|
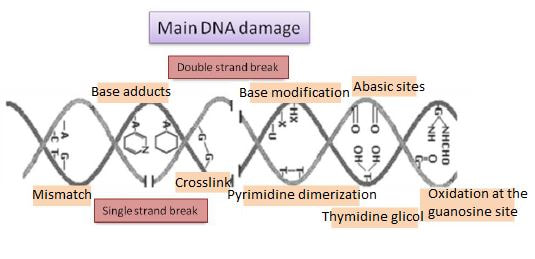
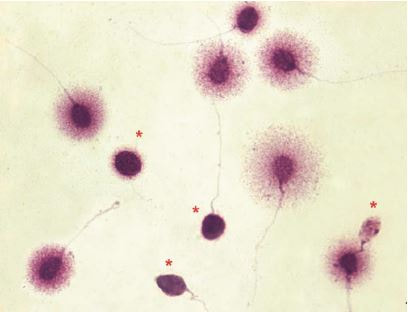
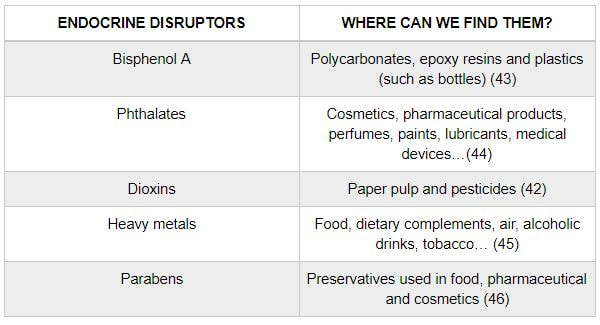
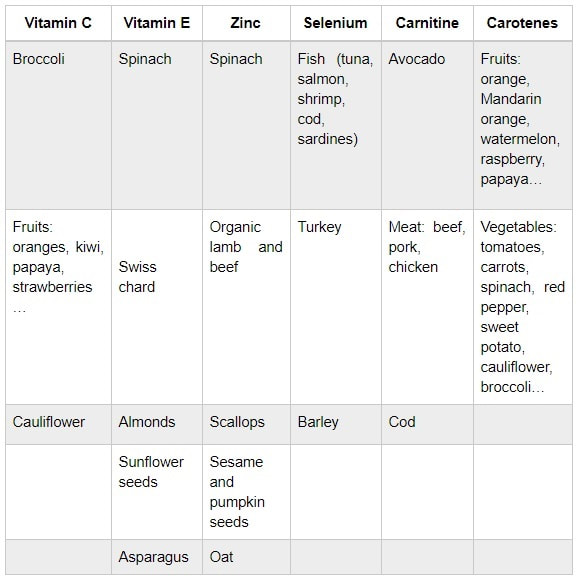
Authors: María Caballero Sastre & Lidón Carretero Vilarroig “Due to the current lifestyle and habits, high production of ROS and oxidative stress are the most common mechanisms responsible for DNA damage” 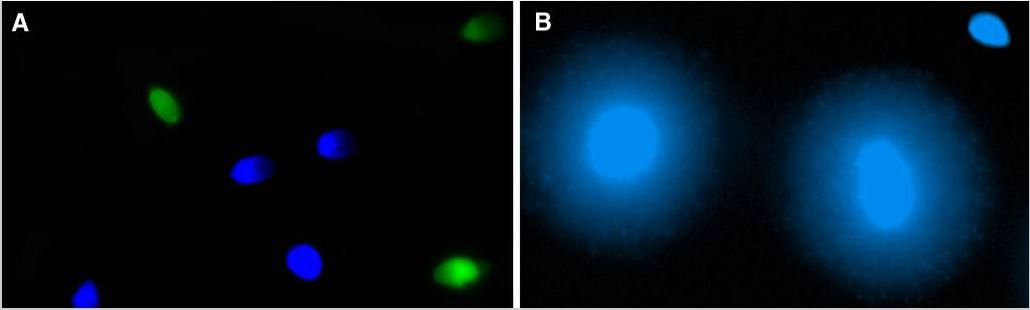 Fig 1. Example of two techniques to detect DNA fragmentation in a sperm sample. A) TUNEL assay: sperm in green indicate DNA fragmentation. B) Sperm Chromatin Dispersion test: the halo around both central sperm nuclei indicate non-fragmented DNA, while the absence of halo in the sperm in the upper right hand corner indicates DNA fragmentation (1). INTRODUCTION Male factor plays a role in 33% of couples experiencing infertility problems. Although it is less understood than its female counterpart, a great effort has been made in order to understand its basis. The World Health Organization (WHO) periodically updates its Laboratory Manual for the Examination and Processing of Human Semen, as well as the reference parameters (2). However, since results from conventional seminograms vary depending on temporal factors such as stress, period of abstinence or sample collection, it is difficult to diagnose male infertility using conventional semen analyses (3). This makes also necessary to understand the process behind male gamete formation (spermatogenesis) in order to achieve a better comprehension of the molecular basis of male infertility. Within spermatogenesis, meiotic problems may occur resulting in sperm cells carrying chromosome alterations, and during spermiogenesis sperm DNA becomes highly compacted by replacing histones by protamines. Humans express two protamines (P1 and P2) in a 1:1 ratio; an imbalance of this ratio results in male infertility (2). Measurement of sperm DNA integrity as a marker of sperm quality has recently gained interest for embryologists. The study of sperm DNA fragmentation is highly indicated in cases of implantation failure or repeated miscarriages. There is a wide range of tests currently used aimed to analyze sperm DNA integrity (Fig. 1). These techniques make it possible to determine whether there is a high rate of single-strand or double-strand breakpoints (SSB or DSB) in the DNA; in such cases the fertilizing and replicative ability of the sperm cell may be jeopardised. CONVENTIONAL SEMEN ANALYSIS Conventional semen analyses are the first step to measure semen parameters. Using fresh ejaculated semen, andrologists evaluate sperm motility, vitality and morphology, as well as its concentration along with other seminal components, and total volume of the ejaculate. WHO gives the following results as a reference for normality (2): - Volume of at least 1.5 mL. The total amount of fresh ejaculated semen may vary depending on the abstinence period. Thus, a period of abstinence of 3-7 days before the analysis is recommended. - Concentration of at least 15 million sperm cells per millilitre (15 mil/mL). Samples with less than 15 mil/mL are classified as oligozoospermic, whereas samples with no sperm cells are classified as azoospermic. Samples with a concentration below 100,000 sperm cells per millilitre are considered cryptozoospermic. - Motility and vitality. Even though sperm cells acquire their characteristic forward and rapid movement during capacitation, the way they move within a fresh sample is a quality indicator. Proper movement consists in progressive and linear displacement, while those sperm cells with circular or even missing movement are considered less capable of fertilising. Such samples are classified as asthenozoospermic. Following these WHO’s reference guidelines, sperm samples are considered normal if linear progressive movement is found in at least 32% of the cells. - Correct morphology in at least 4% of the sperm cells. This feature refers to (4): (i) sperm cells with one head and one tail; (ii) sperm cells with a round/oval-shaped head; (iii) lack of cytoplasmic droplet. Samples with less than 4% of morphologically normal sperm cells are classified as teratozoospermic. Fluorescent In Situ Hybridization (FISH) in spermatozoa is a complementary technique to conventional semen analyses. This test is indicated in cases of altered seminal values, miscarriages, implantation failure, non-oocyte-driven fertilization failure cause or anomalous embryonic development (5). The objective of this procedure is to verify the chromosome number in human sperm cells. As haploid cells, these must contain 23 chromosomes each. FISH assays use fluorescent probes to recognize specific regions of certain chromosomes in every test. The 5-probe FISH test analyzes chromosomes X, Y, 13, 18 and 21, while the 9-probe FISH test also includes chromosomes 15, 16, 17 and 22. Altered results can suppose a high percentage of diploid, dysomic or hyperploidic sperm cells, which may compromise fertilization and embryo development (Fig. 2). In such cases, a cycle of in vitro fertilization is indicated, followed by preimplantation genetic test (IVF-PGT) by aneuploidy screening, in order to transfer only euploid embryos. Due to all different variables that may influence the result of a conventional semen analysis (6-8) and FISH assay (7), DNA fragmentation analysis has gained attention over the last years. 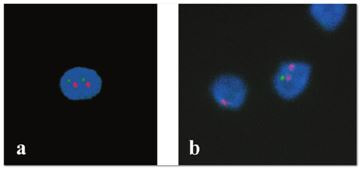 Fig 2. FISH analysis of testicular human sperm. Sperm nuclei hybridized by chromosome 18 (green) and Y (red) probes. (a) Diploid sperm nucleus exhibiting signals for chromosomes 18,18 and YY.. (b) Nucleus with an absent signal for chromosome 18 (left) and nucleus displaying gonosomal dysomy for the Y chromosome (5). SPERM DNA FRAGMENTATION According to Sakkas et al., DNA damage in spermatozoa can be induced by six main mechanisms: [1] apoptosis during spermatogenesis, [2] DNA strand breaks produced during remodeling of sperm chromatin typical of spermiogenesis, [3] oxygen radicals during sperm transport through the seminiferous tubules and the epididymis, [4] endogenous caspases and endonucleases, [5] radiotherapy and chemotherapy and [6] environmental toxicants (8). Some of these mechanisms seem to be involved in specific cases of DNA damage due to a variety of factors such as varicocele (10), bacterial infections (11), age (15) or abstinence (13). The main types of DNA damage include are mismatch, base adducts, crosslink, base modification, pyrimidine dimerization, abasic sites, thymidine glycol and oxidation at guanosine sites. When evaluating DNA fragmentation, two kind of breaks are considered: single strand breaks (SSBs) and double strand breaks (DSBs) (Fig. 3). Although both type of DNA lesions are present in sperm of both fertile and infertile men, the latter have been observed to present higher levels of DNA fragmentation (14). Each kind of DNA break has a different origin. For instance, nucleases usually generate both types, whereas chromatin remodelling may cause DSBs produced by topoisomerase II and reactive oxygen species (ROS), and other radical molecules generate mainly SSBs, DNA adducts and abasic sites (16, 17). Whereas SSBs are slightly detrimental and easy to repair, DSBs represent serious harmful lesions that can lead to chromosome aberrations (16, 18). Fortunately, cells present different pathways to face and repair DNA damage. The main mechanisms present in germline cells are direct reversal repair, nucleotide excision repair, base excision repair, mismatch repair, post replication repair and DNA double strand break repair (19). These DNA repair processes can take place during spermiogenesis (20), in the fertilized oocyte (21) or during early embryonic development (22). The capacity to repair DNA damage depends on the type of DNA damage present in the sperm; should it fail, the embryo may not develop or miscarriage may eventually occur (14). DIFFERENT TECHNIQUES TO ASSES DNA DAMAGE - Sperm Chromatin Structure Assay (SCSA): This technique, first proposed in 1980 by Evenson and coauthors (23), uses flow cytometry to measure sperm DNA integrity. It tests the vulnerability of DNA to acid-induced denaturation by exposure to acridine orange media (AO) (24). AO binds to both double-stranded and single-stranded DNA, but fluoresces in different colour, being red or green, respectively. The DNA fragmentation index (DFI), which represents the percentage of spermatozoa with red fluorescence, is analyzed and represented on a histogram. Green fluorescence in turn shows the percentage of mature undamaged sperm (24). - TUNEL test: This test detects SSBs and DSBs by labelling 3’-free ends of DNA fragments (25) with fluorescent dUTP nucleotides in a reaction catalyzed by the terminal deoxynucleotidyl transferase. The evaluation process can be performed using light and fluorescence microscope or flow cytometry (26, 27). - Single cell gel electrophoresis assay (COMET): It enables the detection of both SSB and DSBs. It consists in the lysis of sperm cells with detergents and high salt-density components. After lysis, the sample is introduced into a gel and subjected to an electrophoretic field resulting in the migration of both DNA and proteins. DNA fragments move faster than intact DNA; as a consequence, fragmented DNA from sperm cells migrate through the gel leaving a comet-like trace, whereas DNA without breaks is seen as a dot. Samples can be analyzed by fluorescence microscopy. The ratio between comet-like trace and dots defines the degree of fragmentation of the sample. - Sperm cromatin dispersion (SCD) test: SCD test allows for the differentiation between sperm with fragmented DNA and intact DNA. This technique denatures DNA that contains breaks using an acid solution. Subsequently, spermatozoa are treated with a lysis buffer to remove membranes and proteins and, in so doing, a peripheral halo of DNA loops is formed around the sperm. The observation of a large halo means the sperm nucleus presents low levels of DNA fragmentation. On the contrary, a small or absent halo is indicative of highly fragmented DNA (Fig. 5). Results can be evaluated using either fluorescence or bright-field microscopy (29). - γH2AX test: Not only is γH2AX labelling assay used to assess DNA fragmentation in sperm cells, but it has been also used in order to evaluate DNA damage in tumour cells during radio- or chemotherapy, thus providing a potential prediction marker (30). Once DSBs occur, histone H2AX becomes phosphorylated at Ser139 (γH2AX), playing a role in signalling the break sites to be repaired (31). The presence of γH2AX in this context is commonly assessed by flow cytometry, being the percentage of γH2AX higher among infertile men. LIFESTYLE AND POTENTIAL HARMFUL FACTORS FOR DNA Among the six mechanisms above mentioned that can produce DNA damage, oxidative stress and ROS are the most common (17), which is mainly due to current lifestyle and habits. The term ROS includes highly oxidative radicals like OH, non-radical species like O2- and H2O2, as well as reactive nitrogen species. Spermatozoa produce ROS themselves, since these are required for several functions such as sperm motility, capacitation, acrosomal reaction, zona pellucida binding (32) and chromatin compaction (17). Because ROS concentration can be harmful, it is important to guarantee the action of antioxidant mechanisms in order to maintain cell homeostasis (33). Therefore, decompensation in ROS final concentration, due to either excess production or to failure in antioxidant mechanisms, would eventually lead to oxidative stress and, as a consequence, to DNA damage (34-36). Alcohol and smoking Several studies have demonstrated both alcohol and tobacco to be responsible for the significant increase in ROS at sperm production (37,38). Although some studies have shown reduced efficiency of antioxidant mechanisms in smokers and people with a high intake of alcohol (39), contradictory results have been reported as well (40). These studies showed an unbalance in favour of ROS despite the increased antioxidant activity, suggesting the antioxidant mechanisms were somehow unable to protect from oxidative damage (40). The various chemical products found in tobacco such as cadmium, lead (17) and nicotine, and alcohol itself most likely seem to result harmful for semen parameters, since they have been reported to cause DNA damage (40). Obesity A recent study by Jin-Chun Lu and collaborators has shown data from a correlation between sperm DNA fragmentation index and obesity (41). Samples from 1010 subfertile men from 18 to 50 years old were analysed using SCSA. Although the authors found no correlation between obesity and sperm index fragmentation, they did find a positive correlation between sperm fragmentation and seminal plasma triglyceride and total cholesterol levels.A separate study on these matters concluded there is no correlation between body mass index and DNA integrity. The authors stated obesity might affect lipid metabolism and reproductive hormones, leading to a decreased sperm quality (42). Varicocele Varicocele is an enlargement of the veins located within the scrotum. It is postulated that varicocele causes a rise in temperature that leads to higher DNA fragmentation ratio and ROS production (43). About 15% of adult men suffer from this condition (44), but authors suggest that varicocelectomy can improve semen parameters after 3 months (8), which is the expected period for de novo sperm production in humans. Environmental contaminants Exposure to environmental contaminants has been demonstrated to be the main cause for male infertility, even over genetic defects (45). These substances, known as endocrine disruptors (ED) (Table 1), can negatively affect the endocrine system by mimicking natural hormones in the organism like estrogens and androgens, and so they can block interactions between natural hormones and their receptors (46). Exposure of an individual to ED may cause important specific epigenetic modifications in sperm (that are eventually passed down to several generations), as well as severe fertility disorders (45). Electromagnetic radiation: mobile phones radiation and Wi-Fi It has been suggested that the electromagnetic radiation (EMR) emitted by some electrical devices such as mobile phones or Wi-Fi suppresses the natural antioxidant mechanisms and increase the oxidative stress, which consequently increases infertility (50). Although studies in both animals and humans have demonstrated the detrimental effect of EMR, there is still controversy. Some studies show that the prolonged use of mobile phones decreases sperm motility (51), concentration (52), viability and morphology (53), while others have found no effects on sperm concentration (reviewed in 54). Moreover, results have been reported on the increase in DNA fragmentation in human sperm associated to exposure to Wi-Fi networks (51). Heat It is important to maintain testicular temperature 2-4ºC lower than the body temperature for spermatogenesis to progress normally. This justifies the existence of several thermoregulatory mechanisms found in the organism, such as the characteristics of the scrotal sac, the musculature and the vasculature (18). Fertility is directly affected by a rise in temperature due to the increased DNA fragmentation index (55) and sperm cell apoptosis (18). Several factors can cause hyperthermia, namely: the use of tight clothes, saunas, laptops and mobile devices closely located to the testes (18), or even an episode of high fever or varicocele (55). Moreover, the severity of damage varies with duration, frequency and intensity of heat (18). ANTIOXIDANT THERAPY Spermatozoa present low levels of antioxidants and DNA repair enzymes. Therefore, the presence of these factors in the seminal plasma is critical. As it has been demonstrated, not only subfertile men have been observed to present lower levels of antioxidants than fertile men (35, 56), but they have been also found to exhibit higher levels of ROS (36). Antioxidants present in semen include vitamins C and E, zinc, selenium, carnitine and carotenoids (57). These components can be found in the food (table 2), meaning that controlling the dietary antioxidant intake, oxidative stress can be minimised or avoided and, as a consequence, DNA damage, too (17). Besides the above-mentioned antioxidants, polyunsaturated fatty acids are also an important source of antioxidants. They are classified into omega-3, omega-6 and omega-9 fatty acids and, with the exception of omega-9, they need to be incorporated into the diet through the intake of vegetable and fish oils. CONCLUSIONS The first step to check male infertility in a couple undergoing infertility issues is the completion of a conventional semen analysis established by the WHO. This kind of test evaluates several parameters such as motility, vitality, morphology, concentration and total volume of the ejaculate. Because of the evidenced involvement of sperm DNA damage in infertility, its study has been gaining significance and, consequently, it is usually indicated in certain cases such as implantation failure or recurrent miscarriages. Techniques like SCSA, SCD, TUNEL, COMET and γH2AX test are normally used to analyze sperm DNA integrity. These approaches are helpful to discriminate between SSBs and DSBs or other types of DNA damage. Due to the current lifestyle and habits, high production of ROS and oxidative stress are the most common mechanisms responsible for DNA damage. Among such habits and lifestyle it is worth mentioning alcohol and smoking, environmental contaminants, electromagnetic radiations and heat sources. An efficient method to prevent DNA fragmentation is to resort to the intake of specific products as an antioxidant dietary therapy. Thus, a proper diet rich in such components may be able to reduce the incidence of sperm DNA fragmentation, and, consequently, to also improve male fertility. REFERENCES
1. Schulte RT, Ohl DA, Sigman M, Smith GD. Sperm DNA damage in male infertility: Etiologies, assays, and outcomes. J Assist Reprod Genet. 2010;27(1):3–12. 2. Edition F. Examination and processing of human semen. World Health. 2010;Edition, V(10):286. 3. Saleh RA, Agarwal A, Nelson DR, Nada EA, El-Tonsy MH, Alvarez JG, et al. Increased sperm nuclear DNA damage in normozoospermic infertile men: A prospective study. Fertil Steril. 2002;78(2):313–8. 4. Menkveld R. Clinical significance of the low normal sperm morphology value as proposed in the fifth edition of the WHO Laboratory Manual for the Examination and Processing of Human Semen. Asian Journal of Andrology. 2010;12(1):47-58. 5. J. Bataller Sánchez, A. Barberá Alberola, X. Vendrell Montón, M. Ruiz Jorro CCL. Valoración del área media de la cabeza espermática como posible indicador para solicitar FISH en espermatozoides. ASEBIR 2017. 6. Dunphy BC, Neal LM, Cooke ID. The clinical value of conventional semen analysis. Fertil Steril. Elsevier Masson SAS; 1989;51(2):324–9. 7. Intasqui P, Camargo M, Antoniassi M, Barradas V, Spaine DM, Bertolla RP. Sperm functional testing and conventional semen analysis: correlations, distributions, and putative mechanisms. Fertil Steril. Elsevier Ltd; 2013;100(3):S214. 8. Kruger T. Critical appraisal of conventional semen analysis in the context of varicocele. Asian J Androl. 2016;18(2):202. 9. Sakkas D, Alvarez JG. Sperm DNA fragmentation: mechanisms of origin, impact on reproductive outcome, and analysis. Fertil Steril. 2010;93(4):1027–36. 10. Roque M, Esteves SC. Effect of varicocele repair on sperm DNA fragmentation: a review. Int Urol Nephrol. Springer Netherlands; 2018;50(4):583–603. 11. González-Marín C, Roy R, López-Fernández C, Diez B, Carabaño MJ, Fernández JL, et al. Bacteria in bovine semen can increase sperm DNA fragmentation rates: A kinetic experimental approach. Anim Reprod Sci. 2011;123(3–4):139–48. 12. Vagnini L, Baruffi RLR, Mauri AL, Petersen CG, Massaro FC, Pontes A, et al. The effects of male age on sperm DNA damage in an infertile population. Reprod Biomed Online. 2007;15(5):514–9. 13. Comar VA, Petersen CG, Mauri AL, Mattila M, Vagnini LD, Renzi A, et al. Influence of the abstinence period on human sperm quality: Analysis of 2,458 semen samples. J Bras Reprod Assist. 2017;21(4):306–12. 14. González-Marín C, Gosálvez J, Roy R. Types, causes, detection and repair of DNA fragmentation in animal and human sperm cells. Int J Mol Sci. 2012;13(11):14026–52. 15. Vagnini L, Baruffi RLR, Mauri AL, Petersen CG, Massaro FC, Pontes A, et al. The effects of male age on sperm DNA damage in an infertile population. Reprod Biomed Online. 2007;15(5):514–9. 16. Cortés-Gutiérrez EI, López-Fernández C, Fernández JL, Dávila-Rodríguez MI, Johnston SD, Gosálvez J. Interpreting sperm DNA damage in a diverse range of mammalian sperm by means of the two-tailed comet assay. Front Genet. 2014;5:1–11. 17. Wright C, Milne S, Leeson H. Sperm DNA damage caused by oxidative stress: Modifiable clinical, lifestyle and nutritional factors in male infertility. Reprod Biomed Online. Reproductive Healthcare Ltd.; 2014;28(6):684–703. 18. Durairajanayagam D, Agarwal A, Ong C. Causes, effects and molecular mechanisms of testicular heat stress. Reprod Biomed Online. Reproductive Healthcare Ltd.; 2015;30(1):14–27. 19. Jaroudi S, Kakourou G, Cawood S, Doshi A, Ranieri DM, Serhal P, et al. Expression profiling of DNA repair genes in human oocytes and blastocysts using microarrays. Hum Reprod. 2009;24(10):2649–55. 20. Baarends WM, van der Laan R, Grootegoed JA. DNA repair mechanisms and gametogenesis. Reproduction. 2001;121(1):31–9. 21. Men NT, Kikuchi K, Furusawa T, Dang-Nguyen TQ, Nakai M, Fukuda A, et al. Expression of DNA repair genes in porcine oocytes before and after fertilization by ICSI using freeze-dried sperm. Anim Sci J. 2016;87(11):1325–33. 22. Derijck A, Van der heijden G, Giele M, Philippens M, De boer P. DNA double-strand break repair in parental chromatin of mouse zygotes, the first cell cycle as an origin of de novo mutation. Hum Mol Genet. 2008;17(13):1922–37. 23. Evenson DP, Darzynkiewicz Z MM. Relation of mammalian sperm chromatin heterogeneity to fertility. Science. 1980;210(4474):1131–3. 24. Khatun A, Rahman MS, Pang M-G. Clinical assessment of the male fertility. Obstet Gynecol Sci. 2018;61(2):179–91. 25. Ribas-Maynou J, García-Peiró A, Fernández-Encinas A, Abad C, Amengual MJ, Prada E, et al. Comprehensive analysis of sperm DNA fragmentation by five different assays: TUNEL assay, SCSA, SCD test and alkaline and neutral Comet assay. Andrology. 2013;1(5):715–22. 26. Rex AS, Aagaard J, Fedder J. DNA fragmentation in spermatozoa: a historical review. Andrology. 2017;5(4):622–30. 27. Kucuk N. Sperm DNA and detection of DNA fragmentations in sperm. Türk Üroloji Dergisi/Turkish J Urol [Internet]. 2018;44(1):1–5. 28. Pandir D. Assessment of the DNA Damage in Human Sperm and Lymphocytes Exposed to the Carcinogen Food Contaminant Furan with Comet Assay. 2015;58:773–80. 29. Fernández JL, Muriel L, Goyanes V, Segrelles E, Gosálvez J, Enciso M, et al. Simple determination of human sperm DNA fragmentation with an improved sperm chromatin dispersion test. Fertil Steril. 2005;84(4):833–42. 30. Xuan Huang H. Dorota Halicka Frank Traganos Toshiki Tanaka Akira Kurose Zbigniew Darzynkiewicz. Cytometric assessment of DNA damage in relation to cell cycle phase and apoptosis. Cell Prolif. 2005;8(4):223-43. 31. Jha KN, Tripurani SK, Johnson GR. TSSK6 is required for γH2AX formation and the histone-to-protamine transition during spermiogenesis. J Cell Sci. 2017;130:1835–44. 32. Doshi S, Khullar K, Sharma R, Agarwal A. Role of reactive nitrogen species in male infertility. Reprod Biol Endocrinol. 2012;10:109. 33. Sharma RK1 AA. Role of reactive oxygen species in male infertility. Urology. 1996;48(6):835–50. 34. Muratori M, Tamburrino L, Marchiani S, Cambi M, Olivito B, Azzari C, et al. Investigation on the Origin of Sperm DNA Fragmentation: Role of Apoptosis, Immaturity and Oxidative Stress. Mol Med. 2015;21(1):109–22. 35. R2 BSD. Oxidative stress: Major executioner in disease pathology, role in sperm DNA damage and preventive strategies. Front Biosci. 2017;9:420–47. 36. Aktan G, Doǧru-Abbasoǧlu S, Küçükgergin C, Kadioǧlu A, Özdemirler-Erata G, Koçak-Toker N. Mystery of idiopathic male infertility: Is oxidative stress an actual risk? Fertil Steril. 2013;99(5):1211–5. 37. Tweed JO, Hsia SH, Lutfy K, Friedman TC. The endocrine effects of nicotine and cigarette smoke. Trends Endocrinol Metab. Elsevier Ltd; 2012;23(7):334–42. 38. Anderson RA Jr, Willis BR, Oswald C, Reddy JM, Beyler SA ZL. Hormonal imbalance and alterations in testicular morphology induced by chronic ingestion of ethanol. Biochem Pharmacol. 1980;29(10):1409–19. 39. Lesgards JF, Durand P, Lassarre M, Stocker P, Lesgards G, Lanteaume A, et al. Assessment of lifestyle effects on the overall antioxidant capacity of healthy subjects. Environ Health Perspect. 2002;110(5):479–86. 40. Aboulmaouahib S, Madkour A, Kaarouch I, Sefrioui O, Saadani B, Copin H, et al. Impact of alcohol and cigarette smoking consumption in male fertility potential: Looks at lipid peroxidation, enzymatic antioxidant activities and sperm DNA damage. Andrologia. 2018;50(3):1–7. 41. Lu JC, Jing J, Chen L, Ge YF, Feng RX, Liang YJ, et al. Analysis of human sperm DNA fragmentation index (DFI) related factors: A report of 1010 subfertile men in China. Reprod Biol Endocrinol. Reproductive Biology and Endocrinology; 2018;16(1):1–9. 42. Bandel I, Bungum M, Richtoff J, Malm J, Axelsson J, Pedersen HS, et al. No association between body mass index and sperm DNA integrity. Hum Reprod. 2015;30(7):1704–13. 43. Silber S. The varicocele argument resurfaces. J Assist Reprod Genet. Journal of Assisted Reproduction and Genetics; 2018 44. Lundy SD, Sabanegh ES. Varicocele management for infertility and pain: A systematic review. Arab J Urol. 2017;16(1): 157-170 45. Jenardhanan P, Panneerselvam M, Mathur PP. Effect of environmental contaminants on spermatogenesis. Semin Cell Dev Biol. Elsevier Ltd; 2016;59:126–40. 46. Claude Monneret. What is an endocrine disruptor? C R Biol. Academie des sciences; 2017;340:403–5. 47. Fromme H, Bolte G, Koch HM, Angerer J, Boehmer S, Drexler H, et al. Occurrence and daily variation of phthalate metabolites in the urine of an adult population. Int J Hyg Environ Health. 2007;210(1):21–33. 48. Wirth JJ, Mijal RS. Adverse effects of low level heavy metal exposure on male reproductive function. Syst Biol Reprod Med. 2010;56(2):147–67. 49. Castelain F, Castelain M. Parabens: A real hazard or a scare story? Eur J Dermatology. 2012;22(6):723–727. 50. La Vignera S, Condorelli RA, Vicari E, D’Agata R, Calogero AE. Effects of the exposure to mobile phones on male reproduction: A review of the literature. J Androl. 2012;33(3):350–6. 51. Choy JT, Brannigan RE. Re: Use of laptop computers connected to internet through Wi-Fi decreases human sperm motility and increases sperm DNA fragmentation. Eur Urol. 2012;62(6):1196–7. 52. Kesari KK, Behari J. Microwave exposure affecting reproductive system in male rats. Appl Biochem Biotechnol. 2010;162(2):416–28. 53. Agarwal A, Deepinder F, Sharma RK, Ranga G, Li J. Effect of cell phone usage on semen analysis in men attending infertility clinic: an observational study. Fertil Steril. 2008;89(1):124–8. 54. Adams JA, Galloway TS, Mondal D, Esteves SC, Mathews F. Effect of mobile telephones on sperm quality: A systematic review and meta-analysis. Environ Int. Elsevier B.V.; 2014;70:106–12. 55. Ahmad G, Moinard N, Esquerr-Lamare C, Mieusset R, Bujan L. Mild induced testicular and epididymal hyperthermia alters sperm chromatin integrity in men. Fertil Steril. 2012;97(3):546–53. 56. Tremellen K. Oxidative stress and male infertility — a clinical perspective. 2008;14(3):243–58. 57. Talevi R, Barbato V, Fiorentino I, Braun S, Longobardi S, Gualtieri R. Protective effects of in vitro treatment with zinc, d-aspartate and coenzyme q10 on human sperm motility, lipid peroxidation and DNA fragmentation. Reprod Biol Endocrinol. 2013;11(1):1–10.
6 Comments
|
Entries
March 2019
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media