|
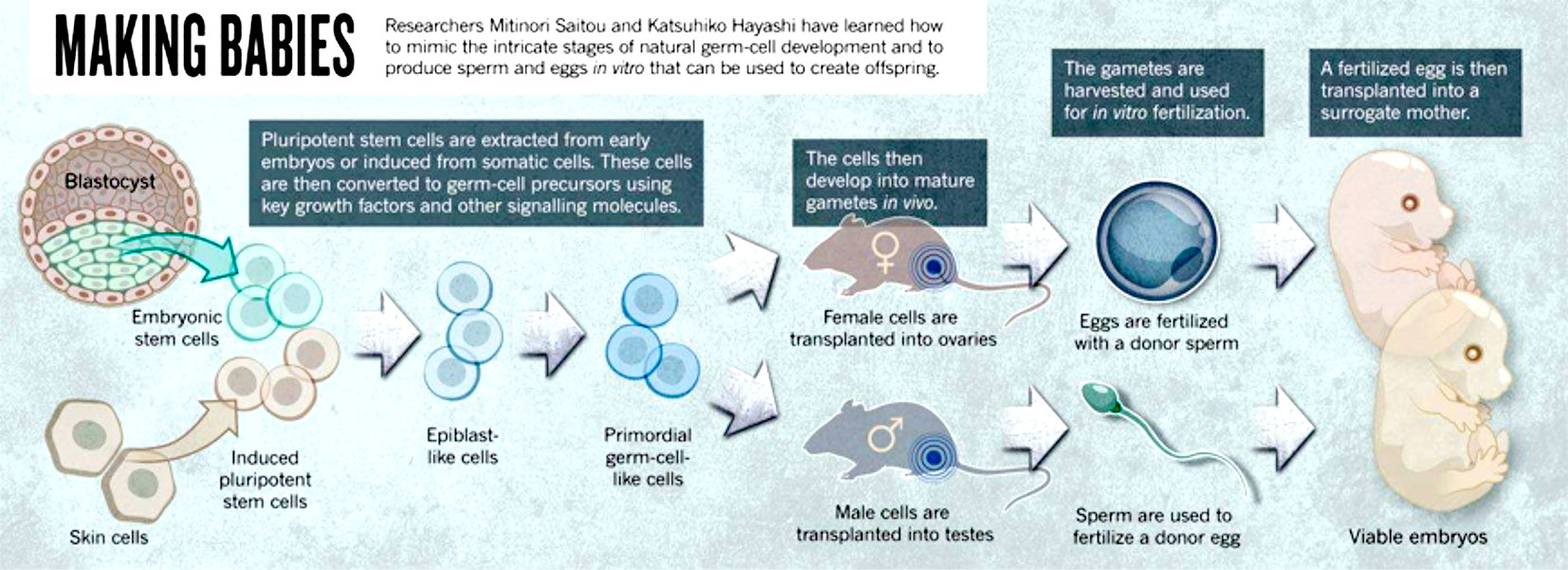
“Most of the couples with no children are found in developing countries". Author: Iñaki Arroyos INTRODUCTION Nowadays, assisted reproduction technologies (ART) are routine medical procedures that help couples with problems to conceive naturally. Since the birth of Louise Brown in 1978, the first in vitro fertilization (IVF) baby, important advances in the field of infertility have been achieved. For instance, ovulation can now be controlled by the use of modern and improved drugs, and the latest enhancements in culture conditions allow to better mimic the actual natural environment for embryo development. Additionally, in recent years new technologies have arisen that allow to perform better embryo selection, and remarkable progress in cryopreservation has been achieved, too, thus resulting in improved pregnancy rates for current treatments. Nevertheless and in spite of these advances, there is still progress to be made: what kind of challenges will ART face in the upcoming years? The following points are three of these challenges: 1. Biomarkers 2. In vitro-generated gametes 3. ART in developing countries FUTURE CHALLENGES TO FACE 1. Biomarkers A biomarker is defined as “a characteristic that is objectively measured and evaluated as an indicator of normal biologic processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention” (2). Biomarkers are important in reproductive medicine because of their potential to: (a) reveal different causes of infertility; and (b) improve treatments and clinical outcomes. Consequently, research on biomarkers is quite an active area in reproduction and other medicine disciplines. The term OMICS collectively refers to a group of technologies that aim to study different types of molecules and their relation with biological functions. Relevant examples of such areas are epigenomics, genomics, transcriptomics, proteomics and metabolomics, among others (3). The appearance of these research fields helped scientist to find new biomarkers for different subjects. However, regarding reproduction medicine this effort has resulted uncoordinated (4), and so there is currently a lack of studies able to validate the actual quality of biomarkers. Within the huge area of research on ART biomarkers, two topics of paramount importance are presented below: 1. Embryo selection A key factor in IVF is to determine which embryo exhibits the highest implantation potential. Even though a great progress has been made in order to clarify the process of embryo development, the actual physiology of embryos and the contributing factors for successful implantation are not yet entirely understood. Embryos generated in the laboratory present different implantation potential to each other (5); it just so happens that embryologists may not know for sure which embryo has the highest implantation potential for transfer. This leads to performing multiple embryo transfers that may result in multiple pregnancies, which entail associated problems such as increased perinatal mortality for twins and triplets (6). In order to avoid such problems, different embryo scoring systems have been developed to allow a better and more accurate embryo selection. However, since these systems are based on morphological traits they do not offer objective and reliable data, and so classification can actually vary depending on the observer. The introduction of time-lapse allowed to follow embryo development from zygote state up to blastocyst in a continuous fashion. Additionally, the improved culture conditions have made possible to perform single embryo transfer (SET) much more frequently, which avoids multiple pregnancies. These advances undoubtedly represented a leap forward in ART, and yet sometimes there are still difficulties when it comes to selecting the best embryo. The development of OMICS unveiled the existence of a series of factors that may improve embryo selectivity, although related new technologies are actually far from being used on a daily basis in the clinical world. 2. Sperm biomarkers Male fertility problems represent approximately 50% of infertility (7), reason why it is important to accurately diagnose male factor disorders. Semen analysis is currently one of the most common tools for sperm assessment; however, even though it is certainly helpful for making a primary diagnosis, it lacks objectivity and accuracy (8). In fact, false negatives are occasionally reported to patients who still fail to conceive. Perhaps the most successful approach to improve semen analyses has been finding certain biomarkers that provide extra information about sperm quality. Spermatozoa are not just a means to deliver the paternal genetic material into the oocyte, but they also provide instructions for the proper embryo development. Such instructions are epigenetically encoded, which means the genome is modified depending on the association and regulation of specific proteins to DNA. For instance, differences in protamin ratio (proteins essential for DNA compaction in the sperm nucleus) have been found between fertile and infertile men (9). Other proteins found in the seminal plasma may also serve as biomarkers for the regulation of specific genes, thus improving the accuracy of semen analyses. Taken together, these and other similar observations make reasonable to think that epigenomics and proteomics may provide useful information for sperm assessment. The use of intracytoplasmic sperm injection (ICSI) has expanded during the last years. However, there is still a need of a biomarker able to discriminate which individual spermatozoa is the most suitable for injection. Different methods based on the quality of sperm DNA or percentage of cell death in a given semen population have been developed, and yet they are not able to select the best individual sperm for ICSI. 2. In vitro-generated gametes What about those patients who constantly fail to conceive through IVF or who have lost the capacity to produce gametes due to a disease or a specific condition such as some sort of cancer? Will they actually have any chance in the future to pass on their genetic material? Stem cells may provide an answer for these people. Stem cells have the potential to generate any type of cell in the organism. In order to generate artificial gametes, two types of stem cells can be used: 1. Adult stem cells from male and female gonads (that naturally differentiate into either sperm or eggs). 2. Pluripotent stem cells. Two types: i) embryonic stem cells (ESCs), derived from the inner cell mass of the embryo (10) and able to generate any cell lineage; and ii) induced pluripotent stem cells (iPSCs), obtained from somatic cells by overexpression of specific genes (11, 12), which can also turn them into any type of cell. Despite previous achievements in generating gametes from somatic cells in mice, there are serious technical and ethical issues that nowadays keep this approach away from the clinical practice. However, this scientific breakthrough represented new challenges in ART: - Transplantation of stem cells into gonads may be no longer necessary, but gamete development in the laboratory still requires certain factors to substitute the function of ovaries and testes. - Molecular interactions and signalling pathways are different in men and mice, and so improving the technique is absolutely necessary. “Cells from a man’s skin could theoretically be used to create eggs that are fertilized with a partner’s sperm, then nurtured in the womb of a surrogate” (1). This science-fiction scenario may be actually possible within the next twenty-five years. 3. ART in developing countries Epidemiological studies show that the majority of couples with no children live in developing countries (13). By contrast, high-quality ART treatments are almost exclusively performed in western countries (14). Developing countries still have quite a limited offer of treatments due to their high cost (15). Among all technologies available in developing countries, IVF constitutes the main treatment, followed by intrauterine insemination (IUI). However, these techniques are still unaffordable for the majority of the population in certain countries. Over the years, different strategies have arisen to provide more affordable solutions. The addition of natural cycles (16) or minimal/mild stimulations in order to reduce the amounts of hormones (17), in combination with intravaginal culture of oocytes (INVO) (18) or new developed devices offer reachable alternatives at a lower cost (19). High prices of treatments is not the only reason of the slow progress of ART in developing countries. Their fertility issues have not drawn western countries’ attention, and so interest in this research field has decreased. Overpopulation is, perhaps, one of the main reasons; countries lacking a well-developed social structure (healthcare or education, among others) and a strong economy able to sustain the present population should not foster population growth. On the contrary, it should be controlled; by doing so, life quality of next generations could be guaranteed. Additionally, developing areas have other priorities as thousands of people are still suffering from diseases such as malaria, tuberculosis or HIV. Interesting enough is the fact that the main cause for infertility is the high incidence of pelvic infections; therefore, approaches to reduce fertility issues in these countries should be firstly focused on decreasing sexually transmitted diseases (STDs) (20). CONCLUSIONS Within the last forty years assisted reproduction has experienced a true revolution. Looking back in time, seventy years ago all these approaches could have been regarded as science fiction, but nowadays infertility can be effectively treated. Whereas the main disadvantage is the high cost of assisted reproduction procedures, new lines of research are currently focused on reducing it, and new methodologies are becoming more easily available. Perhaps, in twenty-five years, ART as a whole maybe a worldwide affordable approach. REFERENCES 1. Cyranosky D. Stem cells: Egg engineers. Nature. 2013; 500:392-394. 2. Woodcock J. Assessing the clinical utility of diagnostics used in drug therapy. Clin Pharmacol Ther. 2010; 88:765–73. 3. Rocío Rivera Egea, Nicolás Garrido Puchalt, Marcos Messeguer Escrivá, Alex C Varghese. OMICS: Current and future perspectives in reproductive medicine and technology. Journal of Human Reproductive Sciences. 2014; 7(2): 73-92. 4. Palmer Stephen S, Barnhart Kurt T. Biomarkers in Reproductive Medicine: The promise, and can it be fulfilled? Fertil Steril. 2013; 99(4): 954-962. 5. Bromer JG, Seli E. Assessment of embryo viability in assisted reproductive technologies: shortcomings of current approaches and the emerging role of metabolomics. Curr Opin Obstet Gynecol. 2008; 20:234-241. 6. Lieberman B, Ali R, and Rangarajan S. Towards the elective replacement of a single embryo transfer (eSET) in the United Kingdom. Human Fertility. 2007; 10(2):123-7. 7. Mosher WD, Pratt WF. Fecundity and infertility in the United States: incidence and trends. Fertil Steril.1991; 56:192-3. 8. Murray KS, James A, McGeady JB, Reed ML, Kuang WW, et al. The effect of the new 2010 World Health Organization criteria for semen analysis on male infertility. Fertil Steril 2012; 98:1428-31. 9. Carrell DT, Liu L. Altered protamine 2 expression is uncommon in donors of known fertility, but common among men with poor fertilizing capacity, and may reflect other abnormalities of spermiogenesis. J Androl 2001; 22:604-10. 10. Aguilar-Gallardo C, Poo M, Gomez E, Galan A, Sanchez E, Marques-Mari A, et al. Derivation, characterization, differentiation, and registration of seven human embryonic stem cell lines (VAL-3, -4, -5, -6M, -7, -8, and -9) on human feeder. In Vitro Cell Dev Biol Anim 2010; 46:317-26. 11. Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult broblast cultures by de ned factors. Cell 2006; 126:663-76. 12. Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 2007; 318:1917-20. 13. Rutstein SO, Iqbal HS. Infecundity, Infertility and Childessness in Developing Countries. DHS Comparative Reports, WHO. 2004; p24. 14. Sharma S, Mittal S, Aggarwal P. Management of infertility in low resources countries. BJOG. 2009; 116 (Suppl1): 77-83. 15. Murage A, Muteshi MC, Githae F. Assisted reproduction services provision in a developing country: time to act? Fertil Steril. 2011; 96(4):966-8. 16. Shahin AY. The problem of IVF cost in developing countries: has natural cycle IVF a place? Reprod Biomed Online. 2007; 15(1):51-6 17. Kim CH, Kim SR, Cheon YP, et al. Minimal stimulation using gonadotropin-releasing hormone (GnRH) antagonist and recombinant human follicle-stimulating hormone versus GNRH antagonist multiple-dose protocol in low responders undergoing in vitro fertilization/intracytoplasmic sperm injection. Fertil Steril. 2009; 92(6):2082-4 18. Lucena E, Saa AM, Navarro DE, et al. INVO procedure: minimally invasive IVF as an alternative treatment option for infertile couples. Sci World J. 2012; 2012:571-596. 19. Klerkx EPF, Janssen M, van Blerkom J et al. O-014. First pregnancies with a simplified IVF procedure: a crucial step to universal and accessible infertility care Human Reprod. Abstracts of the 29th Annual Meeting of the European Society of Human Reproduction and Embryology 2013; 28(Suppl1):i4-i6. 20. Ombelet W. Global access to infertility care in developing countries: a case of human rights, equity and social justice. F, V& in ObGyn. 2011; 3(4):257-266.
2 Comments
|
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media