|
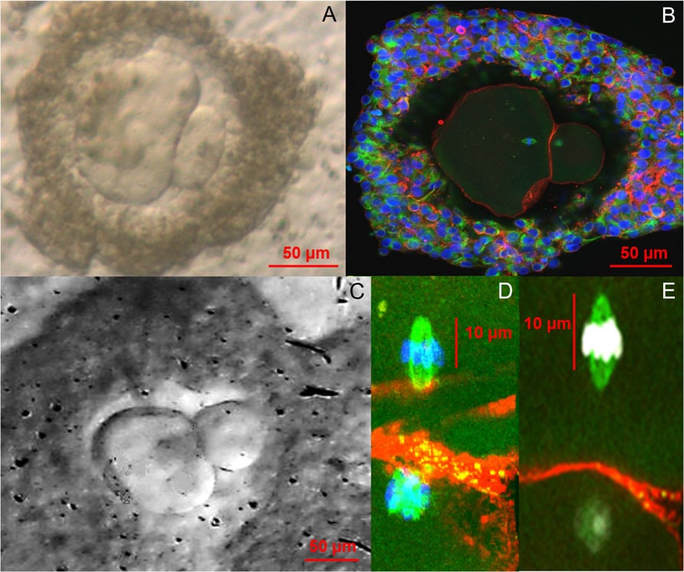
Author: Javier del Río “This could eventually help in developing new fertility preservation treatments”. Currently, there are many research lines focused on the improvement of assisted reproduction technologies (ART) and the development of new ones that guarantee new reproductive options for patients. Among the most recent investigated topics there are some worth to mention, like 3D-printed ovaries, stem cells-derived gametes or the use of CRISPR-Cas9 technology for embryos. All these promising approaches have opened new paths of research in ART, too. Recently, a paper has been published reporting functional human eggs grown in the laboratory for the first time. The study was carried out by McLaughlin and colleagues from University of Edinburgh (UK), whose research focuses on the mechanisms of follicle development. As part of the procedure, ovarian samples were collected from 10 women, aged between 25 and 39, who were undergoing elective caesarean section. Tissue pieces that showed no damage or abundant stromal tissue were dissected in the laboratory. A total of 87 fragments with follicles of a mean diameter of 40 μm were cultured for 8 days at 37°C in humidified air with 5% CO2 and renewing half the media every two days. Secondly, the growing follicles (presumably secondary follicles) were mechanically dissected and cultured individually for another 8 days in the same incubation conditions. In the following step, those follicles with visible antral cavities were isolated, from which 48 oocyte-granulosa cell complexes were removed. These were then placed on a membrane with growth-supporting proteins for the next 4 days. A total of 32 complexes containing oocytes >100 μm in diameter showed significant oocyte growth and were selected for in vitro maturation. After 24 hours, only 9 emitted polar bodies and presented cumulus cell expansion. In their paper, the authors point out the larger size of these polar bodies compared to those from in vivo grown oocytes, suggesting the possibility that oocytes may have lost a valuable part of cytoplasm. Finally, the team confirmed the proper organization of the polar bodies and the presence of a meiotic spindle in all 9 oocytes by confocal analysis, which supported the in vitro maturation process. Although complete oocyte growth and maturation had been previously achieved in mice, this is the first time researchers have developed human oocytes all the way from their earliest stages up to the point at which they would be released from the ovaries in vivo. The authors point out that these oocytes were of low quality, which explains their low maturity and the low probabilities of resuming the meiotic division. These results provide proof of concept in developing a complete in vitro growth system to support human oocyte maturation. However, there is a need to optimize each of the stages of development and to enhance our understanding of how culture systems affect the growth process, in particular the epigenetic status of any embryo derived from these eggs. Also, genetic analysis of the obtained oocytes is needed to confirm their healthy condition. Despite these issues, researchers believe that this could represent a new approach for women whose oocytes fail to fully develop in their body or who need to undergo any sort of harmful treatment such as chemotherapy or radiotherapy, which can damage oocytes and cause infertility. It would also be an alternative for girls suffering from cancer who have not yet hit puberty. Because these patients cannot produce mature oocytes, cryopreservation and reintroduction of ovarian tissue may represent a valid option, but they imply a potential risk of reinserting cancer cells, too. In addition, it may be useful for women who experiment premature fertility loss and for low responders, whose follicles do not respond to external stimulation. Potential applications of this technique would aim to women who opt for in vitro fertilization treatments and whose oocytes are too immature to be used. Furthermore, it will help to understand the mechanisms involved in the development of human oocytes. Definitely, there is a chance that the new technique could increase the total number of oocytes available for in vitro fertilization and a woman’s odds of conceiving. Much more work is needed though to make sure the technique is safe before it can be ascertained whether these oocytes remain normal during the process, and if they could actually lead to healthy live births once fertilized. REFERENCES
1. McLaughlin M, Albertini DF, Wallace WHB, Anderson R, Telfer E. Metaphase II oocytes from human unilaminar follicles grown in a multi-step culture system. Mol Hum Reprod. 2018;1-8.
2 Comments
2/26/2018 10:08:32
Yes this is an exiting development. But the clinical problem we face is premature ovarian failure and age related infertility. Developing oocytes from autologous stem cells would be much more useful.
Reply
Javier del Río
2/28/2018 19:35:20
Thank you for your comment M.D. Kini. More studies are needed to improve this new technique and to guarantee its effeciency. Also, stem cell therapy would be a good solution for premature ovarian failure, so that they can use their own gametes without having to resort to donated oocytes.
Reply
Leave a Reply. |
Entries
October 2018
Categories
All
Join our mailing list and keep yourself up to date!
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media