|
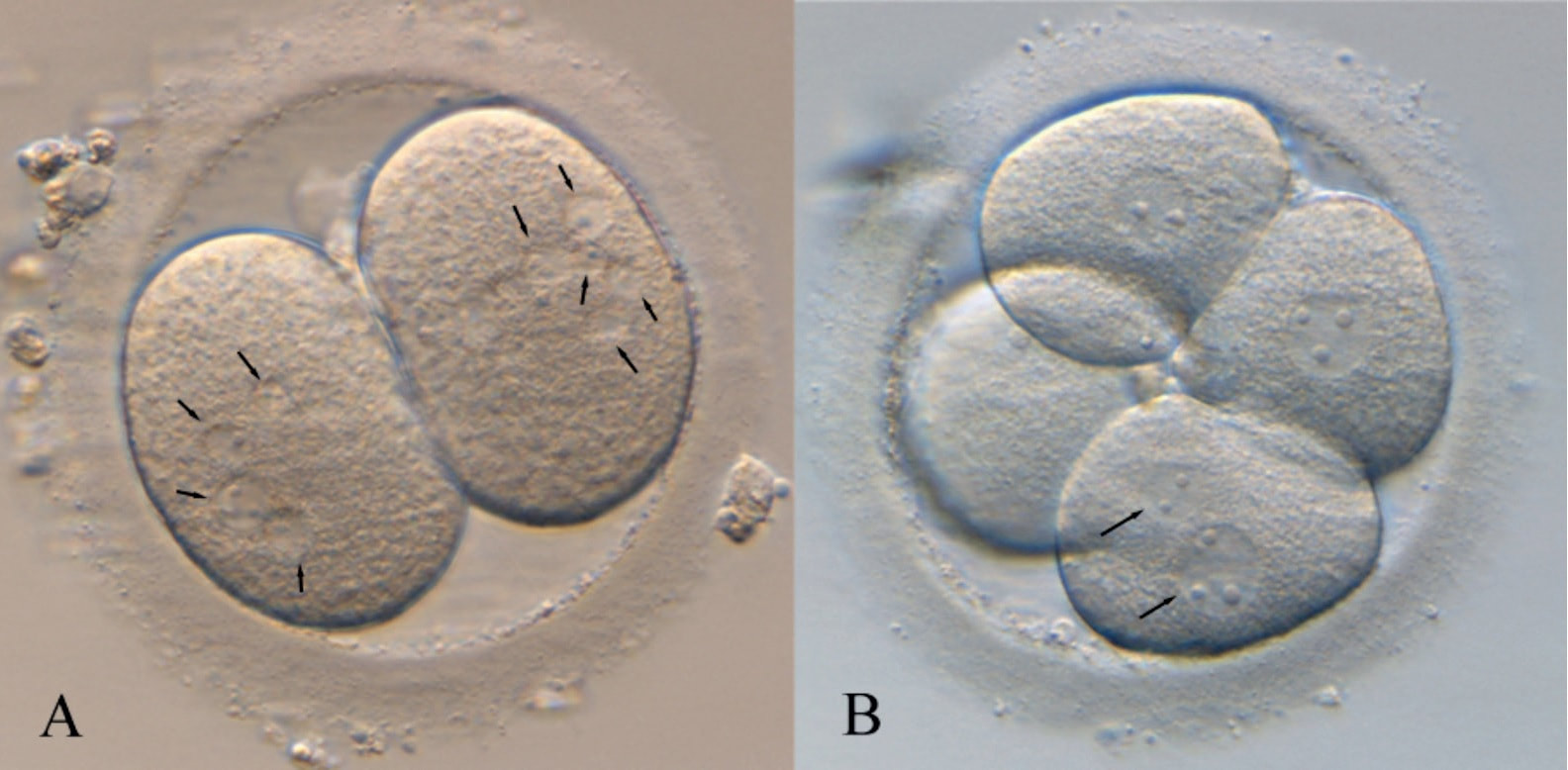
Authors: Iris Martínez Rodero and Raquel Pillado González "Multinucleated blastomeres (MNBs) present in embryos are morphological abnormalities of unclear origin, which have been extensively correlated with chromosomal defects, lower blastocyst formation and implantation rates". INTRODUCTION Selection of high-quality embryos is an important factor for the successful outcome of assisted reproduction technologies (ART). Nowadays, criteria for selection are mainly based on morphological features such as embryo fragmentation, cell number, blastomeres uniformity, etc. (2). The parameters studied so far have been demonstrated to be useful indicators of embryo quality. Their evaluation is performed through non-invasive light microscopy-based analyses, usually carried out once a day at specific time points. This approach intends to minimise the events of taking the embryos out of the incubators and exposing them to undesired harmful conditions. The presence of multinucleated blastomeres (MNBs) can be regarded as one of those indicators, and even though previous studies had already connected it to DNA abnormalities and low pregnancy rates as early as in the 90s (3,4,5) the origin of this phenomenon still remains unclear. Several possible factors seem to influence its genesis, but the specific cause for its occurrence is yet to be determined (6). Since the introduction of time-lapse imaging and monitoring technology, IVF laboratories have been able to carry out more exhaustive and continuous observations on embryo development, keeping risks at a minimum (7,8). By identifying the precise timing of specific key events of blastomere cell cycle interludes and of the embryo´s overall growth it is possible to assess its quality (7). Furthermore, time-lapse imaging and monitoring systems have facilitated the study of multinucleation (MN) in relation to its incidence in time and in the population, as well as its correlation with other morphological features and clinical variables (6,7,9). In the IVF context multinucleation is defined as the presence of two or more nuclei in one or more blastomeres. Multiples studies even differentiate between binucleated and multi-/micro-nucleated (three or more nuclei) blastomeres (Fig 1) (1,4,7). Reports on this phenomenon range from 17% to 69% of the total of cultured embryos, depending on the groups used within assays and authors (10). Factors that may influence MNB appearance are numerous and have been repeatedly studied (6,7,9,11). To date, several explanations have been proposed for MNBs: dysfunction of the mitotic spindle or the occurrence of karyokinesis without cytokinesis (12); DNA breaks or imperfect mitosis (13); nuclear membrane alterations (14) and even other factors not directly responsible for MNBs and yet linked to its presence (1). FACTORS THAT MAY RELATE TO THE APPEARANCE OF MULTINUCLEATION
Results from various studies have shown a difference in the percentage of multinucleated embryos between groups that had been fertilised by traditional IVF vs ICSI. Van Royen and colleagues showed 32.7% of MN embryo in the IVF group, compared to 34.5% in the ICSI group (6). Accordingly, Walmsley et al. 2003 reported 17,2% vs 18,3% of MN in embryos derived from IVF and ICSI, respectively (11).
Apparently, the type of infertility factor seems not to affect MN rates. Some studies have reported no significant differences in the percentages of MN embryos between cases of female factor-only infertility and male factor-only infertility (32.7% and 34.7%, respectively). In addition, differences were not found between these cases and those with both partners affected by some sort of infertility, either (6). However, different studies support the relevance of oocyte culture, specially regarding certain processes that occur naturally in in vivo conditions and that are essential for the proper embryo development. Data show that both oocytes subjected to negative conditions when cultured in vivo (like stern hypoxia, for instance) and oocytes cultured in vitro derive in a higher percentage of MN embryos (15,16). Regarding male infertility, results are more controversial; whereas some studies reflect a higher MN rate for cases in which male factor is especially severe (normally derived for ICSI rather than IVF) (11), other authors show no significant differences (6).
Records exhibit that cycles with an accelerated ovulation induction response present increased MN rates (6,17). Furthermore, several studies have reported that embryos derived from patients from whom ten or more oocytes had been collected presented a significantly higher MN rate than embryos from groups of nine or fewer oocytes (6,8,18). This is in accordance with the fact that patients who need high doses of FSH present higher MN rates (6). Very short cycles and cases where high doses of GnRH are needed trigger the development of high numbers of premature follicles that produce oocytes, which despite being able to reach metaphase II and become fertilized fail to go through proper nuclear cleavage (6,18). Lastly, even though differences in GnRH doses have been associated to significant differences in the incidence of MN embryos, similar results have not been observed when using different hormones (like rFSH, r-hFSH, purified urinary FSH or urinary gonadotropin, for instance) (6).
Multiple studies have been conducted on patients ranging from 25 to 45 years old. Several authors divided data in 5-year interval groups in order to verify whether patient age correlates with MN. However, the only significant difference was found when comparing women of +40 with younger ones of -35, presenting higher degree of MN in the first group (6,9).
Chromosome polymorphisms consist in heterochromatin variability. These are usually located in the long arms of chromosomes 1, 9 and 16, and the short arms of chromosomes from the groups D and G (13, 15 and 21, 22 and Y) (19). Even though such polymorphisms are generally regarded as normal within karyotypes (20), studies indicate that some of them might be associated with certain clinical problems such as abnormal spermatogenesis (21), infertility (22,23), recurrent miscarriages (24,25) and higher rate of chromosome abnormalities among blastomeres at the cleavage stage (26,27). Sun and collaborators hypothesized that couples with chromosome polymorphisms might experience a higher rate of embryo multinucleation (19). Nevertheless, the authors found no association between chromosome polymorphisms and MN embryo formation in couples undergoing IVF (19).
- Cellular fragmentation Although MN may appear regardless of the cellular fragmentation levels, several papers support the correlation between these two features (6,28,29). In particular, Van Royen et al. divided the level of fragmentation in three categories: F1 (≤10%), F2 (10-20%) and F3 (20-30%); this study presented evidence for higher MN in F2 and F3 when compared to F1, but similar to each other (6). - Cleavage rate When 3-cell and 5‐cell day-2 embryos were observed under the microscope, both types exhibited significantly higher multinucleation (28.2% -50%) than regular ones with the ideal 4-cell cleavage pattern (with only 16.8% MN). Similarly, day-3 embryos with the typical 8‐cell stage showed significantly lower multinucleation (15.5%) than 7‐cell and 9‐cell embryos (6). As it has been mentioned, ideal 4-cell and 8-cell stages show similar MN percentages. However, application of time-lapse imaging has revealed a significant decrease in MN from the 2-cell to the 4-cell stage (from 43.2% to 15.0%). The analysis of MN in 2-cell embryos indicated that, after cleavage, the majority (52%) of 2-cell MN embryos became mononucleated, whereas only a lower percentage (34%) showed MNBs, and about 14% were of poor quality (with only one or no visible nucleus at all) (9). This decrease in the MN rate suggests that 2-cell MN embryos are able to self-correct their nuclear abnormalities. But this repair mechanism has been observed in both euploid and aneuploid embryos, therefore it cannot be used as an indicator of chromosomal normality during embryo selection (8,9,30). An extended duration of both 2-cell and 4-cell stages has been proposed as a possible indicator of the occurrence of nuclear self-correction (9). INCIDENCE OF MULTINUCLEATION IN CLINICAL IVF As previously exposed, it is through time-lapse imaging that a far higher percentage of multinucleation (25%) has been detected compared to static observations on day 2 at 42 hours post-insemination (hpi) (<5%) (7). These observations have demonstrated that multinucleation is a frequent event that, according to Yilmaz et al., is present in at least one embryo in 41.3% of IVF cycles (31). Data provided by Desai and colleagues reported that approximately 56% of binucleated embryos and 48% of those with three or more nuclei went on to form blastocysts that met the appropriate criteria for vitrification (7). In addition, data from different studies point to binucleation being more frequent than blastomeres with 3 or more nuclei (7,31,32). At the same time, multinucleation has provided an additional criterion for embryo selection, since it is mainly observed in those of poor quality and is associated with direct and/or reverse cleavage (7). It has been observed that, out of all embryos found showing direct and/or reverse cleavage, at least one fourth were also multinucleated (7). By using time-lapse, multinucleation has been repeatedly observed to be a reversible event in a high proportion of embryos (7,32). Multinucleation reversibility has been reported to be as high as 73.4% (32); this has been calculated as the proportion of embryos in which multinucleation was detected at 2-cell stage, but not visible at 4-cell stage (likely due to self-correction mechanisms, as above-mentioned). In fact, Aguilar and collaborators reported 127 multinucleated embryos at 4-cell stage out of the 479 ones initially observed to present this feature at 2-cell stage. De novo multinucleation at the 4-cell stage in turn was observed in 36 embryos (32). IMPACT OF MULTINUCLEATION ON IVF OUTCOMES Multinucleation has traditionally been related to both low blastocyst formation (33) and implantation rates (5,6,17,28,34), and linked to the likely presence of chromosome abnormalities, which consequently results in embryo arrest (35). Nevertheless and despite all the existing evidences, there is still much controversy regarding multinucleation; reports have been published revealing cases in which fully binucleated 4-cell stage embryos had eventually developed into euploid blastocysts and genetically normal children (31,34).
Although some preimplantation genetic testing (PGT) studies have shown that not all multinucleated embryos are chromosomally abnormal (31,32,36) multinucleation is predominantly associated to chromosomal defects and poor implantation prognosis (3,31,37). Kligman and colleagues published that 74.5% of multinucleated embryos were chromosomally abnormal, compared to 32.3% of non-multinucleated embryos (3). Years later, Ambroggio et al. revealed an increased incidence of aneuploidy of MN 4-cell stage embryos when compared to single-nucleated embryos (85% vs 78%), suggesting that multinucleated embryos should not be recommended for transfer in IVF cycles (37). These results were confirmed when, from 395 MN embryos tested for PGT, Yilmaz et al. reported that 82.5% of MN blastomeres exhibited two nuclei, whereas the remaining blastomeres presented a single or three or more nuclei (31). Noteworthy, binucleated patterns of multinucleation may be less detrimental, since a high percentage of embryos with such feature are euploid, compared to embryos exhibiting three or more nuclei in a single blastomere (38).
Embryo morphokinetics was studied and related to the multinucleation status in a study conducted by Meseguer’s team (32). In the study, 53.4% of a total 1676 embryos included were MN. Based upon the reported data, differences in morphokinetics between multinucleated and non-multinucleated embryos at both 2-cell and 4-cell stages comprise cleavage events involving the completion of the first mitosis and the length of the S-phase. These differences affected the following parameters: t2, t3, second cell cycle (cc2=t3-t2), t4, t6, t7 and t8. These results allowed to conclude that, if multinucleation remains at 4-cell stage, it takes longer for the embryos to complete the next cell cycle (cc3=t5-t3). Should this be true, the restoration system would not be efficient if MNBs were still observed after the 2-cell stage (32). The origin of the multinucleation phenotype has been suggested to be multiple: disruption of intracellular restructuring, remodelling or imprinting in the developing oocyte, or even alterations in DNA replication, cytokinesis or compaction during the first cell cycle (16). If multinucleation appears as a result of defects in cell function, differences in morphokinetics between MN and non-MN embryos during these early stages may be expected (32).
Opinions on the impact of the multinucleation phenotype on implantation rates diverge from each other: On one hand, cell stage for MN appearance has been proposed to exert the highest effect on the implantation rate. Authors supporting this claim are divided into two positions: those who affirm that the presence of MN at the 2-cell stage is actually insignificant in terms of differences on implantation rates, but it is at 4-cell stage when it does have a measurable negative effect (32); and the authors who argue implantation rates to be significantly reduced when MN is already observed at the 2-cell stage (8). On the other hand, the school of Meriano and coauthors affirm that binucleation is less harmful than any other type of multinucleation (16). However, Aguilar and colleagues explained that their differences with Meriano were found on the frequency of image acquisition and the systems used to measure multinucleation (32); whereas the former acquired one picture in seven different focal planes every 20 minutes, the latter recorded images every 2.5 minutes (32). In any case, it has been demonstrated that patterns of multinucleation at 4-cell stage are correlated with low implantation rates, while any of the other cases has been reported to decrease the chances to achieve pregnancy (16,32). CONCLUSIONS Multinucleation is a common and reversible event observed in human IVF embryos, and it is specially frequent as binucleation at two-cell stage. It is associated with chromosomal defects and altered morphokinetic parameters, eventhough binucleation patterns seem to be the less severe. Regarding multinucleation impact on implantation rate, results are controverted. It seems that implantation rates are not affected when multinucleation appears as two nuclei in two-cell stage. Although the presence of multinucleated blastomeres in human embryos has been associated with the above-mentioned undesired characteristics in IVF embryos, the reasons explaining its appearance and occurrence in time and its relationship with patient specifications have not been deeply studied until time-lapse systems became available. Even though different causes have been suspected to lie behind MNB development, none of them have been proved actually represent the main responsible. Nevertheless, a growing number of studies provide data untangling the relationship between MN and assisted reproduction fertilization methods (IVF and ICSI), stimulation cycles, infertility factors, culture conditions and other embryonic morphological characteristics. Even though sometimes results from different studies may seem contradictory, this might be accredited to the differences in sample sizes. All the above said, it seems reasonable to highlight the need for further research on this issue. It would be highly helpful to unveil the actual triggers of multinucleation, to develop optimal ART practices that avoid increasing MN incidence, and to unravel any other correlation with adverse embryo features during development. Deeper knowledge would help improving embryo assessment methods and, consequently, increase the rates of successful ART outcomes. REFERENCES
0 Comments
|
Entries
March 2019
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media