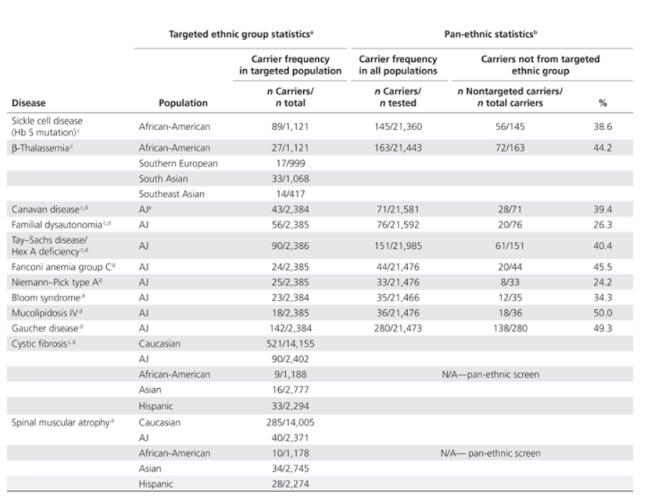
1. Broekmans F.J., Soules M. R., Fauser B. C. Clinical and endocrinological aspects of ovarian ageing. Endocrine Reviews [Internet] August 2009.[cited 2018 Jun 27] 30(5):465–493. 2. MANIFIESTO DE LA SEF SOBRE LA DONACIÓN DE GAMETOS EN ESPAÑA [Internet] Sociedad Española de Fertilidad. n.d. [cited 2018 Jul 25]. Available from: https://goo.gl/KZ6ZD9. 3. Egg donation fact sheets 3 [Internet] European Society of Human Reproduction and Embryology. January 2017 [cited 2018 Jul 25]. Available from: https://goo.gl/T86TLR. 4. Association of Biomedical Andrologists, Association of Clinical Embryologists, British Andrology Society, British Fertility Society, Royal College of Obstetricians and Gynaecologists. UK guidelines for the medical and laboratory screening of sperm, egg and embryo donors (2008). Hum Fertil (Camb). 2008 Dec;11(4):201–10. 5. Weinig JRM, Hernández JH. Estudio y tratamiento de la pareja estéril: Recomendaciones de la Sociedad Española de Fertilidad, con la colaboración de la Asociación Española para el Estudio de la Biología de la Reproducción, de la Asociación Española de Andrología y de la Sociedad Española de Contracepción [Internet]. Adalia farma; 2007 [cited 2018 Jul 25]. Available from: https://dialnet.unirioja.es/servlet/libro?codigo=562165. 6. Barratt C, Englert Y, Gottlieb C, Jouannet P. Gamete donation guidelines. The Corsendonk consensus document for the European Union. Hum Reprod [Internet]. 1998 Feb 1 [cited 2018 Jul 25];13(2):500–1. Available from: https://academic.oup.com/humrep/article/13/2/500/876942. 7. 2008 Guidelines for gamete and embryo donation: a Practice Committee report. Fertility and Sterility [Internet]. 2008 Nov 1 [cited 2018 Jul 5];90(5):S30–44. Available from: https://www.fertstert.org/article/S0015-0282(08)03714-X/fulltext. 8. Trounson A, Leeton J, Besanko M, Wood C, Conti A. Pregnancy established in an infertile patient after transfer of a donated embryo fertilised in vitro. Br Med J (Clin Res Ed) [Internet]. 1983 Mar 12 [cited 2018 Jul 5];286(6368):835–8. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1547212/. 9. Sociedad Española de Fertilidad [Internet] Registro Nacional de Actividad 2015: Informe estadístico de Técnicas de Reproducción Asistida 2015 [cited 2018 Jul 25]. Available from: https://goo.gl/ZB8EhA. 10. Genetic Science Learning Center (2013, July 15) [Internet]. August 21, 2018. Epigenetics & Inheritance [cited 2018 Jul 5]. Available from: http://learn.genetics.utah.edu/content/epigenetics/inheritance/. 11. Dolinoy DC. The agouti mouse model: an epigenetic biosensor for nutritional and environmental alterations on the fetal epigenome. Nutr Rev [Internet]. 2008 Aug [cited 2018 Aug 22];66(Suppl 1):S7-11. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2822875/. 12. Lorite Mingot D, Gesteiro E, Bastida S, Sánchez-Muniz FJ. Epigenetic effects of the pregnancy Mediterranean diet adherence on the offspring metabolic syndrome markers. Journal of Physiology and Biochemistry [Internet]. 2017 [cited 2018 Aug 26];73(4):495–510. Available from: http://link.springer.com/10.1007/s13105-017-0592-y. 13. Geraghty AA, Lindsay KL, Alberdi G, McAuliffe FM, Gibney ER. Nutrition During Pregnancy Impacts Offspring’s Epigenetic Status—Evidence from Human and Animal Studies. Nutr Metab Insights [Internet]. 2016 Feb 16 [cited 2018 Aug 26];8(Suppl 1):41–7. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4758803/. 14. Armstrong DA, Green BB, Blair BA, Guerin DJ, Litzky JF, Chavan NR, et al. Maternal smoking during pregnancy is associated with mitochondrial DNA methylation. Environ Epigenet [Internet]. 2016 Aug 1 [cited 2018 Aug 26];2(3). Available from: https://academic.oup.com/eep/article/2/3/dvw020/2415103. 15. Joubert BR, Håberg SE, Bell DA, Nilsen RM, Vollset SE, Midttun Ø, et al. Maternal smoking and DNA methylation in newborns: In utero effect or epigenetic inheritance? Cancer Epidemiol Biomarkers Prev [Internet]. 2014 Jun [cited 2018 Aug 26];23(6):1007–17. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4140220/. Author: Lidón Carretero Vilarroig "Scientists have developed a genetic test to detect carriers of recessive mutations, which can reduce a couple’s chances of having a child with a genetic disorder". Regarding genetic disorders, a couple with no family history may have an affected child without knowing they were asymptomatic carriers. This situation is mostly common in related couples. It is estimated that each one of us carries at least one genetic disorder. Nevertheless, this does not mean suffering from them, as they are inherited with a recessive pattern. What does this mean? For an autosomal genetic disease with a recessive inheritance pattern to develop, it is necessary that each parental passes on one mutation to their offspring. Thus, people with only one mutation will never suffer from the disease. This way, when a couple randomly carries one mutation in the same gene the probability of having an affected baby is 25% (Figure 1). As much, related couples are more likely to be carriers of the same genetic disorder, since they probably share the same ancestral chromosomes. This scenario is more common in little cities or villages, where marriage between relatives were common not too long ago, just a few generations back. That is exactly what happened in the European royalty in the 19th and 20th centuries with Haemophilia B. This is a recessive disorder caused by a mutation in the X chromosome, which causes a bleeding disorder. Due to consanguinity marriages, the incidence of haemophilia B in the royal family was higher than in the rest of the population, which caused a great weakness in the royalty. I AM PLANNING TO HAVE A BABY. IS THERE ANY WAY TO AVOID THIS KIND OF RISK? Yes, there is. Scientists have developed a genetic test to detect carriers of recessive mutations, which can reduce a couple’s chances of having a child with a genetic disorder. It is known as Carrier Screening Panels, and it is performed using only blood sample. In the first instance only one member of the couple needs to be tested, since a negative result in one of them eliminates the possibility of having an affected child. It is important to point out that there is always a residual risk of being a carrier, since these panels do not screen for all genetic disorders. Carrier screening tests are available for a wide range number of diseases, depending on the trademark. The more expensive a panel is, the more mutations it screens. Nevertheless, there are many common diseases that can be avoided using this tool. A great example would be cystic fibrosis (CF). CF is a recessive genetic disorder that mostly affects the respiratory tract (with a variety of diverse consequences). There is no cure, and the treatment depends on the case, being 37 the average life expectancy for patients who live past childhood (2). There are a lot of CF healthy carriers worldwide who do not know their carrier status and are at risk of having a child affected by this condition. WHAT ARE THE MAIN CONSIDERATIONS WHEN SCIENTIST CHOOSE THE GENETIC DISEASES FOR THEIR COMMERCIAL PANEL?
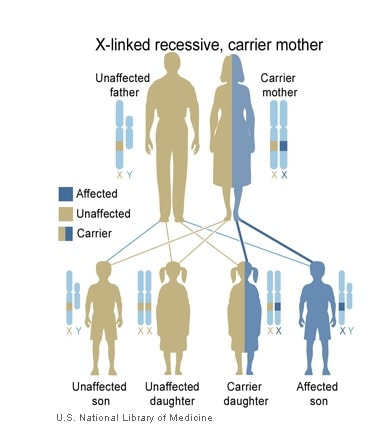
WHAT IF MY PARTNER AND I ARE BOTH CARRIERS OF THE SAME GENETIC DISORDER? If both members of a couple are carriers for the same recessive genetic disorder, they have a chance of 1 in 4 of having a baby affected of this disease, or 1 in 2, if it is the case of an X-linked recessive trait. Fortunately, there are other alternatives for them:
TAKE HOME MESSAGE Everybody carries at least one genetic disorder. Since carriers are usually healthy, they are not aware of their risk of having a child with a genetic disorder. Carrier screening tests are aimed at minimizing the random risk each couple have because of being carriers of the same recessive genetic condition. This test consists of a panel that screens a variable number of mutations and genetic disorders depending on the commercial name. By performing a carrier screening test a couple can minimize the risk of having an affected child, but remember that commercial panels only screen for the most common genetic disorders. If you are planning to have a baby and are interested in carrying out a Carrier Screening test, you should speak with your gynaecologist and seek for advice on these matters. REFERENCES
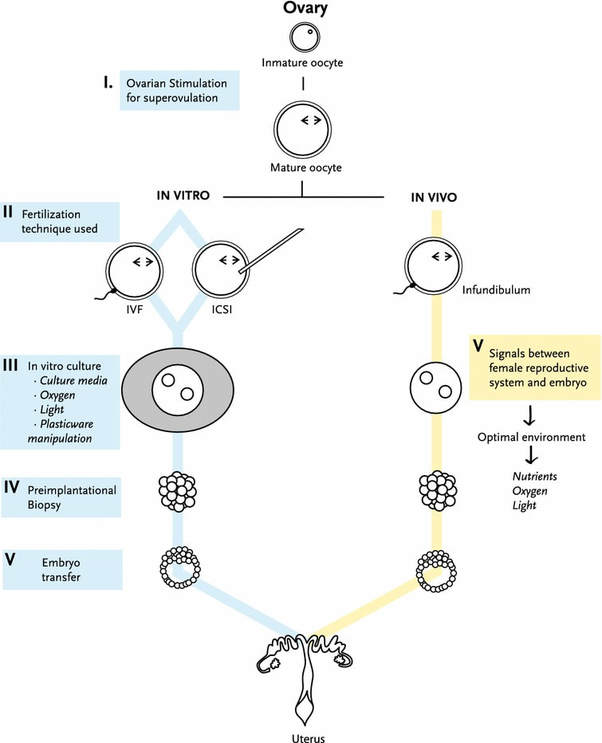
1. https://geneticsupportfoundation.org/archive/genetics-and-you/autosomal-recessive-inheritance 2. https://cysticfibrosisnewstoday.com/cystic-fibrosis-life-expectancy/ 3. Rohlfs EM, Zhou Z, Heim RA, Nagan N, Rosenblum LS, Flynn K, et al. Cystic fibrosis carrier testing in an ethnically diverse US population. Clin Chem. 2011;57(6):841–8. 4. http://www.treat-nmd.eu/dmd/about/x-linked-recessive/ 5. Gabriel A. Lazarin et al. An empirical estimate of carrier frequencies for 400+ causal Mendelian variants: results from an ethnically diverse clinical sample of 23,453 individuals Genet Med. 2013 Mar; 15(3): 178–186. Author: Belén Gómez Giménez "It is without question that embryologists work tirelessly to help families achieve their dream of bringing a healthy baby home". It is estimated that 186 million people in the world have infertility issues. Infertility is defined as the inability to achieve conception by natural means through unprotectected sexual intercouse after one year. Gynaecologists, andrologists, nurses and psychologists are the professionals who attend to couples from the very beginning. However, there are also other important professionals in fertility centres who work discreetly and with no direct contact with patients, and yet help them in their aspiration to raise a family. They are mainly biologists, but there are also chemists or biotechnologists whose role inside the laboratory is crucial. The present text intends to explain more accurately who embryologists are, with details on their specific tasks in the laboratory. WHO ARE THE EMBRYOLOGISTS? An embryologist is a reproduction biology specialist. In particular, clinical embryologists are in charge of manipulating eggs, sperm and embryos in order to achieve fertilisation. Although most of the decisions related to treatments are often taken by doctors, it is the embryologists' call to decide on several aspects of the appropriate approach for the specific treatment (1), as well as coordinating the performance of every activity in the laboratory. In some countries, embryologists are even responsible for informing patients about certain laboratory procedures and, to some extent, embryo development, too (1). These responsibilities require embryologists to have wide experience in clinics, high degree of technical skills and extensive knowledge of reproductive human biology (2). TREATMENTS IN ASSISTED REPRODUCTION CLINICS Couples who decide to go through a fertility treatment are normally asked for blood tests to determine their hormone profile. Men usually go through semen analyses, too, and it is not uncommon for both partners to be subjected to physical examination if required, looking to discard specific abnormalities of the reproductive system. Once gynaecologists and/or andrologists have diagnosed the specific issue, they deliberate along with the senior embryologist on the most appropriate treatment depending on woman age, sperm quality or genetic problems. Broadly speaking, three different approaches can be considered: intrauterine insemination, in vitro fertilization and intracytoplasmatic sperm injection. Intrauterine insemination (IUI) is probably the easiest and least invasive procedure. It is indicated in very clear and specific cases such as tube obstruction, cervical factor infertility (3) or erectile dysfunction, and even some other unclear fertility issues that result in the inability to conceive by natural means (3). IUI consists in increasing gamete (spermatozoa) density at the site of fertilization by introducing sperm directly into the uterus (4). This technique requires previous sperm capacitation in the laboratory; capacitation is a natural process that comprises several physiological changes in the sperm necessary for fertilisation. Embryologists perform different procedures based on their evaluation of motile sperm that allows for selection of the best gametes (5) [find more about sperm selection in our previous post here]. Couples suffering from low sperm count or low sperm motility on the male side, or damaged Fallopian tubes or endometriosis on the female side (and more complex cases of infertility for either of them) are usually recommended to resort to either in vitro fertilization (IVF) or intracytoplasmatic sperm injection (ICSI) (depending on the particular situation). In both cases, ovarian stimulation is required for women. Hormonal stimulation of the ovary aims to increase the number of oocytes produced during the cycle; these oocytes must reach a minimum size in order to mature and be retrieved for the following procedures (either IVF or ICSI) (6, 7). Collection of oocytes is done through follicular puncture by means of laparoscopic transvaginal surgery. Whereas for IVF the sperm concentration used is similar to natural conditions in order to mimic the natural environment of the oocyte in the Fallopian tubes, cases of severe male factor infertility may require the use of ICSI (8). This procedure consists in the injection of an individual spermatozoon into the mature oocyte, thus being actually fertilised by the embryologist, one by one (9). Sperm selection is based on morphology and motility [learn more about sperm selection through our previous post here]. Twenty-four hours after the procedure, embryologists verify whether fertilisation has occurred by looking for two pronuclei under the microscope. The presence of two pronuclei is the first sign of fertilisation (one from each gamete), and they are to be fused into a single one in a process called syngamy (10). In some cases, fertile couples with genetic family history attend clinics in order to avoid passing the disease down to their offspring. Such situation requires performing preimplantation genetic diagnosis (PGD). This analysis, usually carried out at day 3 or 5 of embryo development, identifies embryos affected with the genetic disorder (11). Before being transferred, embryos are cultured for 3, 5 or 6 days (depending on a variety of factors and upon the embryologist´s own criterion). Based on PGD results, only healthy embryos will be selected for transfer into the womb. The number of embryos to be transferred is determined by specific regulations in each country, being just one the ideal situation, whereas spare embryos are derived for cryopreservation. Once all the above is done, the extensive work of the embryologist normally gets to an end. It is important to highlight that every single procedure is carried out in the laboratory under strict hygienic and sterile conditions. Also, the use of any perfume, deodorant and alike is completely forbidden just to prevent potential volatile organic compounds (VOS) from affecting developing embryos. In the laboratory, the use of caps, masks and the appropriate scrubs is mandatory for embryologists, and embryos are quickly placed in incubators that mimic the natural environment of the uterus (12). As for the timelines, embryologists work in shifts with the purpose of being available twenty-four hours a day, seven days a week. Assisted reproduction treatments must follow a strict timeline given the fact that procedures must be done at an exact time point in order to guarantee their success until transfer day. As a consequence, the work of embryologists is intense and often requires long-hour shifts. CONCLUSIONS Embryologists are extensively-trained professionals with wide experience in assisted reproduction technologies (ART) (1). To increase their knowledge and technical skills to perform ART-related procedures, they are continually trained in new methodologies and go through continuous education in the field of embryology (2). These highly-qualified professionals are in charge of the routine maintenance of the laboratory and equipment, sperm handling and processing for IUI, IVF and ICSI, cryopreservation of biological material, tissue sampling for biopsies for genetic analyses and PGD, or even embryo culture, among other tasks (2). These also include a strict and exhaustive record of the procedures for safety purposes. It is actually on the embryologists how to evaluate gametes and embryos, and to choose which ones are the most appropriate to be used and which should be kept in case future use is needed. It is without question that embryologists work tirelessly to help families achieve their dream of bringing a healthy baby home. REFERENCES
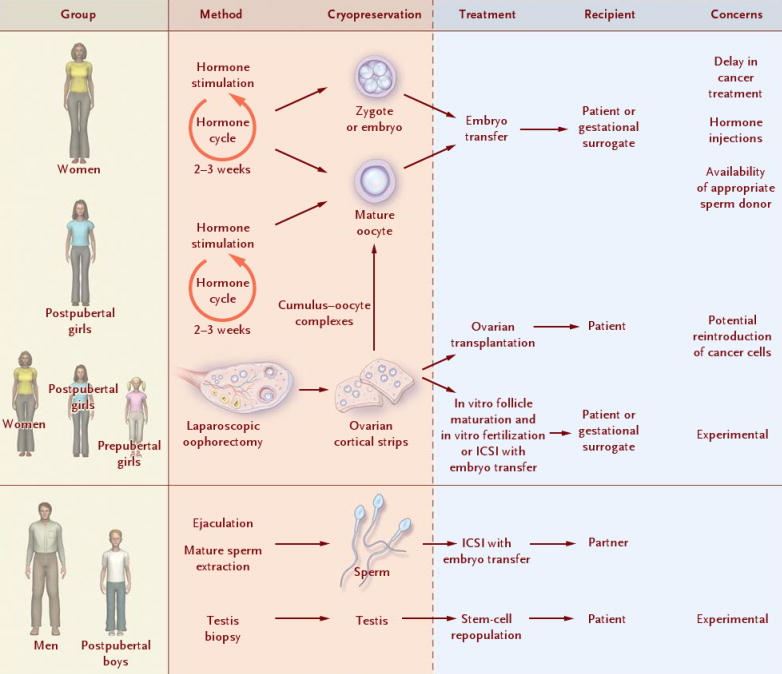
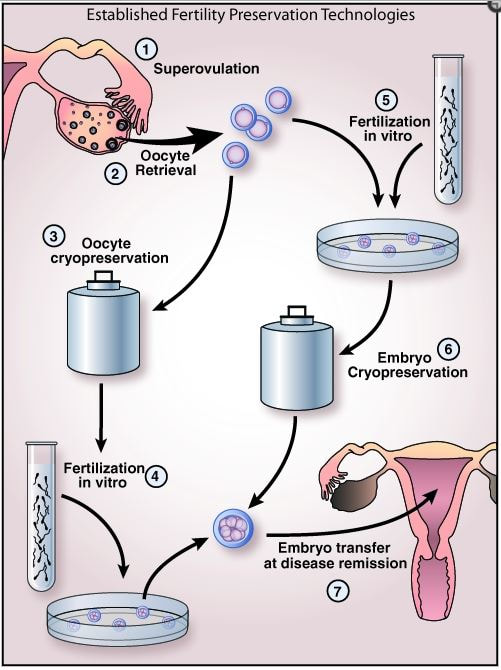
1-Medicine ASiR. The Alpha Consensus Meeting on the professional status of the clinical embryologist: proceedings of an expert meeting. Reprod Biomed Online. 2015;30(5):451-61. 2-Go KJ. 'By the work, one knows the workman': the practice and profession of the embryologist and its translation to quality in the embryology laboratory. Reprod Biomed Online. 2015;31(4):449-58. 3-Allen, N. C., Herbert, C. M., Maxson, W. S., Rogers, B. J., Diamond, M. P., & Wentz, A. C. (1985). Intrauterine insemination: A critical review. Fertility and Sterility, 44(5), 569-580. 4-Ombelet W, Van Robays J. Artificial insemination history: hurdles and milestones. Facts Views Vis Obgyn. 2015;7(2):137-43. 5-Fraser LR. Sperm capacitation and the acrosome reaction. Hum Reprod. 1998;13 Suppl 1:9-19. 6-Brinsden PR. Superovulation strategies in assisted conception. In: Brinsden PR. A Textbook of In Vitro Fertilization and Assisted Reproduction: The Bourn Hall Guide to Clinical and Laboratory Practice. Cambridge: Taylor and Francis group; 2005. p. 177-188. 7-Ventura-Juncá P, Irarrázaval I, Rolle AJ, Gutiérrez JI, Moreno RD, Santos MJ. In vitro fertilization (IVF) in mammals: epigenetic and developmental alterations. Scientific and bioethical implications for IVF in humans. Biol Res. 2015;48:68. 8-Eftekhar M, Mohammadian F, Yousefnejad F, Molaei B, Aflatoonian A. Comparison of conventional IVF versus ICSI in non-male factor, normoresponder patients. Iran J Reprod Med. 2012;10(2):131-6. 9-Simopoulou M, Gkoles L, Bakas P, Giannelou P, Kalampokas T, Pantos K, et al. Improving ICSI: A review from the spermatozoon perspective. Syst Biol Reprod Med. 2016;62(6):359-71. 10-Elder K, Dale B. Sperm-oocyte interaction. In: In-Vitro Fertilization. New York: Cambridge University Press; 2011. p. 50-63. 11-Beyer CE, Willats E. Natural selection between day 3 and day 5/6 PGD embryos in couples with reciprocal or Robertsonian translocations. J Assist Reprod Genet. 2017;34(11):1483-92. 12-Agarwal N, Chattopadhyay R, Ghosh S, Bhoumik A, Goswami SK, Chakravarty B. Volatile organic compounds and good laboratory practices in the in vitro fertilization laboratory: the important parameters for successful outcome in extended culture. J Assist Reprod Genet. 2017;34(8):999-100. Author: María Caballero "(..) it is important to choose the appropiate fertility preservation procedure according to the treatment". Once someone is diagnosed with any kind of disease, the first step is to decide the appropriate treatment. In many cases, such treatment can seriously affect the reproductive capacity, and so it is important to consider the option of fertility preservation (FP) when it comes to individuals at reproductive age or prepubertal children (2, 3) (Figure 1). Although most of the articles about FP are related to oncology patients (4, 5, 6, 7), there are other diseases and situations in which considering FP may be also important. Some of these diseases include autoimmune diseases like systemic lupus erythematosus (8) or genetic conditions that can cause premature ovarian insufficiency in women, such as Turner’s syndrome or fragile X-related mental retardation, or hypogonadism and azoospermia/oligozoospermia in men, like in individuals with Klinefelter’s syndrome (reviewed in 2). Moreover, other circumstances exist in which FP must be considered, such as sex reassignment surgeries (2) or a variety of social reasons that may end up hampering or delaying parenthood to otherwise perfectly fertile couples (9). Below, currently available procedures for both women and men are addressed, as well as other procedures still at experimental stage. AVAILABLE FERTILITY PRESERVATION PROCEDURES Male FP Nowadays, two options for FP in males can be found: gonadal shielding and sperm cryopreservation. In cases in which radiotherapy is unavoidable, gonadal shielding may result helpful since it seems to mitigate the effects of radiation, especially in children (10, 11). This procedure consists in covering the testes with a lead block or apron to avoid direct exposure to radiation (10); however, it does not completely prevent from radiation damage. Therefore, the best option for FP in pubertal and adult males is most likely sperm cryopreservation (Figure 1). Even though the sample is usually collected by masturbation, it may be also obtained by other methods including testicular sperm extraction (TESE) or electroejaculation. These methods are commonly applied in cases in which the previous approach is not possible due to specific reasons, such as certain physical/psychological conditions or even religious beliefs (2, 11). Female FP Several options are currently available for FP in female patients, including oocyte and embryo cryopreservation, ovarian transposition or gonadal shielding. These options are more invasive and time-consuming than for the male counterpart, and so it is important to choose the appropriate FP procedure according to the treatment (3). Oocyte and embryo cryopreservation have been established as standard methods by the American Society of Reproductive Medicine since 2012 (3, 12) (Figure 2). Between these two techniques, oocyte cryopreservation is usually the preferred one (2); even though embryo cryopreservation is offered to women with a partner and shows great results, it may result in issues past some years; for instance, if the couple divorces or the patient does not recover from the disease (12). Both methods require medical ovarian stimulation at the beginning, as well as follicular monitoring and transvaginal oocyte retrieval. The whole process can take around two weeks (3, 11). Sometimes, harvesting of immature oocytes and the subsequent in vitro maturation may be indicated for those patients who cannot undergo controlled ovarian stimulation, like prepubertal girls or women suffering from polycystic ovarian syndrome (PCOS) (2, 13). Once the oocyte matures, it will be eventually cryopreserved (to learn more about oocyte cryopreservation, read our post on “Social egg freezing”). In those cases in which oocyte/embryo cryopreservation is not possible due to the urge to commence the treatment, patients may also resort to the other methods. Both gonadal shielding and ovarian transposition are indicated when radiation on the pelvic area is necessary (11, 15). Ovarian transposition requires surgical intervention to remove the ovaries and fix them, usually to the anterolateral abdominal wall. In these cases, it is recommended to collect a biopsy sample of ovarian tissue, since certain risk of failure is expected (12). EXPERIMENTAL TECHNIQUES AND FUTURE PROSPECTS The only available method for FP in prepubertal male children so far is gonadal shielding. However, the option of testicular tissue cryopreservation (TTC) is sometimes offered by some clinics, as well. This technique remains experimental and there are currently no reports on reimplantation of testicular tissue or achievement of pregnancy (11). Due to the risk of reintroducing malignant cells (2), TTC still depends on the improvement of other techniques for the maturation of spermatogonial stem cells into mature sperm, which is necessary for current assisted reproduction techniques (IVF and ICSI) (3). Regarding female FP methods, the experimental ovarian tissue cryopreservation technique is also offered by some clinics, and it is the only option for prepubertal females and for those patients who cannot delay the initiation of the treatment (3). In this technique, part of the ovarian cortex with the primordial follicles is harvested and vitrified (4). Certain studies have demonstrated successful pregnancies in patients with transplanted ovarian tissue (3) (to learn more about ovarian tissue cryopreservation, read our post “Ovarian tissue cryopreservation. Current procedures and outcomes”). Treatments with gonadotropin-releasing hormone agonists (GnRHa) are a common practise in different situations, including certain cases of infertility or sex hormone-related issues. FP approaches required in cases of chemotherapy treatments may include administration of GnRHa, since they have been proved to be efficient in keeping ovaries quiescent and reducing their regular activity. This means inhibition of the so-called hypothalamic-pituitary-gonadal axis, which is the main hormone pathway regulating ovarian and follicular activity. By administering GnRHa’s, it would be possible to reduce the blood flow to the ovary, thus preserving follicles (4), so that they can grow and mature later on once chemotherapy treatment is over. However, evidence for its efficiency is still relatively low, and so further research is still necessary on this field (4, 16). Looking into the future, potential prospectives for female FP may include in vitro activation of cryopreserved follicles within the ovarian tissue, in vitro follicle culture and development of an artificial ovary into which primordial follicles could be eventually transplanted and cultured (2). The use of stem cells has been demonstrated to have promising results in the generation of both male and female artificial gametes (2, 17). Embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs) and embryonic germ cells (EGCs) are different types of stem cells with distinct degrees of pluripotency; this means they are able to differentiate into any (or almost any) cell type and tissue present in the body (17). In human embryos, primordial germ cells (PGCs) represent the earliest stage of gametes that, after a series of reprogramming changes and differentiation, will result in sperm or oocytes in the adult. Using the above-mentioned pluripotent (or totipotent) cells, several studies have reported it possible to generate germ cells that will eventually differentiate into fully functional sperm and eggs (17). On the other hand, the use of PGCs present in the gonads of the progenitors, which are spermatogonial stem cells (SSC) in the male and ovarian stem cells (OSC) in the female, have been also studied for artificial gamete generation by in vitro propagation of SSC and by in vitro propagation and re-aggregation of OSC in the ovarian tissue (reviewed in 17). CONCLUSIONS FP is an essential issue to keep in mind in situations in which the fertility capacity is at stake, which usually happens when undergoing certain medical treatments (or other circumstances). Currently, there exist several available options for patients and people, both at prepubertal and reproductive age, who need to preserve their fertility. Even though the most used one is gamete cryopreservation, other experimental techniques, such as the ovarian/testicular tissue cryopreservation, should be born in mind. There is still a clear need for improvement of those experimental techniques, since every disease and situation are different, and because the use of gamete cryopreservation is not always a viable option. REFERENCES
1. Jeruss JS, Woodruff TK. Preservation of Fertility in Patients with Cancer. N Engl J Med. 2009;360(9):902-911. 2. Martinez F. Update on fertility preservation from the Barcelona International Society for Fertility Preservation–ESHRE–ASRM 2015 expert meeting: indications, results and future perspectives. Hum Reprod. 2017;32(9):1802-1811. 3. Burns KC, Hoefgen H, Strine A, Dasgupta R. Fertility preservation options in pediatric and adolescent patients with cancer. Cancer 2018. 4. Harada M, Osuga Y. Fertility preservation for female cancer patients. Int J Clin Oncol. 2018. 5. Vakalopoulos I, Dimou P, Anagnostou I, Zeginiadou T. Impact of cancer and cancer treatment on male fertility. Hormones (Athens). 2015;14(4):579-589. 6. Kemertzis MA, Ranjithakumaran H, Hand M, Peate M, Gillam L, McCarthy M, Super L, McQuillan S, Drew S, Jayasinghe Y, Orme L. Fertility Preservation Toolkit: A Clinician Resource to Assist Clinical Discussion and Decision Making in Pediatric and Adolescent Oncology. J Pediatr Hematol Oncol. 2018;40(3):e133-e139. 7. Joshi S, Savani BN, Chow EJ, Gilleece MH, Halter J, Jacobsohn DA, Pidala J, Quinn GP, Cahn JY, Jakubowski AA, Kamani NR, Lazarus HM, Rizzo JD, Schouten HC, Socie G, Stratton P, Sorror ML, Warwick AB, Wingard JR, Loren AW, Majhail NS. Clinical guide to fertility preservation in hematopoietic cell transplant recipients. Bone Marrow Transplant. 2014;49(4):477-484. 8. Choux C, Cavalieri M, Barberet J, Samson M, Bonnotte B, Fauque P, Sagot P. [Immunosuppressive therapy and fertility preservation: Indications and methods]. Rev Med Interne 2018. 9. Daniluk JC, Koert E. Childless women's beliefs and knowledge about oocyte freezing for social and medical reasons. Hum Reprod. 2016;31(10):2313-2320. 10. Sayan M, Cassidy RJ, Butker EE, Nanda RH, Krishnamurti L, Khan MK, Esiashvili N. Gonadal shielding technique to preserve fertility in male pediatric patients treated with total body irradiation for stem cell transplantation. Bone Marrow Transplant. 2016;51(7):997-998. 11. Saraf AJ, Nahata L. Fertility counseling and preservation: considerations for the pediatric endocrinologist. Transl Pediatr. 2017;6(4):313-322. 12. Donnez J, Dolmans MM. Fertility Preservation in Women. N Engl J Med. 2018;378(4):400-401. 13. Hirshfeld-Cytron J, Gracia C, Woodruff TK. Nonmalignant diseases and treatments associated with primary ovarian failure: an expanded role for fertility preservation. J Womens Health (Larchmt). 2011;20(10):1467-1477. 14. Shirasawa H, Terada Y. In vitro maturation of human immature oocytes for fertility preservation and research material. Reprod Med Biol. 2017;16(3):258-267. 15. Kanda Y, Wada H, Yamasaki R, Kawamura K, Ishihara Y, Sakamoto K, Ashizawa M, Sato M, Machishima T, Terasako-Saito K, Kimura S, Nakasone H, Kikuchi M, Yamazaki R, Kanda J, Kako S, Nishida J, Tsunoda H, Omori Y, Nakazawa M, Tanaka O. Protection of ovarian function by two distinct methods of ovarian shielding for young female patients who receive total body irradiation. Ann Hematol. 2014;93(2):287-292. 16. Senra JC, Roque M, Talim MCT, Reis FM, Tavares RLC. Gonadotropin-releasing hormone agonists for ovarian protection during cancer chemotherapy: systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2018;51(1):77-86. 17. Vassena R, Eguizabal C, Heindryckx B, Sermon K, Simon C, van Pelt AM, Veiga A, Zambelli F; ESHRE special interest group Stem Cells. Stem cells in reproductive medicine: ready for the patient? Hum Reprod. 2015;30(9):2014-2021. "Patients must be aware that laws and regulations from other countries will likely be different from home" Author: Raquel Pillado WHAT IS “CROSS BORDER REPRODUCTIVE CARE”? Accessibility of patients suffering from infertility to appropriate treatments may greatly vary depending on which country or region they reside in. The availability of appropriate care, its quality, cost or regulatory restrictions may push patients into seeking treatment abroad (2). Furthermore, these limitations may also include privacy issues, involving the social stigma that may be associated with certain treatments (surrogacy, sex selection, preimplantation genetic diagnosis…) in certain cultures. Such situation is known as Cross Border Reproductive Care (CBRC), and it is defined as the practice of travelling to another region or to a different country to receive fertility treatments (3). CBRC is also commonly known as reproductive, procreative or fertility tourism because many patients take the chance to combine their fertility treatments with regular tourist activities. Also, some authors refer to this phenomenon as “reproductive exile”, because they see it as the patient being pushed to this alternative in order to achieve their desire to become parents (4). PREFERRED DESTINATIONS FOR CBRC The destination choice often represents an arduous task for patients due the travel logistics, including the geographical and cultural proximity and the specificities of treatments required (5). As a result, some companies have appeared to offer assistance for patients interested in CBRC. These companies help their clients to organize their trip, from accommodation booking to arranging all medical meetings. Moreover, some clinics directly advertise specific programs for international clients, and they even include sections on their websites specifically dedicated to provide information on every step of the process and tourist attractions. Nowadays, the main global markets or hubs for CBRC industry are Denmark for sperm donation, Belgium for in vitro fertilization (IVF) or Spain and Czech Republic for egg and embryo donation. Likewise, the most popular destinations for fertility preservation seem to be Denmark and Belgium in Europe and the United States in the American continent. This country is also a preferred choice for cryopreservation of gametes, embryos and ovarian and testicular tissues. Furthermore, and along with Russia and India, it is a popular destination for preimplantation genetic diagnosis (PGD) and sex selection procedures (3). The selected country depends on where patients are originally from and the treatment they seek (Table 1). For example, North Americans visit countries with comparative lower prices, Britons travel to other European countries looking for sperm, eggs and embryo donors and some Arab couples engage in “return reproductive tourism”* (6) to the Middle East (5). FACTORS TO CONSIDER BEFORE TRAVELLING ABROAD FOR CBRC Patients must be aware that laws and regulations from other countries will likely be different from home. There are two main ways of regulating the practice and the access to ART. First, proper regulation can be achieved through sets of rules or guidelines followed by the practitioners on a voluntary basis. Usually these guidelines are developed by professional organizations related to the field. Secondly, governmental legislation, which implies specific rules in the shape of laws ruling the use of ART and legal penalties for those who violate them (7). It is important to study the specific ART legislation and/or guidelines of both the origin and chosen destination countries. In order to find information about these regulations the first step is to find out whether there is a national independent regulator overseeing ART practice. For example, in the United States, United Kingdom and India these are the American Society of Reproductive Medicine (ASRM) (8), the Human Fertilisation and Embryology Authority (9), and the National Registry of Assisted Reproductive Technology (ART) Clinics and Banks (10), respectively. These organizations usually provide information concerning national legislation and/or guidelines, and some of them even offer data of officially approved clinics. In the case of the US and UK there are legislation and guidelines, while in India there are only guidelines (6). Regarding this country, the National Registry is still in the process of verifying whether clinics follow these guidelines and awarding them with official certificates accordingly (10). The information obtained from guidelines/legislation will provide a general idea about the standards required for ART in those countries. When looking for a gestational carrier or sperm, eggs or embryos donors, it is important to specifically look into the requirements asked to potential donors and gestational carriers (genetic tests, STD reports, etc.). Additionally, in the case of surrogacy it is also advisable to double check any law related to legal rights over the offspring, both in the surrogate mother and the couple´s respective countries of origin (for more information about surrogacy, read one of our previous posts here). Lastly, the language barrier must not be forgotten. Before travelling, one should verify with the fertility centre the availability of interpreters and the ability of the staff to speak the required language. Also, it will be necessary to ask the centre to provide copies of every document in that language (certificates, bills, informed consents, etc.). THE FUTURE OF CBRC Travelling between countries and easy access to information is nowadays part of our routine life due to globalization and internet. As a result, it is very attractive for assisted reproduction clinics to offer their services to international clients. However this market has quickly developed without any international regulation ruling over it. As a future objective to improve CBRC, governs and health organizations should work on an international regulation in order to protect both patients and professionals (2). Ideally, the creation of an international joint database and standardization of data collection would help to improve ART efficiency (11). In addition, special attention should be given to writing up laws in order to: [1] prevent exploitation of gamete donors and/or gestational carriers in destination countries; and [2] avoid the price rise that would make it more difficult for local patients to access ART treatments. CONCLUSIONS Even though CBRC seems to provide a solution for most patients to access to certain treatments, this option needs a much clearer international regulation. Such improvement should help to collect data to increase the current knowledge on ART field, which will enable professionals to offer a better care in the future. Furthermore, governments should probably implement specific measurements to prevent misinformation to international patients. Simultaneously, this would help defending local citizens from exploitation in specific cases; for instance, those in which a third party is involved such as gestational carriers or donors. Finally, the decision of signing on CBRC should be made after extensive research about the treatment one wishes to undergo and the regulations in the destination country. *Situation in which a couple or an individual who lives abroad returns home specifically for ART treatments due to a variety of cultural, moral and psychological reasons (6). REFERENCES 1. https://www.babygest.es/turismo-reproductivo/que-es-el-turismo-reproductivo-o-de-fertilidad/ (10-03-2018) 2. Storrow RF (2011) Assisted reproduction on treacherous terrain: the legal hazards of cross-border reproductive travel. Reproductive BioMedicine Online (2011) 23, 538– 545. 3. Salama M (2014) Cross Border Reproductive Care (CBRC): A Global Perspective. Obstet Gynecol Int J 1(2): 00008. DOI: 10.15406/ ogij.2014.01.00008. 4. Inhorn and Patrizio (2009) Rethinking reproductive ‘‘tourism’’ as reproductive ‘‘exile’’, American Society for Reproductive Medicine, Fert and Steril, 92 (3): 904-906. 5. Gürtin ZB et al (2011) Introduction: travelling for conception and the global assisted reproduction market. Reprod Biomed Online, 23(5): 535-537. 6. Inhorn MC (2011) Diasporic dreaming: return reproductive tourism to the Middle East. Reprod Biomed Online, 23(5): 582 - 91. 7. Präg P and Mills MC (2017) Assisted Reproductive Technology in Europe: Usage and Regulation in the Context of Cross-Border Reproductive Care. In: Kreyenfeld M, Konietzka D (eds) Childlessness in Europe: Contexts, Causes, and Consequences. Demographic Research Monographs (A series of the Max Planck Institute for Demographic Research). Springer, Cham. 8. American Society For Reproductive Medicine. Montgomery Highway Birmingham, Alabama. Availability: http://www.asrm.org/arm/ 9. Human Fertilization and Embryology Authority. 10 Spring Gardens London Availability: https://www.hfea.gov.uk/treatments/eexplor-all-treatments/fertility-treatment-abroad/ 10. National Registry of Assisted Reproductive Technology (ART) Clinics and Banks in India. ICMR. Ansari Nagar, New Delhi Availability: http://www.icmr.nic.in/icmrnews/art/art.htm 11. Shenfield F (2011) Implementing a good practice guide for CBRC: perspectives from the ESHRE Cross-Border Reproductive Care Taskforce. Reprod BioMed Online 23: 657– 664. “Most of the couples with no children are found in developing countries". Author: Iñaki Arroyos INTRODUCTION Nowadays, assisted reproduction technologies (ART) are routine medical procedures that help couples with problems to conceive naturally. Since the birth of Louise Brown in 1978, the first in vitro fertilization (IVF) baby, important advances in the field of infertility have been achieved. For instance, ovulation can now be controlled by the use of modern and improved drugs, and the latest enhancements in culture conditions allow to better mimic the actual natural environment for embryo development. Additionally, in recent years new technologies have arisen that allow to perform better embryo selection, and remarkable progress in cryopreservation has been achieved, too, thus resulting in improved pregnancy rates for current treatments. Nevertheless and in spite of these advances, there is still progress to be made: what kind of challenges will ART face in the upcoming years? The following points are three of these challenges: 1. Biomarkers 2. In vitro-generated gametes 3. ART in developing countries FUTURE CHALLENGES TO FACE 1. Biomarkers A biomarker is defined as “a characteristic that is objectively measured and evaluated as an indicator of normal biologic processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention” (2). Biomarkers are important in reproductive medicine because of their potential to: (a) reveal different causes of infertility; and (b) improve treatments and clinical outcomes. Consequently, research on biomarkers is quite an active area in reproduction and other medicine disciplines. The term OMICS collectively refers to a group of technologies that aim to study different types of molecules and their relation with biological functions. Relevant examples of such areas are epigenomics, genomics, transcriptomics, proteomics and metabolomics, among others (3). The appearance of these research fields helped scientist to find new biomarkers for different subjects. However, regarding reproduction medicine this effort has resulted uncoordinated (4), and so there is currently a lack of studies able to validate the actual quality of biomarkers. Within the huge area of research on ART biomarkers, two topics of paramount importance are presented below: 1. Embryo selection A key factor in IVF is to determine which embryo exhibits the highest implantation potential. Even though a great progress has been made in order to clarify the process of embryo development, the actual physiology of embryos and the contributing factors for successful implantation are not yet entirely understood. Embryos generated in the laboratory present different implantation potential to each other (5); it just so happens that embryologists may not know for sure which embryo has the highest implantation potential for transfer. This leads to performing multiple embryo transfers that may result in multiple pregnancies, which entail associated problems such as increased perinatal mortality for twins and triplets (6). In order to avoid such problems, different embryo scoring systems have been developed to allow a better and more accurate embryo selection. However, since these systems are based on morphological traits they do not offer objective and reliable data, and so classification can actually vary depending on the observer. The introduction of time-lapse allowed to follow embryo development from zygote state up to blastocyst in a continuous fashion. Additionally, the improved culture conditions have made possible to perform single embryo transfer (SET) much more frequently, which avoids multiple pregnancies. These advances undoubtedly represented a leap forward in ART, and yet sometimes there are still difficulties when it comes to selecting the best embryo. The development of OMICS unveiled the existence of a series of factors that may improve embryo selectivity, although related new technologies are actually far from being used on a daily basis in the clinical world. 2. Sperm biomarkers Male fertility problems represent approximately 50% of infertility (7), reason why it is important to accurately diagnose male factor disorders. Semen analysis is currently one of the most common tools for sperm assessment; however, even though it is certainly helpful for making a primary diagnosis, it lacks objectivity and accuracy (8). In fact, false negatives are occasionally reported to patients who still fail to conceive. Perhaps the most successful approach to improve semen analyses has been finding certain biomarkers that provide extra information about sperm quality. Spermatozoa are not just a means to deliver the paternal genetic material into the oocyte, but they also provide instructions for the proper embryo development. Such instructions are epigenetically encoded, which means the genome is modified depending on the association and regulation of specific proteins to DNA. For instance, differences in protamin ratio (proteins essential for DNA compaction in the sperm nucleus) have been found between fertile and infertile men (9). Other proteins found in the seminal plasma may also serve as biomarkers for the regulation of specific genes, thus improving the accuracy of semen analyses. Taken together, these and other similar observations make reasonable to think that epigenomics and proteomics may provide useful information for sperm assessment. The use of intracytoplasmic sperm injection (ICSI) has expanded during the last years. However, there is still a need of a biomarker able to discriminate which individual spermatozoa is the most suitable for injection. Different methods based on the quality of sperm DNA or percentage of cell death in a given semen population have been developed, and yet they are not able to select the best individual sperm for ICSI. 2. In vitro-generated gametes What about those patients who constantly fail to conceive through IVF or who have lost the capacity to produce gametes due to a disease or a specific condition such as some sort of cancer? Will they actually have any chance in the future to pass on their genetic material? Stem cells may provide an answer for these people. Stem cells have the potential to generate any type of cell in the organism. In order to generate artificial gametes, two types of stem cells can be used: 1. Adult stem cells from male and female gonads (that naturally differentiate into either sperm or eggs). 2. Pluripotent stem cells. Two types: i) embryonic stem cells (ESCs), derived from the inner cell mass of the embryo (10) and able to generate any cell lineage; and ii) induced pluripotent stem cells (iPSCs), obtained from somatic cells by overexpression of specific genes (11, 12), which can also turn them into any type of cell. Despite previous achievements in generating gametes from somatic cells in mice, there are serious technical and ethical issues that nowadays keep this approach away from the clinical practice. However, this scientific breakthrough represented new challenges in ART: - Transplantation of stem cells into gonads may be no longer necessary, but gamete development in the laboratory still requires certain factors to substitute the function of ovaries and testes. - Molecular interactions and signalling pathways are different in men and mice, and so improving the technique is absolutely necessary. “Cells from a man’s skin could theoretically be used to create eggs that are fertilized with a partner’s sperm, then nurtured in the womb of a surrogate” (1). This science-fiction scenario may be actually possible within the next twenty-five years. 3. ART in developing countries Epidemiological studies show that the majority of couples with no children live in developing countries (13). By contrast, high-quality ART treatments are almost exclusively performed in western countries (14). Developing countries still have quite a limited offer of treatments due to their high cost (15). Among all technologies available in developing countries, IVF constitutes the main treatment, followed by intrauterine insemination (IUI). However, these techniques are still unaffordable for the majority of the population in certain countries. Over the years, different strategies have arisen to provide more affordable solutions. The addition of natural cycles (16) or minimal/mild stimulations in order to reduce the amounts of hormones (17), in combination with intravaginal culture of oocytes (INVO) (18) or new developed devices offer reachable alternatives at a lower cost (19). High prices of treatments is not the only reason of the slow progress of ART in developing countries. Their fertility issues have not drawn western countries’ attention, and so interest in this research field has decreased. Overpopulation is, perhaps, one of the main reasons; countries lacking a well-developed social structure (healthcare or education, among others) and a strong economy able to sustain the present population should not foster population growth. On the contrary, it should be controlled; by doing so, life quality of next generations could be guaranteed. Additionally, developing areas have other priorities as thousands of people are still suffering from diseases such as malaria, tuberculosis or HIV. Interesting enough is the fact that the main cause for infertility is the high incidence of pelvic infections; therefore, approaches to reduce fertility issues in these countries should be firstly focused on decreasing sexually transmitted diseases (STDs) (20). CONCLUSIONS Within the last forty years assisted reproduction has experienced a true revolution. Looking back in time, seventy years ago all these approaches could have been regarded as science fiction, but nowadays infertility can be effectively treated. Whereas the main disadvantage is the high cost of assisted reproduction procedures, new lines of research are currently focused on reducing it, and new methodologies are becoming more easily available. Perhaps, in twenty-five years, ART as a whole maybe a worldwide affordable approach. REFERENCES 1. Cyranosky D. Stem cells: Egg engineers. Nature. 2013; 500:392-394. 2. Woodcock J. Assessing the clinical utility of diagnostics used in drug therapy. Clin Pharmacol Ther. 2010; 88:765–73. 3. Rocío Rivera Egea, Nicolás Garrido Puchalt, Marcos Messeguer Escrivá, Alex C Varghese. OMICS: Current and future perspectives in reproductive medicine and technology. Journal of Human Reproductive Sciences. 2014; 7(2): 73-92. 4. Palmer Stephen S, Barnhart Kurt T. Biomarkers in Reproductive Medicine: The promise, and can it be fulfilled? Fertil Steril. 2013; 99(4): 954-962. 5. Bromer JG, Seli E. Assessment of embryo viability in assisted reproductive technologies: shortcomings of current approaches and the emerging role of metabolomics. Curr Opin Obstet Gynecol. 2008; 20:234-241. 6. Lieberman B, Ali R, and Rangarajan S. Towards the elective replacement of a single embryo transfer (eSET) in the United Kingdom. Human Fertility. 2007; 10(2):123-7. 7. Mosher WD, Pratt WF. Fecundity and infertility in the United States: incidence and trends. Fertil Steril.1991; 56:192-3. 8. Murray KS, James A, McGeady JB, Reed ML, Kuang WW, et al. The effect of the new 2010 World Health Organization criteria for semen analysis on male infertility. Fertil Steril 2012; 98:1428-31. 9. Carrell DT, Liu L. Altered protamine 2 expression is uncommon in donors of known fertility, but common among men with poor fertilizing capacity, and may reflect other abnormalities of spermiogenesis. J Androl 2001; 22:604-10. 10. Aguilar-Gallardo C, Poo M, Gomez E, Galan A, Sanchez E, Marques-Mari A, et al. Derivation, characterization, differentiation, and registration of seven human embryonic stem cell lines (VAL-3, -4, -5, -6M, -7, -8, and -9) on human feeder. In Vitro Cell Dev Biol Anim 2010; 46:317-26. 11. Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult broblast cultures by de ned factors. Cell 2006; 126:663-76. 12. Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 2007; 318:1917-20. 13. Rutstein SO, Iqbal HS. Infecundity, Infertility and Childessness in Developing Countries. DHS Comparative Reports, WHO. 2004; p24. 14. Sharma S, Mittal S, Aggarwal P. Management of infertility in low resources countries. BJOG. 2009; 116 (Suppl1): 77-83. 15. Murage A, Muteshi MC, Githae F. Assisted reproduction services provision in a developing country: time to act? Fertil Steril. 2011; 96(4):966-8. 16. Shahin AY. The problem of IVF cost in developing countries: has natural cycle IVF a place? Reprod Biomed Online. 2007; 15(1):51-6 17. Kim CH, Kim SR, Cheon YP, et al. Minimal stimulation using gonadotropin-releasing hormone (GnRH) antagonist and recombinant human follicle-stimulating hormone versus GNRH antagonist multiple-dose protocol in low responders undergoing in vitro fertilization/intracytoplasmic sperm injection. Fertil Steril. 2009; 92(6):2082-4 18. Lucena E, Saa AM, Navarro DE, et al. INVO procedure: minimally invasive IVF as an alternative treatment option for infertile couples. Sci World J. 2012; 2012:571-596. 19. Klerkx EPF, Janssen M, van Blerkom J et al. O-014. First pregnancies with a simplified IVF procedure: a crucial step to universal and accessible infertility care Human Reprod. Abstracts of the 29th Annual Meeting of the European Society of Human Reproduction and Embryology 2013; 28(Suppl1):i4-i6. 20. Ombelet W. Global access to infertility care in developing countries: a case of human rights, equity and social justice. F, V& in ObGyn. 2011; 3(4):257-266. "Only a small fraction of oocytes will reach the correct maturation stage and acquire the potential to be released and fertilized". Author: Edel Rocher 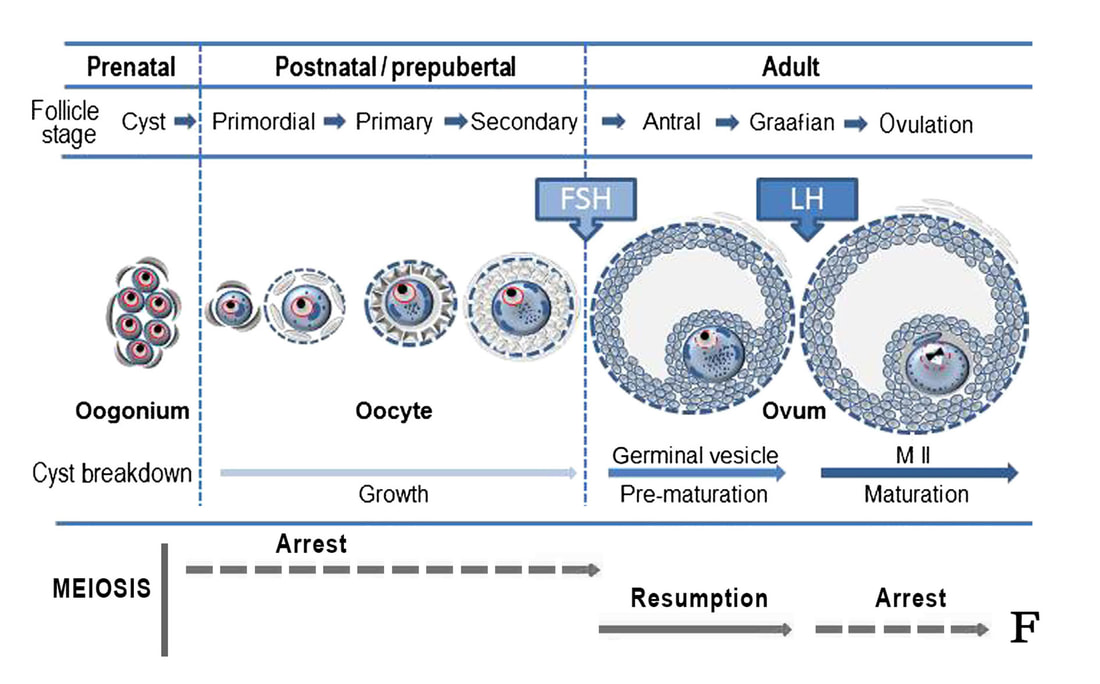 Figure 1. Representation of the parallel growth of follicles and maturation status of oocytes. During embryo development, germ cells are associated to each other in multinucleated cell accumulation called cysts. Each of these cysts contains multiple oogonia, the cells that will eventually become the oocytes. At birth, oogonia are already individualized as oocytes and surrounded by ovarian cells, forming a structure called primordial follicle. Some of the follicles eventually grow due to proliferation of the oocyte-surrounding cells (follicular cells), while oocyte development continues. For this to occur, an intricate balance of hormonal actions is needed, whose main characters are FSH and LH (modified from [1]). During assisted reproduction treatments ("cycles"), immature oocytes (or eggs) are oftentimes found. The following lines try to explain the reader why these cells are so frequent, and why they reduce the success rates of fertility treatments. The egg or oocyte is part of a cell aggregation found in the ovary referred to as follicle. Follicles receive and produce a series of hormone secretions that contribute to the maturation of these eggs, which are eventually released during ovulation. At birth, women normally present somewhat between 1 and 2 million ovarian follicles. These are called primordial follicles, each containing a primary oocyte (Figure 1). Only a small fraction of these oocytes will reach the correct maturation stage and acquire the potential to be released and fertilized. Oocytes and spermatozoa, the gametes, undergo a special kind of cell division called meiosis, which consists of two consecutive divisions, the end result being the reduction of the chromosome number to half. This means that, in the germline, a diploid human cell (46 chromosomes) eventually gives rise to four haploid cells (23 chromosomes each). During fertilization, each haploid gamete contributes half of the chromosome complement, thus generating a normal embryo. In a newborn, primary oocytes are paused at the beginning of the meiotic division (“meiotic arrest”), and remain so throughout childhood. Early stages of oocyte development include the germinal vesicle (GV) and metaphase I (MI) stages (Figure 2). Because these stages are part of the first meiotic division, the cell is still diploid and, therefore, it is still considered "immature". Once puberty is reached, a pituitary gonadotropin called follicle-stimulating hormone (FSH) promotes the growth of a cohort of follicles every month. But only those at the right stage of maturation will go on, along with their respective oocyte, which comes out of the arrest and resumes meiosis. By the time ovulation occurs, triggered by the surge of the luteinizing hormone (LH), oocytes have already completed the first of the two meiotic divisions, which means the primary oocyte has already divided into two cells: the first polar body and the secondary oocyte. The former is just a small cell that contains the corresponding chromosomes that result from the cell division. It never participates in embryogenesis, and so it has no genetic contribution to it. The latter in turn continues through the second meiotic division, until it arrests at MII (metaphase II) stage before ovulation (Figure 2). These oocytes (eggs) are considered mature, and they can be fertilized by sperm once ovulated. However, if fertilization does not occur, meiosis is never completed in the oocyte (2). MAIN ASPECTS NEEDED FOR THE EGG TO ACHIEVE FULL MATURATION Follicle growth and oocyte maturation are both influenced by circulating levels of gonadotropins (FSH and LH) and steroids (progesterone and oestrogens), among other factors. As parallel processes in time, they should accomplish certain events to ensure the formation of a fully mature egg by the time the follicle has acquired its maximum size ("Graafian follicle" = 18-21 mm) (3). Maturation status of the oocyte is defined by: 1. Nuclear maturation of the egg. This means the oocyte needs to reach a specific stage of meiotic division (MII stage), thus ensuring the appropriate chromosome number (genetic material) has been achieved (4). 2. Cytoplasmic maturation. In addition to the organization of the genetic material, the oocyte must complete a series of events such as distribution of mitochondria (the power source of the cell) or formation and relocation of the cortical granules (which will play a key role during fertilization), among others. This will guarantee the readiness of the egg for the sperm arrival (4). 3. Follicle maturation and growth. The follicle must increase its volume accordingly with the progression of the oocyte maturation; follicular cells are to be reorganized and differentiate into distinct layers to ensure blood supply and facilitate communication with the egg. Should any of these events fail to complete, fertilization will be greatly compromised (5). 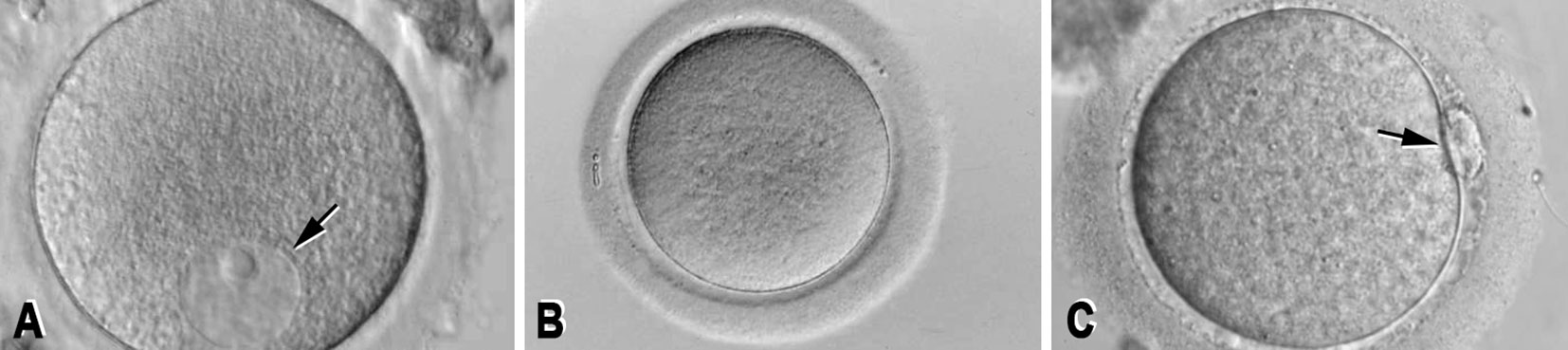 Figure 2. Different stages of oocyte maturation. (A) Germinal vesicle (GV). The nucleus of the oocyte is still visible at an eccentric location (arrow), indicative of an early stage of meiosis. (B) Metaphase I (MI). The nuclear envelope has disorganized and the chromosomes (not seen) are preparing for division. No other evident features can be clearly distinguished under the optical microscope. (C) Metaphase II (MII). First division of meiosis has been completed, evidenced by the presence of the first polar body (arrow). The oocyte is now arrested at the second division; the nucleus is not distinguishable because chromosomes are again prepared for division, should fertilization happen. 400x magnification (original images modified from [6]). POSSIBLE CAUSES FOR FAILURE IN EGG MATURATION DURING ASSISTED REPRODUCTION TREATMENTS During assisted reproduction cycles, women are subjected to controlled ovarian hyperstimulation (COH) by the application of a variety of protocols in order to promote the growth of multiple follicles. One of the main actors used in these stimulation protocols is the human chorionic gonadotropin (hCG), which causes the oocyte to progress through meiosis and take over the role of LH in triggering ovulation (7). The use of conventional protocols allows most of the oocytes obtained to be at MII stage by the time of egg collection. However, some of these eggs could remain immature despite ovarian stimulation (8). Some estimations have suggested that around 20%-30% of collected oocytes do not achieve full maturation (9), although such figures may actually be higher as a consequence of the stimulation of multiple follicles. In some rare cases, it is possible to observe a phenomenon known as "oocyte maturation arrest", in which oocytes keep arrested during the maturation process, thus not being able to complete it. As a result, there is an increased number of collected oocytes actually arrested at either GV or MI stage (10). Explanations for these rare cases include several possibilities: 1. It could be due to failure in ovarian stimulation response. Both LH activity and hCG timing are directly related to the meiotic progression of oocytes, so the abnormal function of the hormone stimulation mechanisms may negatively impact clinical outcomes. In these cases, results might be improved by the modification of doses, hCG forms or stimulation protocols in successive cycles (11). 2. Specific follicular cells may present defective signalling mechanisms, which would result in abnormal response to gonadotropins and the subsequent inability of the egg to progress through meiosis (12,13). 3. Oocytes unable to reach a minimum size cannot progress further than GV or MI stage. In those cases, the growth of the follicle is limited, which means follicular cells do not acquire the ability to respond to gonadotrophic signals and promote maturation up to MII stage of the oocyte (13). 4. The acquisition of "meiotic competence" is controlled by the so-called maturation promoting factor (MPF). The activity of this molecular machinery may be compromised in certain patients suffering from rare heritable molecular defects. In such cases, the end result could be the inability of oocytes to reach the mature stage (14). In spite of these hypotheses, only a few cases of oocyte maturation failure in ART treatments have actually been reported (11). Additionally, the modification of hCG doses, in vitro maturation (IVM) of oocytes and intracytoplasmic injection of MPF seem to be the best options to date to overcome meiotic arrest (15). Even in the most difficult cases, egg donation may be a potential solution. The approaches mentioned above are just suggestions and theoretical solutions; this means further research is needed in order to improve our knowledge on the actual causes for oocyte maturation failure. Only by doing so will we be able to better deal with this situation. REFERENCES
|
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media