|
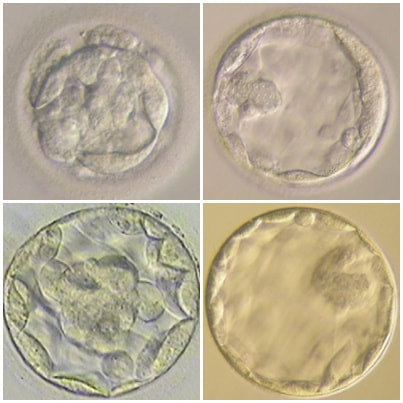
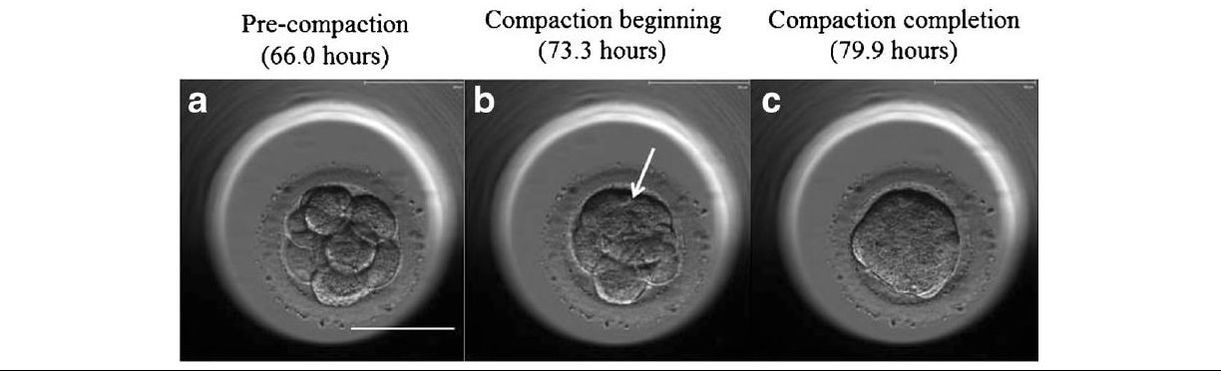
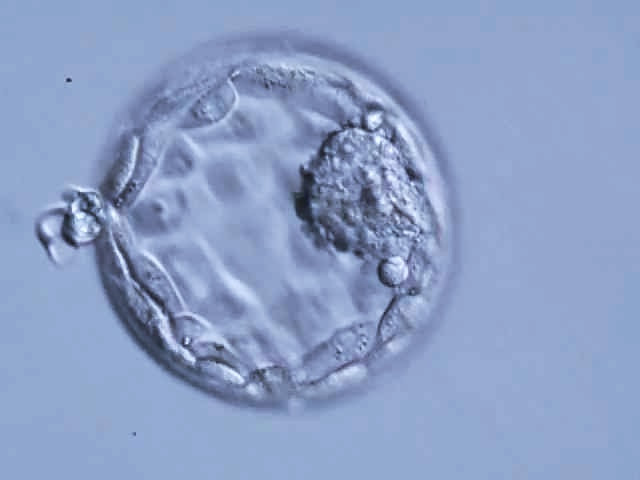
Authors: Javier del Río, Belén Gómez-Giménez & Iris Martínez "The relationship between blastocyst morphology and implantation has been investigated mainly according to expansion, trophectoderm and inner cell mass" The main goal of in vitro fertilization (IVF) is the birth of a single healthy child. However, the consequences and the effects of assisted reproductive techniques on children’s short- and long-term health have always been a source of discussion. Although IVF techniques and protocols have dramatically improved, the overall success rates are still relatively low, and assisted reproduction units still face the challenge of improving pregnancy rates (1). For this purpose, transfer of a single human embryo at blastocyst stage is becoming more common in the practice of assisted reproduction (2). It allows a better synchronization between the endometrium and the embryo and the possible selection of embryos with a higher implantation potential (3). Several morphology- or kinetics-based approaches have been described to select the best blastocyst in order to increase pregnancy rates. However, the yielded results are conflicting and the outcome is a matter of never-ending and controversial debates, specially regarding blastocyst stage (4, 5). BLASTOCYST ASSESSMENT SYSTEMS The relationship between blastocyst morphology and subsequent blastocyst implantation has been investigated according to various criteria. Traditionally, morphology has been evaluated after embryo compaction (6). The significance of examining the embryo after compaction is the ability to examine it after embryonic genome activation. Furthermore, the obvious benefit of looking at the blastocyst is the possibility to examine both cell types. The extent to which the trophectoderm (TE) develops will reflect the embryo’s ability to attach and implant in the endometrium, whereas development of the inner-cell mass (ICM) is obviously crucial for the progress of the foetus (7). There have been described several assessment systems to predict the success of blastocyst implantation. However, Gardner's grading system seems to be a better predictor of pregnancy rates (8, 4, 7). Following this method, blastocysts are initially scored from 1 to 6 based on their degree of expansion and hatching status, and ICM and TE grading is then assessed from A to D depending on their morphology (9). It was felt that expansion was important for cavity formation. This process requires both extensive energy utilization through the Na+/K+ ATPases on the basolateral membrane of the TE and formation of effective tight junctions between TE cells to form a barrier. Therefore, expansion seems to be a reflection of embryo competence (7). Recently, Richardson et al. proposed a simplified blastocyst grading system. These authors demonstrated both its prognostic potential and the inter- and intra-observer variability. This grading scheme was able to effectively predict clinical outcomes in terms of implantation, clinical pregnancy and live birth. Slight variation existed both between and within embryologists grading the embryos but, overall, consistency in their analyses was similar to, if not better than, those associated with more complex grading systems (10). However, most of the grading systems that are currently used for assessing viability of IVF embryos are subjective, relying on visual inspection of morphological characteristics of the embryos that are qualitatively evaluated. Grading based on qualitative criteria is imprecise, and it inevitably results in inter-observer variability and in intra-observer to some extent, as well (10). MORPHOLOGICAL ASSESSMENT AND OUTCOME RESULTS As it has been exposed, there is a need for increased knowledge about the relative impact of each morphology parameter at the blastocyst stage (and their potential correlation) on predicting the probability of successful implantation and pregnancy (1, 2, 11). Shapiro et al. compared up to 25 parameters in order to develop predictive models of clinical pregnancy within a set of blastocyst transfer cycles (12). Among these variables, blastocyst diameter seemed to be the most significant predictor of clinical pregnancy in the multivariate models. The authors concluded that embryos developing into expanded blastocyst stage on day 5 were approximately twice as likely to implant, compared to those for which expansion was delayed until day 6 (13, 3). This is supported by Van den Abbeel and coauthors, who found that high scores of blastocyst expansion and hatching stage, ICM and TE grade were all significantly associated with increased pregnancy and live birth rates after fresh transfers (11). The finding that the expansion and hatching stage is the most important parameter when selecting a blastocyst for transfer (11) is in contrast with some retrospective cohort studies that suggest TE grading to have the strongest predictive power for treatment outcome in fresh transfers (14, 15). On the contrary, Basak Balaban et al. exposed that quantitative measurement of blastocysts and ICM is not a practical way to assess blastocyst quality, arguing that two-dimensional measurements of three-dimensional global structures can be misleading. The reasoning is that the size of a blastocyst may vary depending on the time the blastocyst is assessed under the microscope, and this may easily confuse grading (9). For this purpose, Almagor et al. tried to provide an easily measurable assessment of the ICM and evaluate its correlation with pregnancy rates in a series of single blastocyst transfers. They found a high ICM/blastocyst ratio associated with significantly increased pregnancy rates. Thus, they proposed this measure to be used as an additional strongly predictive parameter of successful implantation (16). Recently, Bouillon et al. have confirmed that clinical pregnancy and live birth rates were significantly higher for blastocysts with good TE and ICM quality, and so it was concluded that both rates decreased with morphology (4). Even though some blastocysts with non-optimal morphology are able to implant, it has been suggested that when selection is made among suboptimal blastocysts, preference should be given to those with a normal ICM (6). However, the current goal for researchers is to establish the optimal perinatal outcome of singletons according to blastocyst morphology. This has been recently analyzed by Bouillon et al., who found no increased rates of adverse obstetric and perinatal outcomes after transfer of blastocysts with poor morphological features (4). 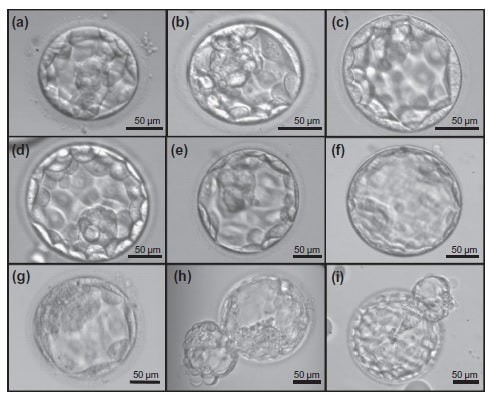 Figure 2. Examples of blastocyst grading: (a) 3AA blastocyst; (b) 3AB blastocyst; (c) 3BA blastocyst; (d) 4AA blastocyst; (e) 4AB blastocyst; (f) 4BA blastocyst; (g) 4CC blastocyst; (h) 5AA blastocyst; (i) 5CA blastocyst. For details of the EH stages and ICM and TE grades, see Materials and methods from Van den Abbeel (11). BLASTOCYST ASSESSMENT BY TIME-LAPSE TECHNOLOGY As previously explained, the most accepted blastocyst grading system is Gardner’s (17), based on the degree of blastocyst expansion and the morphological appearance of both the ICM and TE. However, since embryo development is a dynamic process, conventional grading practices may not detect subtle differences in morphology, which changes significantly over a time span of only a few hours (18). In order to obtain a complete picture of morpho-kinetic events occurring during embryo development a time-lapse system is needed. This technology offers continuous monitoring of embryos rather than just a limited number of discrete observations annotated through conventional assessment. Besides, time-lapse allows embryos to be cultured uninterruptedly, thus getting rid of embryo trafficking from and into the incubator (19). Nevertheless, the actual new and unique contribution of morpho-kinetics is the ability to predict how likely is for a zygote to reach the blastocyst stage in vitro. Several algorithms based on parameters detected by time-lapse, such as early divisions of cleavage-stage embryo, have recently been developed in IVF laboratories to predict blastocyst formation (20). In addition, some authors have made an effort to take time-lapse usefulness further, for instance, to predict the ploidy status of pre-implantation embryos (21, 22). Implantation potential of blastocysts can be evaluated by means of time-lapse during its development. In this regard, three main events are currently being investigated: duration of both compaction and blastulation plus number of blastocyst collapse events (19, 23, 24). Duration of compaction After several cell divisions during the initial stages of embryonic development, the intercellular boundaries become obscured in a process called compaction, which maximizes the intercellular contact and gives rise to the morula (25). Although the compaction of embryos has not received sufficient attention in the IVF field, some studies have focused on the relationships between compaction patterns and embryo developmental potential. Embryos that begin to compact before the eight-cell stage exhibit aberrant in vitro development. Conversely, embryos that complete compaction on day 5 have a lower ability to develop into high-quality blastocysts than those that compact on day 4 (26). These results suggest that the compaction patterns of embryos can facilitate the prediction of their ability to develop both in vitro and in vivo. An interesting work on this issue has been recently published by Mizobe and collaborators (23). The study retrospectively examined the outcome of 299 embryos from 243 patients, which were transferred at blastocyst stage. The whole early development was analysed by comparing morpho-kinetic parameters between implanted and non-implanted embryos, and measuring the time length of specific events, particularly of embryo compaction. Compaction length was calculated by using values of beginning and end of compaction. Beginning of compaction was considered as the time point when the intercellular boundaries became diffuse somewhere in the embryo, while fully compaction was defined as the point when blastomeres were finally unified into one cluster. Compaction length was significantly shorter in blastocysts resulting in pregnancies compared to those that failed to do so. These results indicate a correlation between the length of compaction and implantation potential. This finding is in agreement with the results from previous studies, which observed that the compaction patterns of embryos affected the rates of good-quality blastocyst formation and implantation (26, 27, 28). By contrast, some studies have reported that compaction time of embryos does not affect clinical pregnancy rates (29, 30). Duration of blastulation Blastulation is the process through which a morula becomes a blastocyst. Two different structures will arise to form the blastocyst out of the compacted blastomeres of the morula. The first sign of blastulation is compaction and differentiation of the outer blastomeres, forming the TE. This compaction gives the structure a watertight condition, allowing the fluid later secreted to be contained (31). Then, a different group of blastomeres normally located at the centre of the morula start to get closely attached to each other by the formation of Gap junctions, thus facilitating cell communication. It is these cells that differentiate into the ICM (the future embryoblast) and acquire a polarized location at one edge of the embryo. Such polarization creates a cavity, the blastocoel, and gives rise to the structure termed blastocyst. The trophoblasts (TE cells), in turn, continuously pump fluid into the blastocoel, which results in an enhanced size of the blastocyst. This increased volume leads the embryo to hatch through the zona pellucida (32). A recent study conducted by Mumusoglu analysed whether time-lapse morpho-kinetic variables differ among those euploid blastocysts that result in ongoing pregnancy after single embryo transfer (24). For that purpose, 129 patients who had been transferred a single embryo after an ICSI cycle with PGS were considered. Embryos were cultured in a time-lapse incubator up to the moment of TE biopsy, and 23 time-lapse morpho-kinetic parameters were annotated. After biopsy, blastocysts were vitrified and transferred within the next cycle. When comparing all time-lapse parameters, only blastulation time was statistically different: it had lasted shorter in successfully implanted blastocysts than in those that had not implanted. Blastulation time was calculated as the interval from initiation of blastulation up to full blastocyst formation (33, 34). Even though only a few studies have genetically tested euploid blastocysts (21, 22), all of them have pointed out that faster-developing euploid blastocysts might exhibit higher implantation potential. Even so, further large-scale studies are needed in order to confirm such an association (24). Blastocyst collapse events The phenomenon of blastocyst collapse is actually the shrinkage caused by the efflux of the blastocoel fluid due to the loss of cell bindings along the TE. When blastocysts expand, fluid gradually accumulates in the blastocoel -mediated by the sodium pump (Na+/K+-ATPase) (35), resulting in an increased pressure on both the TE and the zona. In parallel, TE cells produce lysins that are involved in the zona weakening and hatching. Formerly to implantation, the embryo needs to leave the zona behind, place adjacent to the endometrial epithelium and then make first contact with the uterus (36). Thus, embryo hatching from the zona is thought to be related to collapse-expansion cycles. By using a time-lapse monitoring system, it has been observed that many of the human blastocysts that reach stage 5 of expansion experience one or more collapse events of the blastocoel cavity, producing a separation of part (if not all) of the TE cells from the zona (19). In a study conducted by IVI Valencia and IVI Murcia clinics (19), blastocyst collapse was analysed to determine its potential influence on reproductive outcomes and whether it may serve for prognostic purposes. 460 patients and data from over 500 blastocysts known to have implanted were included in the study. Blastocyst collapse was considered to have occurred if the separation between TE and the zona pellucida was higher than 50% of the volume. Blastocysts that had experienced just one collapse event were found to present a significantly reduced implantation potential when compared to those transferred after having experienced none. The authors proposed that the molecular mechanisms underlying this association could be related to the mechanical stress suffered from by the embryo, which could result in an excessive energy consumption that would adversely affect the consequent development (19). 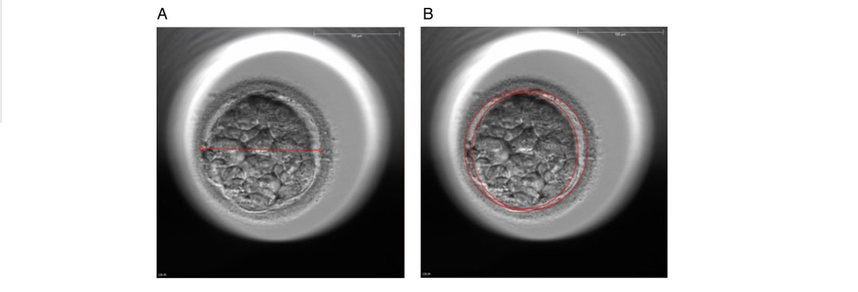 Figure 4. Drawing tools used with Embryovieverw for blastocyst collapse evaluation. First, a line was drawn across the embryo diameter (A). Then, the two circumferences that define the contracted blastocyst and the inner surface of the zona pellucida were outlined (B) [for more details, go to Materials and methods from Marcos (19)]. In spite of the data discussed above, the negative association between blastocyst collapse and implantation potential is not yet clear. In a report by Bodri and colleagues (37), blastocysts were classified according to the number of collapses: embryos with no collapses represented 54% of the total, 22% of the embryos had experienced one single collapse, and multiple collapse events occurred in 24% of the blastocysts. Whereas the live birth rate was observed to decrease as the number of embryo collapse increased, multivariate analyses suggested blastocyst collapse not to be a significant predictor. Rather, it was found to be a confounding factor, along with other morpho-kinetic variables such as time up to two-cell division completion and female age. Therefore, it was concluded that blastocyst collapse patterns should not be evaluated alone without stronger predictors of reproductive outcomes being taken into account (37). TIME-LAPSE AS A MEANS TO EVALUATE EMBRYO QUALITY As previously exposed, the use of time-lapse technology is recently common in embryology laboratories because of its noticeable potential for enhancing embryo selection. Using these technologies, Desai et al. analysed possible kinetic differences between embryos with limited potential and those that accomplished in vitro blastocyst formation and/or implantation (38). Certain parameters such as time of pronuclear formation and cleavage stage were found to be different in embryos reaching blastocyst stage vs. poor-quality embryos. Moreover, a large number of embryos were found to present multinucleation and reverse cleavage, but they were able to form a blastocyst with optimal criteria for freezing (38), which resembles previous reports on the dynamic nuclear formation of blastocysts by Ergin and coauthors (39). With respect to blastocyst formation, Motato et al. (2016) proposed two models to classify embryos based on their probability of reaching blastocyst stage and implantation (40). However, the study was limited by parameters such as subjective criteria from different clinics with different culture media (40). Consequently, it would be reasonable to keep on research on this subject in order to achieve a consensus regarding embryo classification and implantation potential (40). CONSIDERING DAY-2 AND DAY-3 EMBRYO MORPHOLOGY BEFORE DAY-5 TRANSFER Even though blastocyst stage is currently widely accepted as the optimal moment for embryo transfer, cleavage stage has been traditionally regarded as the right moment in global practice. In fact, it still continues to be so in some laboratories, and early transfer into the uterus has been proposed to be advantageous to the embryo due to the limited time exposed to the in vitro environment (41). However, there exist two main arguments supported by extensive scientific literature to explain why blastocyst transfer after extended culture has advantages over the traditional cleavage-stage transfer: First of all, when the embryo arrives to the uterus in natural conditions it has already reached morula stage, which corresponds to, at least, day 4 of in vitro culture (42). This means blastocyst stage is the most physiologically compatible stage for transfer, since it allows a better synchronization between embryonic stage and endometrial receptivity (43) [you can read more about the optimal day for embryo transfer in our previous article here]. Secondly, several studies have reported higher implantation potential for blastocysts compared to cleavage-stage embryos (6, 41), the first transferred blastocyst being reported in 1995 (44). Furthermore, some authors have postulated that a large proportion of morphologically normal day-3 embryos are actually chromosomally abnormal or mosaic, which may contribute to the 80-90% rate of implantation failure observed after cleavage-stage embryo transfer (45). EXTENDING EMBRYO CULTURE UP TO BLASTOCYST STAGE Considering the need for further studies on the subject, and the fact that day-3 embryos can actually implant and develop successfully, does it really make sense to extend embryo culture up to blastocyst stage? As above-stated, morphologically normal embryos may actually present chromosome abnormalities, which proves the insufficiency of morphological criteria to evaluate implantation rates (46). Because of embryo plasticity, the proportion of chromosomally abnormal cells varies within the culture; corrupted cells can be eliminated, thus resulting in a good-quality blastocyst developing from a poor-quality cleavage-stage embryo (47). Some studies have evaluated pregnancy rates derived from transfers of blastocysts with previous poor quality as cleavage-stage embryos, finding an approximate success rate of 45% after culture and freezing of embryos at an early stage for another cycle. The conclusion of this being a valid practice to avoid the repetition of IVF-ICSI treatments (48) agrees with recent findings showing that low-scoring day-2 or 3 embryos, which are not considered transferable, can still result in successful blastulation and end up in a live birth (49, 50). All this said, the right question now would be: should day-2 and day-3 embryo morphology be considered before transfer at day 5 when blastocysts reach a similar good quality? A recent retrospective study by Herbemont has suggested that only the quality of the transferred blastocyst may be predictive of the subsequent clinical outcome, whereas morphological aspects at day 2 or day 3 have limited interest (51). These same results had previously been observed by Guerif; even though early morphological parameters were relatively helpful to predict blastocyst development, their value to predict blastocyst morphology was limited, and so they provided no significant additional information that could prognosticate blastocyst implantation and live birth rates (6). A few years earlier and with the same goal in mind, Zech and coauthors carried out a prospective randomized study in which they compared ongoing pregnancy rates per single embryo transfer between day 3 and day 5. When good-quality embryos were available, pregnancy rates were found to be higher after blastocyst transfer. Therefore, the authors concluded that morphological criteria-based seleccion at day 3 may not be a suitable procedure when just one embryo is to be transferred out of a cohort of all morphologically good ones. Thus, and as stated by the authors, extending embryo culture up to day 5 may result in a better strategy in order to correctly identify and select those embryos with higher implantation potential, provided there is a sufficient number of top-quality eight-cell embryos available (52). On the contrary, a study performed by Silber (2014) found that blastocysts arising from poor-quality embryos displayed lower implantation and pregnancy rates compared to good-quality embryos. These discrepancies could be due to different criteria used to score embryo quality (53). So, in order to minimise discrepancies between studies, the use of time-lapse is currently established as a common approach to evaluate embryo morpho-kinetics. In fact, reduction in the time of embryo exposure to the environment outside the incubator has been demonstrated to enhance both embryo quality and blastulation rates (54). Nevertheless, to answer the question previously postulated, a prospective randomized study would be needed that compare at least two similar good-quality blastocysts, one arising from a good-quality day-2/3 embryo, and to the other from a poor-quality one (51). CONCLUSIONS It is important to take into account that the main population features of different patients, such as paternal age, maternal BMI, parental smoking or cause of infertility may influence clinical outcomes. Moreover, certain methodological aspects also need to be considered, like blastocyst evaluation by the same personal (in order to minimize variation) or the consistent use of the same type of culture media (to avoid potential effects on birth-weight and other traits), just as previously suggested (4). A universal embryo grading system needs to be validated, before widespread implementation in IVF laboratories. Also, it has not yet been clearly established which morphological feature of blastocysts (expansion, TE or ICM) is the most reliable as a predictive factor for post-transfer implantation success. Consequently, there is still a debate between authors about the true outcomes of single transfer of low-quality blastocysts (4). Morpho-kinetics assessment, along with chromosomal screening, may ultimately help identify euploid embryos with the highest developmental potential (55). Since these features are susceptible of being affected by in vitro culture conditions, each embryology laboratory should define their own cut-off points in order to standardise time-lapse variables (24). Finally, it should be taken into account the fact that embryo quality is not the only parameter with influence on implantation rates; endometrial receptivity is also involved, and it may be greatly determined by a variety of factors (56) [to learn more about endometrium status and receptivity, read our previous post here]. REFERENCES
1- Souza Setti A, Ferreira RC, Paes de Almeida, Ferreira Braga D, de Cassia Savio Figueira R, Iaconelli A Jr, Borges E Jr. Intracytoplasmic sperm injection outcome versus intracytoplasmic morphologically selected sperm injection outcome: a meta-analysis. Reprod Biomed Online 2010;21:450-455. 2- Meldrum DR. Blastocyst transfer: a natural evolution. Fertil Steril 1999;72:216–7. 3- Balaban B, Urman B, Sertac A, Alatas C, Aksoy S, Mercan R. Blastocyst quality affects the success of blastocyst-stage embryo transfer. Fertil Steril 2000;74:28-287. 4- Bouillon C, Celton N, Kassem S, Frapsauce C, Guérif F. Obstetric and perinatal outcomes of singletons after single blastocyst transfer: is there any difference according to blastocyst morphology? Reprod Biomed Online 2017;35:197–207. [Internet]. 5- Stecher A, Vanderzwalmen P, Zintz M, Wirleitner B, Schuff M, Spitzer D, Zech NH. Transfer of blastocysts with deviant morphological and morphokinetic parameters at early stages of in-vitro development: a case series. Reprod Biomed Online 2014;28:424-435. 6- Guerif F, Lemseffer M, Leger J, Bidault R, Cadoret V, Chavez C, Gasnier O, Saussereau MH, Royere D. Does early morphology provide additional selection power to blastocyst selection for transfer? Reprod Biomed Online 2010;21:510– 519. 7- Alpha Scientists in Reproductive Medicine and ESHRE Special Interest Group of Embryology. The Istanbul consensus workshop on embryo assessment: proceedings of an expert meeting. Hum Reprod 2011;26:1270 –1283. 8- Gardner DK, Schoolcraft WB, Wagley L, Schlenker T, Stevens J, Hesla J. A prospective randomized trial of blastocyst culture and transfer in in-vitro fertilization. Hum Reprod 1998;13:3434-3440. 9- Balaban B, Yakin K, Urman B. Randomized comparison of two different blastocyst grading systems. Fertil Steril 2006;85:559– 563. 10- Richardson A, Brearley S, Ahitan S, Chamberlain S, Davey T, Zujovic L, Hopkisson J, Campbell B, Raine-Fenning N. A clinically useful simplified blastocyst grading system. Reprod Biomed Online 2015;31:523–530. 11- Van den Abbeel E, Balaban B, Ziebe S, Lundin K, Cuesta MJ, Klein BM, et al. Association between blastocyst morphology and outcome of singleblastocyst transfer. Reprod Biomed Online 2013;27:353–61. 12- Shapiro BS, Daneshmand ST, Garner FC, Aguirre M, Thomas S. Large blastocyst diameter, early blastulation, and low preovulatory serum progesterone are dominant predictors of clinical pregnancy in fresh autologous cycles. Fertil Steril 2008;90:302–9. 13- Shapiro BS, Richter KS, Harris DC, Daneshmand ST. A comparison of day 5 and 6 blastocyst transfers. Fertil Steril 2001;75:1126–30. 14- Ahlström A, Westin C, Reismer E, Wikland M, Hardarson T. Trophectoderm morphology: an important parameter for predicting live birth after single blastocyst transfer. Hum Reprod 2011;26:3289– 3296. 15- Hill MJ, Richter KS, Heitmann RJ, Graham JR, Tucker MJ, Decherney AH, Browne PE, Levens ED. Trophectoderm grade predicts outcomes of single-blastocyst transfers. Fertil Steril 2013;99: 1283–1289. 16- Almagor M, Harir Y, Fieldust S, Or Y, Shoham Z. Ratio between inner cell mass diameter and blastocyst diameter is correlated with successful pregnancy outcomes of single blastocyst transfers. Fertil Steril 2016;106: 1386–1391. [Internet] 17- Gardner DK, Lane M, Stevens J, Schlenker T, Schoolcraft WB. Blastocyst score affects implantation and pregnancy outcome: towards a single blastocyst transfer. Fertil Steril. 2000;73(6):1155–8. 18- Cruz M, Garrido N, Herrero J, Pérez-Cano I, Muñoz M, Meseguer M. Timing of cell division in human cleavage-stage embryos is linked with blastocyst formation and quality. Reprod Biomed Online. 2012;25(4):371–81. 19- Marcos J, Pérez-Albalá S, Mifsud A, Molla M, Landeras J, Meseguer M. Collapse of blastocysts is strongly related to lower implantation success: a time-lapse study. Hum Reprod. 2015;30(11):2501–8. 20- Meseguer M, Herrero J, Tejera A, Hilligsøe KM, Ramsing NB, Remohí J. The use of morphokinetics as a predictor of embryo implantation. Hum Reprod. 2011;26(10):2658–71. 21- Yang Z, Zhang J, Salem SA, Liu X, Kuang Y, Salem RD, et al. Selection of competent blastocysts for transfer by combining time-lapse monitoring and array CGH testing for patients undergoing preimplantation genetic screening: a prospective study with sibling oocytes. BMC Med Genomics. 2014;7(1):38. 22- Kofinas JD, Tiegs A, Kramer YG, McCulloh DH, Grifo JA. Do Time Lapse Morphokinetic (TLM) Parameters Distinguish between Good Versus Poor Prognosis Embryos of Known Ploidy Status? Fertil Steril. 2015;103(2):e27. 23- Mizobe Y, Ezono Y, Tokunaga M, Oya N, Iwakiri R. Selection of human blastocysts with a high implantation potential based on timely compaction. J Assist Reprod Genet. 2017;34(8):991-997. 24-Mumusoglu S, Ozbek IY, Sokmensuer LK, Polat M, Bozdag G, Papanikolaou E, et al. Duration of blastulation may be associated with ongoing pregnancy rate in single euploid blastocyst transfer cycles. Reprod Biomed Online. 2017;35(6):633-639. 25- Fleming TP, Sheth B, Fesenko I. Cell adhesion in the preimplantation mammalian embryo and its role in trophectoderm differentiation and blastocyst morphogenesis. Front Biosci. 2001;6(1):D1000–7. 26- Iwata K, Yumoto K, Sugishima M, Mizoguchi C, Kai Y, Iba Y, et al. Analysis of compaction initiation in human embryos by using time-lapse cinematography. J Assist Reprod Genet. 2014;31(4):421–6. 27- Skiadas CC, Jackson K V, Racowsky C. Early compaction on day 3 may be associated with increased implantation potential. Fertil Steril. 2006;86(5):1386–91. 28- Tao J, Tamis R, Fink K, Williams B, Nelson-White T, Craig R. The neglected morula/compact stage embryo transfer. Hum Reprod. 2002;17(6):1513–8. 29- Chamayou S, Patrizio P, Storaci G, Tomaselli V, Alecci C, Ragolia C, et al. The use of morphokinetic parameters to select all embryos with full capacity to implant. J Assist Reprod Genet. 2013;30(5):703–10. 30-Kirkegaard K, Kesmodel US, Hindkjær JJ, Ingerslev HJ. Time-lapse parameters as predictors of blastocyst development and pregnancy outcome in embryos from good prognosis patients: a prospective cohort study. Hum Reprod. 2013;28(10):2643–51. 31- Forgacs G, Newman SA. Biological physics of the developing embryo. Cambridge University Press; 2005. 32- Larsen WJ. Human embryology. Churchill Livingstone; 2001. 33- Campbell A, Fishel S, Bowman N, Duffy S, Sedler M, Hickman CFL. Modelling a risk classification of aneuploidy in human embryos using non-invasive morphokinetics. Reprod Biomed Online. 2013;26(5):477–85. 34- Ciray HN, Campbell A, Agerholm IE, Aguilar J, Chamayou S, Esbert M, et al. Proposed guidelines on the nomenclature and annotation of dynamic human embryo monitoring by a time-lapse user group. Hum Reprod. 2014;29(12):2650–60. 35- Baltz JM, Smith SS, Biggers JD, Lechene C. Intracellular ion concentrations and their maintenance by Na+/K+-ATPase in preimplantation mouse embryos. Zygote. 1997;5(1):1–9. 36- Biggers JD. Reflections on the culture of the preimplantation embryo. Int J Dev Biol. 2004;42(7):879–84. 37- Bodri D, Sugimoto T, Serna JY, Kawachiya S, Kato R, Matsumoto T. Blastocyst collapse is not an independent predictor of reduced live birth: a time-lapse study. Fertil Steril. 2016;105(6):1476–83. 38-Desai N, Ploskonka S, Goodman LR, Austin C, Goldberg J, Falcone T. Analysis of embryo morphokinetics, multinucleation and cleavage anomalies using continuous time-lapse monitoring in blastocyst transfer cycles. Reprod Biol Endocrinol. 2014;12:54. 39- Ergin EG, Calişkan E, Yalçinkaya E, Oztel Z, Cökelez K, Ozay A, et al. Frequency of embryo multinucleation detected by time-lapse system and its impact on pregnancy outcome. Fertil Steril. 2014;102(4):1029-33.e1. 40- Motato Y, de los Santos MJ, Escriba MJ, Ruiz BA, Remohí J, Meseguer M. Morphokinetic analysis and embryonic prediction for blastocyst formation through an integrated time-lapse system. Fertil Steril. 2016;105(2):376-84.e9. 41- Glujovsky D, Farquhar C, Quinteiro Retamar AM, Alvarez Sedo CR, Blake D. Cleavage stage versus blastocyst stage embryo transfer in assisted reproductive technology. Cochrane Database Syst Rev. 2016(6):CD002118. 42- Croxatto HB, Fuentealba B, Díaz S, Pastene L, Tatum HJ. A simple nonsurgical technique to obtain unimplanted eggs from human uteri. Am J Obstet Gynecol. 1972;112(5):662-8. 43- Valbuena D, Martin J, de Pablo JL, Remohí J, Pellicer A, Simón C. Increasing levels of estradiol are deleterious to embryonic implantation because they directly affect the embryo. Fertil Steril. 2001;76(5):962-8. 44- Edwards RG, Brody SA. History and ethics of assisted human conception. Principles and Practice of Assisted Human Reproduction. Philadelphia: WB Sauders, 1995:17–47. 45- Magli MC, Gianaroli L, Munné S, Ferraretti AP. Incidence of chromosomal abnormalities from a morphologically normal cohort of embryos in poor-prognosis patients. J Assist Reprod Genet. 1998;15(5):297-301. 46- Meseguer M, Rubio I, Cruz M, Basile N, Marcos J, Requena A. Embryo incubation and selection in a time-lapse monitoring system improves pregnancy outcome compared with a standard incubator: a retrospective cohort study. Fertil Steril. 2012;98(6):1481-9.e10. 47- Fragouli E, Alfarawati S, Spath K, Jaroudi S, Sarasa J, Enciso M, et al. The origin and impact of embryonic aneuploidy. Hum Genet. 2013;132(9):1001-13. 48- Kaartinen N, Das P, Kananen K, Huhtala H, Tinkanen H. Can repeated IVF-ICSI-cycles be avoided by using blastocysts developing from poor-quality cleavage stage embryos? Reprod Biomed Online. 2015;30(3):241-7. 49- Hershko Klement A, Ovadia M, Wiser A, Berkovitz A, Shavit T, Nemerovsky L, et al. What we learned from extended culture of 'rejected' day-3 cleavage stage embryos: a prospective cohort study. J Ovarian Res. 2017;10(1):35. 50- Sallem A, Santulli P, Barraud-Lange V, Le Foll N, Ferreux L, Maignien C, et al. Extended culture of poor-quality supernumerary embryos improves ART outcomes. J Assist Reprod Genet. 2017. 51- Herbemont C, Sarandi S, Boujenah J, Cedrin-Durnerin I, Sermondade N, Vivot A, et al. Should we consider day-2 and day-3 embryo morphology before day-5 transfer when blastocysts reach a similar good quality? Reprod Biomed Online. 2017;35(5):521-8. 52- Zech NH, Lejeune B, Puissant F, Vanderzwalmen S, Zech H, Vanderzwalmen P. Prospective evaluation of the optimal time for selecting a single embryo for transfer: day 3 versus day 5. Fertil Steril. 2007;88(1):244-6. 53- Silber, S., 2014. Blastocysts that came from better quality day 3 embryos give higher pregnancy rates than blastocysts that derive from poor quality day 3 embryos. Ovarian Club, Paris. 54- Zhang JQ, Li XL, Peng Y, Guo X, Heng BC, Tong GQ. Reduction in exposure of human embryos outside the incubator enhances embryo quality and blastulation rate. Reprod Biomed Online. 2010;20(4):510-5. 55- Zaninovic N, Irani M, Meseguer M. Assessment of embryo morphology and developmental dynamics by time-lapse microscopy: is there a relation to implantation and ploidy? Fertil Steril. 2017;108(5):722-9. 56- Valbuena D, Valdes CT, Simon C. Introduction: Endometrial function: facts, urban legends, and an eye to the future. Fertil Steril. 2017;108(1):4-8.
4 Comments
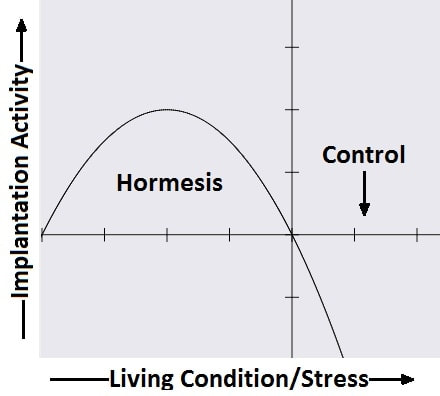
Authors: Javier del Río, Noemi Díaz and Belén Gómez INTRODUCTION What is cryopreservation? The first successful in vitro fertilization (IVF) treatment was in 1978. Since that, there have been a remarkable number of advances in assisted reproductive technologies (ART). Initially, all available embryos were transferred in IVF treatments owing to its low success rate. However, improvements on clinical and laboratory aspects led not only to increased pregnancy rates, but also to increased risk of multiple pregnancies. To prevent this, fewer embryos are transferred and leftover embryos are cryopreserved for potential future cycles use (1). The first pregnancy resulting from transferring a thawed cryopreserved human embryo was reported in 1983 in Australia (2), and the first live birth following embryo cryopreservation was reported in 1984 in The Netherlands (3). Subsequently, the need for an effective cryopreservation program arose from rapid development and improvements of assisted reproductive technology protocols (1). Cryopreservation is a method that requires cells and embryos to be exposed to non-physiological ultra-low temperatures (from -20°C to -196°C) (Fig.2). It aims to achieve “cryogenic suspension of life” through multiple steps, although this puts the elements at risk of damage or “cryoinjury” during temperature changes and phase transitions. These damages could be chilling injury or ice crystal formation, for instance, as a result of the water exchange between the intra- and extracellular compartments, consequence of dramatic changes in osmotic potential (osmotic shock). Therefore, vitrification requires the use of cryoprotectants to avoid the formation of ice crystals in the cells. Two types of cryoprotectants are necessary: permeating and non-permeating. Mixing both at different relative concentrations reduces intracellular ice formation by removing water from inside the cell. Additionally, it creates an osmotic gradient that helps restrict water movement across the cell membrane, thereby preventing osmotic shock (4). There are two typical methods used for cryopreservation: slow freezing and rapid freezing to achieve vitrification. Vitrification is a term used to describe the transformation of a solution into glass by a dramatic increase in viscosity. This method requires to minimize the time for the sample to be exposed to temperature ranges associated with chilling injury and ice crystal formation. As slow freezing, vitrification causes cell dehydration using cryoprotectants. However, unlike that, there is no attempt to maintain equilibrium on both sides of cell membrane (4). The time frame required to reach ultralow temperatures by vitrification is very brief, almost instantaneous. But, the main concern is the need for using high concentrations of cryoprotectant solutions. These might lead to osmotic shock and it could be toxic to cells, affecting embryo survival. Nevertheless, it is possible to limit toxicity by mixing different cryoprotectants, thereby decreasing their relative concentration and the exposure time of embryos to the solution (5). How efficient is the vitrification? This technique seems to be more attractive than slow freezing because it does not require expensive equipment. It uses small amount of liquid nitrogen and it is a simpler technique to perform once the embryologist has gained enough experience in it (6). A recent research performed by Viladimoiv et al. suggests advantages arising from the freezing and thawing process; the authors hypothesize a theory about “cryo-treatment of the embryo”. According to these authors, as a result of freezing or thawing of the embryos there is a decrease in reactive oxygen species levels, in the rate of mitochondrial DNA mutation and cells detoxification is carried out. Also, the authors describe another mechanism involved in restoring the mitochondrial activity (“jumping effect”) which is part of the physiological process of implantation. However, current available data cannot confirm the hypothesis yet (7). Advantages and disadvantages of fresh and frozen cycles Nowadays, fresh embryo transfers (ET) are the most common choice in IVF cycles (8). Nevertheless, in the last years, controlled ovarian stimulation has increased the uncertainty on the possible adverse effects of the ovarian hyperstimulation syndrome (OHSS), and also on possible deleterious effects on the endometrium and implications in obstetric and perinatal results (9). In spite of this, recent developments in cryopreservation of oocytes and embryos have led to substantial improvement in IVF outcomes. This resulted in a significant increase in the number of cycles with frozen embryo transfer (FET), which subsequently led to the enhancement of live births rate (10). What are the advantages of a frozen cycle? Ovarian hyperstimulation syndrome The first strong argument for FET strategy is the prevention of OHSS, that results from an increase in vascular permeability (11,8). OHSS is a medical condition affecting the ovaries of some women who take fertility medication to stimulate oocyte growth. OHSS arguably remains a major cause of morbidity in IVF treatment (10). During a fresh cycle, a woman has to undergo hormonal treatment to regulate her menstrual period, to stimulate the development of multiple oocytes (superovulation), and to encourage their maturation (11, 12). However, in a frozen cycle (FC) the patient does not have to go through ovarian stimulation or egg retrieval depending on their circumstances (13). Many people find that FETs are less stressful than fresh cycles because they do not have to worry about oocytes production or whether there will be viable embryos, since those procedures have already been done (9). Deleterious effects on the embryo The optimization of vitrification protocols has reduced the deleterious effects that this process may produce in embryos. Also, it have been observed similar survival and embryo development in FCs compared to fresh cycles (10). Moreover, best quality embryos, morphologywise, can be stored and transferred in a future cycle in better conditions. These data have allowed for an increment of success rates and the confidence of sanitary personnel and patients over FCs (5). Endometrial receptivity The implantation process, one of the crucial steps in the success of ART, requires a reciprocal interaction between the embryo and the endometrium during a small period of time called window of implantation. This interaction involves the embryo, along with its inherent molecular program of cell growth and differentiation, as well as differentiation of endometrial cells into an adequate uterine receptivity (11). Some patients may find easier to turn to FCs, since dealing with the whole process of medication during a normal cycle for ovarian stimulation may result psychologically and emotionally overwhelming. In this regard, FC may also provide a better outcome (3). The importance of an adequate endometrial environment in ART is highlighted in those patients who resort to oocyte donation, where there must be a synchronization between donor and recipient in fresh cycles. Those cases that require an improvement in endometrial receptivity to stimulate implantation of these donor oocytes seem to obtain better results in frozen cycles or in the next fresh cycle (8). Multiplet pregnancies are one of the major safety concerns of IVF due to the increased risk of neonatal and maternal complications. To achieve good results, to would be ideal to select the optimal single embryo to be transferred. Elective single embryo transfer (eSET) is the most effective way to reduce those risky pregnancies (14). How can cryopreservation damage embryos? Upon analyzing some ART studies and results, embryos are able to adapt and develop in a large range of culture media, showing different gene expression models in different environments. Cryopreservation causes stress in embryos and it is known as “hormesis”(5) (Fig.3). However, if the conditions are too unfavorable or toxic, mitochondrial activity is suppressed below the threshold necessary for the development of the embryo, so that implantation in the endometrium will be affected (5). Results of embryo transfer in fresh cycles vs. frozen cycles The main current objective of IVF professionals is to improve pregnancy rates in both fresh and frozen-thawed cycles. It is clear that embryo and endometrial receptivity are important factors to promote pregnancy rate. Recently, many researches showed FET can enhance the embryo utilization rate and improve the success rate in contrast to other research lines (15). In Roque et al. systematic meta‐analysis for 633 cycles in women aged 27-33 years old showed that FET resulted in a statistically significant increase in the ongoing pregnancy rate and clinical pregnancy compared with the fresh transfer group (8). Interestingly, the fresh group showed a higher miscarriage rate, but no statistical difference was found when compared with the frozen group. According to these data, it seems that the results of IVF-ICSI cycles can be improved by performing the FET especially in patients with normal or high follicular response. This advantage could be explained thanks to a more physiological preparation of endometrium. Several studies have also shown good results with cryopreservation of all embryos and subsequent FET in those patients most susceptible to develop OHSS (8, 16-19). In contrast, Shavit et al. found lower rates of clinical pregnancy and live births in the vitrified-warmed blastocyst group. The difference in implantation and pregnancy rates could be attributed to a higher proportion of good-quality embryos in the fresh blastocysts transfer group. They suggest that in fresh cycles highest quality blastocyst is selected for transfer and the rest are usually vitrified. Thus, vitrified-warmed blastocysts may have slightly poorer grade after warming and prior to transfer (20). In addition, it is necessary to take into account those cycles with frozen oocytes. Braga et al. found that warmed oocytes transferred in endometrial prepared cycles yield better clinical outcomes than fresh ETs. Indeed, they found that fertilization rate, embryo quality, and developmental competence was decreased in embryos derived from vitrified oocytes (12). Conversely, previous studies have suggested that the results of oocyte vitrification followed by ICSI are not inferior with regard to fertilization, embryo developmental competence, pregnancy rates, and live birth (21, 22, 23). An interesting point found in Braga et al. research is that even with lower embryo developmental quality, warmed oocytes transferred in endometrial prepared cycles resulted in higher pregnancy and implantation rates compared with transfer in fresh cycles. This finding strongly suggests that controlled ovarian stimulation impacts endometrial receptivity, which may be a possible cause of implantation failure after ovarian stimulation (12). Indeed, some studies have suggested that pregnancy rate is inversely related to serum progesterone levels on the day of hCG administration (24-27). It has been demonstrated that elevated progesterone levels on hCG trigger day negatively influence the pregnancy, regardless of the oocyte quality. Raised concentrations of progesterone in the late follicular phase are likely to influence the secretory changes of the endometrium, leading to an asynchrony between embryo and endometrial dialogue, which may result in reduced implantation rate (12). Another issue to consider is the obstetric and perinatal outcomes of frozen-thawed cycles. Maheshwari et al. quantified in a meta-analysis the obstetric and perinatal risks for singleton pregnancies after FET and compared it with those after fresh embryo transfer (28). They indicated better perinatal outcomes in singleton pregnancies after the transfer of frozen‐thawed embryos when compared to fresh IVF embryos. This could be explained by antepartum hemorrhage, very preterm birth (delivery at <32 weeks), preterm delivery (delivery at <37 weeks), small for gestational age, low birth weight (birth weight <2500 g), and perinatal mortality significantly lower in women who received frozen embryos than those transferred with fresh embryos (29, 28). It is important to note that most studies comparing perinatal outcome of singleton births conceived after fresh and cryopreserved ETs included both single and multiple ETs. Therefore, part of the adverse perinatal outcome may be attributed to the vanishing twin phenomenon, which occurs in up to 10% of multiple ETs resulting in a singleton live birth (20). What can we conclude? Elective embryo cryopreservation followed by single FET has attracted increasing attention and has been regarded as a potential innovation of IVF treatment. Choosing the well-selected embryo could further increase the chance of live birth of a eSET, which is of high clinical significance. However, there are great gaps in the literature about the risk/benefit ratio of this strategy, which includes multiple steps of treatment (30). The good outcomes in FC might be associated with having a well‐balanced embryo‐endometrium interaction in FC, and also with lacking controlled ovarian hyperstimulation, which may adversely affect endometrial receptivity during fresh IVF cycles. In addition, when hormone replacement cycles were applied in FETs, estrogen and progesterone were given in physiological doses to mimic natural cycles, while supraphysiological doses of gonadotropins were given in fresh cycles (31). On the other hand, other authors find fresh cycles as the best choice, especially in patients who resort to oocyte donation. In fact, it seems that there is a higher proportion of good-quality embryos in fresh blastocysts compared to vitrified-warmed blastocysts, which may have slightly poorer grade after warming and prior to transfer. (8, 20). In conclusion, each case must be individualized in relation to clinical characteristics of the patients and to oocyte, seminal and embryo quality. By doing so, results will be optimized in each cycle and the chances of achieving a live birth will be highly improved. REFERENCES:
1. Wong KM, Mastenbroek S, Repping S. Cryopreservation of human embryos and its contribution to in vitro fertilization success rates. Fertil Steril. 2014;102(1):19-26. 2. Trounson A, Mohr L. Human pregnancy following cryopreservation, thawing and transfer of an eight-cell embryo. Nature. 1983;305(5936):707-9. 3. Zeilmaker GH, Alberda AT, van Gent I, Rijkmans CM, Drogendijk AC. Two pregnancies following transfer of intact frozen-thawed embryos. Fertil Steril. 1984; 42(2):293-6. 4. Sparks AE. Human embryo cryopreservation-methods, timing, and other considerations for optimizing an embryo cryopreservation program. Semin Reprod Med. 2015;33(2):128-44. 5. Konc J, Kanyó K, Kriston R, Somoskői B, Cseh S. Cryopreservation of embryos and oocytes in human assisted reproduction. Biomed Res Int. 2014;2014:307268. 6. Loutradi KE, Kolibianakis EM, Venetis CA, Papanikolaou EG, Pados G, Bontis I, et al. Cryopreservation of human embryos by vitrification or slow freezing: a systematic review and meta-analysis. Fertil Steril. 2008;90(1):186-93. 7. Vladimirov IK, Tacheva D, Diez A. Theory about the Embryo Cryo-Treatment. Reprod Med Biol. 2017;16:118–125. 8. Roque M, Lattes K, Serra S, Solá I, Geber S, Carreras R, Checa MA. Fresh embryo transfer versus frozen embryo transfer in in vitro fertilization cycles: a systematic review and meta-analysis. Fertil Steril. 2013;99(1):156-62. 9. Gurbuz AS, Gode F, Ozcimen N, Isik AZ.Gonadotrophin-releasing hormone agonist trigger and freeze-all strategy does not prevent severe ovarian hyperstimulation syndrome: a report of three cases. Reprod Biomed Online 2014;29:541-544. 10. Lattes K, Prat M, Robles A, Carreras R, Brassesco M, Checa MA. Ciclos de criopreservación y vitrificación de ovocitos y embriones: indicaciones y transferencia diferida. Guía 21 de Práctica Clínica de la SEF y de la SEGO. 11. Lessey BA. Endometrial receptivity and the window of implantation. Baillieres Best Pract Res Clin Obstet Gynaecol. 2000;14(5):775-88. 12. Braga D, Setti A, Figueira R, Azevedo M, Iaconelli A, Lo Turco E et al. Freeze-all, oocyte vitrification, or fresh embryo transfer? Lessons from an egg-sharing donation program. Fertil Steril. 2016;106(3):615-622. 13. Shapiro BS, Daneshmand ST, Garner FC, Aguirre M, Hudson C. Clinical rationale for cryopreservation of entire embryo cohorts in lieu of fresh transfer. Fertil Steril. 2014;102:3-9. 14. Tobias T, Sharara FI, Franasiak JM, Heiser PW, Pinckney-Clark E. Promoting the use of elective single embryo transfer in clinical practice. Fertil Res Pract. 2016;2(1):1-9. 15. Shen C, Shu D, Zhao X, Gao Y. Comparison of clinical outcomes between fresh embryo transfers and frozen-thawed embryo transfers. Iran J Reprod Med. 2014. Jun;12(6):409–14. 16. Griesinger G, von Otte S, Schroer A, Ludwig AK, Diedrich K, Al-Hasani S, et al. Elective cryopreservation of all pronuclear oocytes after GnRH agonist triggering of final oocyte maturation in patients at risk of developing OHSS: a prospective, observational proof-of-concept study. Hum Reprod. 2007;22(5):1348-1352. 17. D'Angelo A. Ovarian hyperstimulation syndrome prevention strategies: cryopreservation of all embryos. Semin Reprod Med. 2010;28(6):513-518. 18. Griesinger G, Schultz L, Bauer T, Broessner A, Frambach T, Kissler S. Ovarian hyperstimulation síndrome prevention by gonadotropin-releasing hormone agonist triggering of final oocyte maturation in a gonadotropin-releasing hormone antagonist protocol in combination with ‘‘freeze-all’’ strategy: a prospective multicentric study. Fertil Steril. 2011;95(6):2029-2033. 19. Devroey P, Polyzos NP, Blockeel C. An OHSS-Free Clinic by segmentation of IVF treatment. Hum Reprod. 2011;26(10):2593-2597. 20. Shavit T, Oron G, Weon-Young S, Holzer H, Tulandi T. Vitrified-warmed single-embryo transfers may be associated with increased maternal complications compared with fresh single-embryo transfers. Reprod Biomed Online. 2017;35(1):94-102. 21. Trokoudes KM, Pavlides C, Zhang X. Comparison outcome of fresh and vitri- fied donor oocytes in an egg-sharing donation program. Fertil Steril. 2011; 95:1996-2000. 22. Herrero L, Pareja S, Aragones M, Cobo A, Bronet F, Garcia-Velasco JA. Oocyte versus embryo vitrification for delayed embryo transfer: an observational study. Reprod Biomed Online. 2014;29:567-72. 23. Rienzi L, Romano S, Albricci L, Maggiulli R, Capalbo A, Baroni E, et al. Embryo development of fresh ‘versus’ vitrified metaphase II oocytes after ICSI: a prospective randomized sibling-oocyte study. Hum Reprod. 2010;25:66-73. 24. Xu, B., Li, Z., Zhang, H., Jin, L., Li, Y., Ai, J. et al, Serum progesterone level effects on the outcome of in vitro fertilization in patients with different ovarian response: an analysis of more than 10,000 cycles. Fertil Steril. 2012;97 (1321-7.e1-4). 25. Wu, Z., Li, R., Ma, Y., Deng, B., Zhang, X., Meng, Y. et al, Effect of HCG-day serum progesterone and oestradiol concentrations on pregnancy outcomes in GnRH agonist cycles. Reprod Biomed Online. 2012;24:511–520. 26. Bosch, E., Labarta, E., Crespo, J., Simon, C., Remohi, J., Jenkins, J. et al, Circulating progesterone levels and ongoing pregnancy rates in controlled ovarian stimulation cycles for in vitro fertilization: analysis of over 4000 cycles. Hum Reprod. 2010;25:2092–2100. 27. Hamdine, O., Macklon, N.S., Eijkemans, M.J., Laven, J.S., Cohlen, B.J., Verhoeff, A. et al, Elevated early follicular progesterone levels and in vitro fertilization outcomes: a prospective intervention study and meta-analysis. Fertil Steril. 2014;102:448–454.e1. 28. Maheshwari A, Pandey S, Shetty A, Hamilton M, Bhattacharya S. Obstetric and perinatal outcomes in singleton pregnancies resulting from the transfer of frozen thawed versus fresh embryos generated through in vitro fertilization treatment: a systematic review and meta-analysis. Fertil Steril. 2012;98:368–77.e1. 29. Qiao J, Zhang L, Yan L, Zhi X, Yan J. Female Fertility: Is it Safe to "Freeze?". Chin Med J (Engl). 2015;128(3):390. 30. Wei D, Sun Y, Liu J, Liang X, Zhu Y et al. Live birth after fresh versus frozen single blastocyst transfer (Frefro-blastocyst): study protocol for a randomized controlled trial. Trials 2017; 18(253): 1-7. 31. Zhang L, Yan LY, Zhi X, Yan J, Qiao J. Female Fertility: Is it Safe to “Freeze?” Chin Med J. 2015;128 (3):390-7. Authors: Paula Brígido, Roberto de la Fuente and Javier Del Río Assisted reproduction technology (ART) can help fertile couples to achieve successful pregnancies. Sometimes, reproductive desires of these couples are affected by the presence of a genetic disease in either partner. In such cases, couples are at a reproductive risk and find themselves in the need of assistance that only ART can provide. Preimplantation genetic diagnosis (PGD) provides an alternative to prenatal diagnosis to detect the specific genetic condition or disease they suffer from, and allows them to avoid passing it on their offspring (2). It requires the analyses of the embryos generated by ART in the in vitro fertilization (IVF) laboratory, by means of accurate and sensitive methodologies such as embryo biopsy, genetics, single cell genomics and, of course, background on prenatal diagnosis and counselling from experts. Clinical application of PGD dates back to the late 60’s, when blastocysts of research animals could be sexed (3) (note that this was already possible ten years before Louis Brown, the first IVF baby, was born in the UK in 1978). At the beginning of the 90’s, early human embryos were sexed before implantation and the first genetic analyses were performed to avoid children inheriting Mendelian diseases. By the end of the century, other nowadays considered basic genetic methodologies were routinely used for preimplantation diagnosis and PGD was applied as a normal procedure to guarantee healthy babies (4). In the present post we aim to give an account of the importance of PGD and the current view of the main clinical approaches for its application. WHEN IS PGD INDICATED? Indications for PGD are multiple and emerge from different motivations. Firstly, the patient may have suffered from a number of terminations due to the embryo having inherited the genetic condition. It could also be motivated by the parents already having a child with a severe genetic disease. In this case they might be willing to avoid passing it on the next one or even looking for a suitable treatment, if possible. However, one of the parents (or both) may be worried about their family history, being aware of the presence of a specific genetic condition, regardless of the type of inheritance. If the parents are carriers of any genetic disease, either an autosomal-dominant disorder like Huntington disease or an autosomal-recessive one like cystic fibrosis, they are at reproductive risk because the resulting embryo may be affected (the probability depending on the specific disorder itself and the way it is inherited) (see [2] for details on inherited conditions). But there are even cases in which motivation is not based on biological but in ethical or religious reasons. Certain families might have serious concerns about going on for abortion of an affected embryo. In such cases, application of PGD may circumvent this kind of ethical conflicts. Applying PGD Broadly speaking, steps for PGD are as follows (2):
PGD vs. PGS Preimplantation genetic screening (PGS) is the general term for a compound of approaches that aim to evaluate the genetic content of the cell, in contrast to genetic tests whose goals are to determine whether an embryo is affected by a specific genetic condition (PGD). Originally termed PGD-AS (preimplantation genetic diagnosis for aneuploidy screening), PGS was developed to confirm the ploidy status of the embryo, searching for possible aneuploidies. Available data suggest that most of miscarriages occurred during the first trimester are a consequence of some sort of aneuploidies (5), and that mainly selected chromosomes were involved in these structural abnormalities (6). Thus, the main approach developed for PGS was the fluorescence in situ hybridization (FISH) for such chromosomes. Types of approaches for PGD in the laboratory Current technical methodologies for preimplantational genetic analyses mainly lie in one of the following:
WHEN TO PERFORM BIOPSY Typical biopsies for PGD (and PGS) are as described as follows:
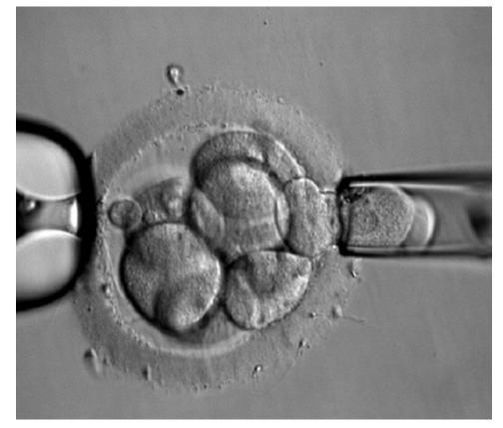
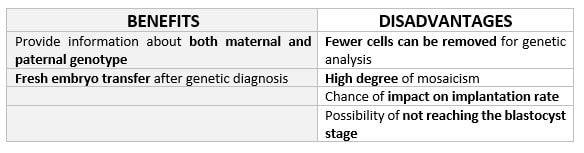
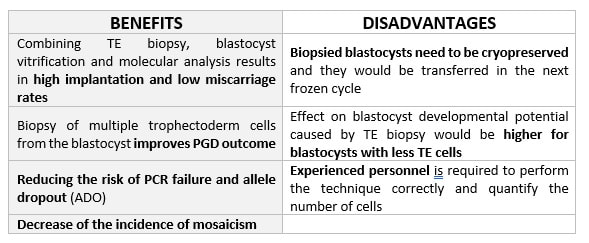
DAY 3. CLEAVAGE STAGE BIOPSY There is a controversy regarding utility of this type of biopsy. In the cleavage stage biopsy, embryos are biopsied at day 3 when individual cells can be differentiated. This technique entails aspiration of one to two blastomeres to obtain the embryonic genetic material for PGD analysis (13). Following genetic diagnosis, embryo transfer may be performed on blastocyst stage. Embryos are usually selected for biopsy based on morphological criteria. Unfortunately, these do not predict the development potential of the embryo, and so it could fail to progress until blastocyst stage. This would compromise the advantages of using the day-3 approach (14). On the other hand, performing biopsy on the cleavage stage allows embryos to be cultured in vitro until they reach the blastocyst. This means they can be fresh transferred (15), whereas embryos biopsied on day 5 must be vitrified and transferred in a subsequent cycle. How many cells should be removed? The number of cells to be removed in the biopsy is still a controversial issue. Aspirating one cell reduces the cellular mass extracted but it can imply the presence of mosaicism. Conversely, aspirating two cells can reduce the risk of mosaicism, but removing such cellular mass could have consequences on the implantation rate (14). Reported data have shown a dramatic reduction of 39% in the implantation rate in cleavage stage biopsy (16). The authors related it with proportion of the embryo total cellular removed. Whereas around five cells pulled out of the embryo in the trophectoderm biopsy represent 2-3% of the total cell content (expanded blastocyst has 200-220 cells approximately), extraction of a single cell from an eight cell embryo supposes 13% of the total content (16). What do experts say? Cleavage stage biopsy produces different opinions among embryologists because of the presence of mosaicism and the possibility of self-correction of aneuploidies from cleavage to blastocyst stage (17). On the contrary, studies using array-comparative genomic hybridization (array-CGH) technology to analyse genetic abnormalities in day-3 blastomeres and confirming it in trophectoderm biopsy showed concordance between day 3 diagnosis and day 5 reanalysis; Treff and coauthors showed more reliable results for SNP-microarray (96% vs. 83%) and also a lower mosaicism degree (31%) for SNP-microarray samples in a study comparing array technology versus FISH technique (18). These data would support the suggestion of some authors, who proposed that the incidence of mosaicism may have been overestimated in previous studies due to technical inconsistency of the FISH technique (17, 18, 19). At present, this matter remains controversial. Regarding pregnancy rates, in both types of biopsies higher pregnancy rates are obtained comparing with the control group, in which no biopsy was performed (14, 19). To sum up: DAY 5. TROPHECTODERM BIOPSY The blastocyst stage is currently supposed to be an optimal time to perform biopsies for PGD/PGS. The combination of improved blastocyst culture, trophectoderm (TE) biopsy, refined cryopreservation techniques, and molecular assays, such as array comparative genomic hybridization that allows for 24-chromosome screening, have led to a renaissance of PGS. TE biopsy will not detect every circumstance in which the embryo is at risk of aneuploidy, but it will detect mosaicism more reliably than cleavage-stage biopsy (which cannot be relied on at all for this purpose) (20, 21). Moreover, when diagnosing monogenic disorders in single blastomere cells using PCR-based protocols, there is a high risk of PCR failure due to either no amplification (allele dropout) or preferential amplification of one of the alleles, potentially resulting in a reduced number of unaffected embryos available for transfer. Increasing the amount of starting DNA template should in principle increase the sensitivity and reliability of genetic diagnosis. Therefore, the biopsy of multiple trophectoderm cells from the blastocyst rather than a single cell from cleavage stage embryos should potentially lead to improved PGD outcome for patients (14). How many cells should be removed? Research to determine the appropriate number of biopsied TE cells in blastocyst biopsies are limited. The exact number of biopsied TE cells is hard to count visually because cells are small and usually remain as a clump. In most studies using comparative genome hybridization or single-nucleotide polymorphism array technology for genetic testing, biopsied TE cells were used for genome amplification and their number was impossible to know. Moreover, some studies showed that removing four to five cells leads to better results. Therefore, the biopsied cell number should be higher in the blastocysts with better TE quality than those with worse characteristics (22, 23). Can biopsies affect blastocyst development and its implantation? Whereas it remains possible that biopsy of cleavage-stage embryos can critically arrest further development through reduction of cell mass, the low miscarriage rates and high term birth rates in the present series, as well as data presently under analysis, suggest that this is not the case for TE biopsy. It can be speculated that the damage to blastocyst development potential caused by TE biopsy would be less for blastocysts with a greater number of TE cells (21, 22). Some experts assured that TE biopsy at the blastocyst stage had no meaningful impact on the developmental competence of the embryo as measured by implantation and delivery rates. This contrasts with the information above-mentioned on the significant reduction in the probability for an embryo to implant and progress up to delivery (16). When combined with TE biopsy and blastocyst vitrification, SNP microarray has resulted in high implantation and low miscarriage rates for some IVF patients (15, 16, 24). Are there any limitations? Owing to the limitations of genetic analysis, most of the biopsied blastocysts need to be cryopreserved by vitrification, and blastocysts with normal results would be transferred in the next frozen cycle. In addition, biopsy of numerous cells from blastocysts with grade B or C may cause damage to the embryo, leading to either its arrest or implantation failure. However, 1-5 cells may be the appropriate biopsied TE cell number to maintain the implantation potential (15, 22). Also, the personnel experience of different embryologists is an influencing factor in this technique. The number of biopsied cells in the blastocyst biopsy is hard to quantify and largely dependent on the experience of embryologist (22). To sum up: WHAT CAN WE CONCLUDE? The availability of new embryology and molecular techniques allow preimplantation genetic diagnosis laboratories to offer patients at genetic risk the transfer of developmentally competent embryos, unaffected by genetic disease. Cleavage stage biopsy allows for fresh embryo transfer after genetic diagnosis. However, there are reports of high levels of mosaicism when the biopsy is performed on day 3. Trophectoderm biopsy, in turn, provides sufficient material for an effective and more reliable diagnosis in embryos compared to those on cleavage stage. Moreover, it seems that it does not compromise embryo implantation and pregnancy rates in PGD cycles. The drawback for this option is the usual need for cryopreservation and transfer in a different cycle. The offer of PGD in fertility centres has increased over the last decade, primarily due to the progress on the application of diagnostic methods. The choice for either development stage relates to successful outcomes in the clinic, which mainly depend on technical challenges and timing of the developing embryo. For the embryologists, both day-3 and day-5 approaches are supported by evidence, but it will be essential to consider every single aspect of them to evaluate the best option for the laboratory. REFERENCES:
|
Entries
March 2019
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media