|
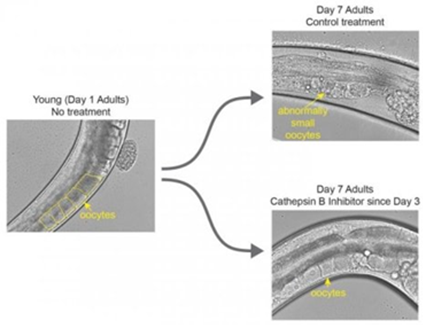
Author: Iris Martínez "In humans, this could translate into a 3- to 6-year extension of female fertility" One of the challenges of modern society is being able to synchronize our own socio-economic context with the biological clock in order to find the most appropriate moment to become parents. Reproductive medicine professionals have been warning about the importance of women awareness about the acute decrease of fertility right before their mid-30s. Should they be fully aware, it would be possible for them to better plan their motherhood by preserving oocytes at young age. However, there are news on this issue: research by Coleen Murphy’s team at Princeton University has reported a protein to be potentially able to extend eggs’ life. This is a key protein present in old and poor-quality oocytes from Caenorhabditis elegans, cathepsin B. In their work, recently published in Current Biology, the authors successfully achieved to extend egg viability longer than their regular life-span, as confirmed by two independent experiments. First, they knocked down cathepsin B-coding genes. The end result was an extension of fertility of the individuals by 10% in time. In humans, this could translate into a 3- to 6-year extension of female fertility. The second approach was to apply a cathepsin B inhibitor at a moment of the worm´s life comparable to the early 30s of a woman. WHY C. elegans? Murphy is professor of Molecular Biology and Director of Paul F. Glenn Laboratories for Aging Research at Princeton. She is specialised in C. elegans, a nematode commonly used as an animal model for a wide variety of studies, including longevity. These tiny animals have several advantages for researchers: not only they present a short life cycle of three weeks, but this species exhibits similar fertility decline towards mid-life, while their oocytes also experiment decrease in quality with age. In addition to this, many of their genes are conserved in humans and, more interestingly, some of them are involved in the regulation of the aging process. One of those genes, with a conserved role in regulation of longevity -and fairly studied by Murphy´s group- is the insulin-like growth factor-1 (onwards, IIS) and its pathway. Even though IIS´s transcriptional targets had been previously characterized, it was not known whether the same mechanisms had any influence on oocyte aging. CATHEPSIN B AND OOCYTE QUALITY EXTENSION When Murphy and colleagues investigated the issue of oocyte aging, they focused on genes and proteins present in higher proportion in healthy and young eggs than in aged ones. But research yielded better results when trying the opposite; this is, by looking at less common proteins in lower-quality oocytes. By doing so, they found a group of proteins absent from high-quality oocytes but abundant in those degraded by age: cathepsin B proteases. In particular, they found cathepsin B protease activity levels in oocytes to be dramatically increased around day 7 of adulthood, which corresponds to mid-life in worms. On the contrary, such levels were low in young adult high-quality oocytes. Cathepsin B knock down If cathepsin B proteases were responsible of oocyte aging and associated to deleterious activity, loss of cathepsin B proteases should lead to decreased oocyte quality damage with aging. Therefore, researchers knocked down individual cathepsin-B genes in adult worms by using iRNA. Surprisingly, those worms had their reproductive life-span increased by 10% compared to wild-type individuals. In addition, aged oocytes exhibited improved morphology. These results point to a deleterious role of cathepsin-B gene in reproductive aging regulation. Cathepsin B inhibitor Once cathepsin B proteases raised as oocyte-aging effectors, Murphy’s team explored pharmacological treatment of aging animals to find out whether the quality of aged oocytes had improved. By treating wild-type adult worms with a potent cell-permeable cathepsin B inhibitor (MDL-28170), the authors showed slowed down decline of oocyte quality over age. Evidences were reduced frequency of cavities between oocytes, misshapen oocytes and abnormally small oocytes in day-5 worms exposed to the inhibitor compared to the control (Figure 1). It is important to highlight that pharmacological inhibition of age-dependent damage in oocyte quality was achieved even when the treatment was applied around mid-life. It suggests that age-related reproductive decline could be pharmacologically treated through mid-life intervention. Figure 1. Image on the left shows a worm at the beginning of its reproductive window with healthy unfertilised eggs, at day 1 of adulthood. The upper-right image represents control cases, in which worms did not receive the treatment by day 7 of their adulthood. Here, eggs are abnormally small and deformed. The lower-right image displays a worm treated with the cathepsin B inhibitor; these individuals show healthy eggs on day 7 (1). WIDER IMPLICATIONS
In an interview to Princeton University press, Murphy showed excited about the doors opened by this study. What she likes the most is the possibility of worms to be useful in mammalian research, even though most people think “worms are so different from mammals”. Dr. Murphy and colleagues have set new horizons on interventions to slow down the loss of reproductive aging, although cathepsin B inhibitor is nowhere near ready for testing in humans. However, in Dr. Murphy’s words: “ (...) if you could do something mid-reproduction to improve the rest of reproduction- for me, that’s a game changer”. REFERENCE 1. Templeman N, Luo S, Kaletsky R, Shi C, Ashraf J, Keyes W et al. Insulin Signaling Regulates Oocyte Quality Maintenance with Age via Cathepsin B Activity. Current Biology. 2018;28(5):753-760.e4.
0 Comments
Leave a Reply. |
Entries
October 2018
Categories
All
Join our mailing list and keep yourself up to date!
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media