|
Authors: Inés Abad, Roberto de la Fuente and Sara Sanz INTRODUCTION Fertility treatments are more and more common in our days, reason why it is important to perform these procedures accurately resembling in vivo conditions. Additionally, male factor may oftentimes be underrated, and yet it is 50% of the treatment. The following text aims to establish an updated comparison between in vivo and in vitro semen preparation methods. In the first part a general description of the processes of maturation and capacitation of sperm are presented. MATURATION Where does sperm maturation take place? Once spermatogenesis is completed in the seminiferous epithelium, immature spermatozoa migrate towards the epididymis, the organ in which sperm maturation and storage take place. The epididymis is usually divided into three different parts: caput (head), corpus (body) and cauda (tail) (2). Typical changes in sperm during maturation 1. Acquisition of progressive motility. Even though immature sperm have functional movement machinery, motility of these cells begins in the caput segment. Whereas beating intensity is similar throughout the whole epididymis, flagellar amplitude is modified within this path. This is due to changes on the sperm surface, such as acquisition of new proteins and molecular changes involving inactivation of Ser/Thr phosphatases (3, 4). 2. Migration of the cytoplasmic droplet (CD). This droplet migrates from the neck towards the annulus of the mammalian spermatozoa (in humans, the CD is more proximal, located at the neck as opposed to the distal position of the annulus). The role of this droplet is to regulate ion homeostasis. It contains K+, Cl- and water channels, which have been suggested to work in regulation of sperm volume during the different regions of the epididymis. It also accumulates Ca2+, which has a biphasic role controlling phosphorylation pathways in sperm cells. In immature spermatozoa, it has been hypothesized that high Ca2+ levels found in the CD maintain low levels of tyrosine phosphorylation (5). 3. Changes in sperm protein and lipid profile. - Protein and lipid content Even though changes in these profiles are not well understood, there are three complementary mechanisms that participate in completing maturation:
Perhaps the most important among these changes is the significant reduction in cholesterol content of the sperm membrane. This reduction involves a decrease in the cholesterol/phospholipid ratio that facilitates protein trafficking from and onto the membrane and enhances its fluidity, which will eventually play a role in triggering capacitation and fertilisation (8). - Post-translational protein changes Additionally, certain post-translational modifications of proteins have also been hypothesised to occur during sperm maturation. This is the case for oxidation of thiol groups, which promotes the formation of disulphide bonds (S-S) and stabilises components of both the head and flagellum (9). 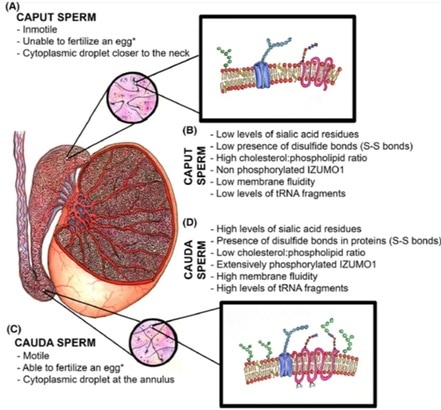 Fig.2. Schematic representation of the main items during sperm maturation. (A) Principal functional and morphological aspects in immature caput spermatozoa. (B) Molecular characteristics of immature spermatozoa. (C) Main morphofunctional traits of cauda mature spermatozoa and (D) their molecular features (4) (*) Ability to fertilize the egg will ultimately depend on completing capacitation. CAPACITATION What is the composition of the ejaculate? In mammals, semen is composed of two different phases:
Following ejaculation, semen is deposited to the anterior wall of the vagina, adjacent to the ectocervical tissues. From here on, for sperm to progress towards the egg through the oviduct or Fallopian tubes, semen must undergo liquefaction. This process usually takes about 20-30 minutes (11). Semen goes through the cervix and reaches the distal portion of the Fallopian tube, where sperm is stored and maintained by interacting with the endosalpingeal epithelium (12, 13). In 1951, Austin and Chang individually observed that a certain period of time in the female tract was required for sperm before fertilization could take place (14, 15). Later, observations in multiple mammalian species confirmed these first notions, and certain studies showed a delay of at least 2 hours before sperm entry into the egg. This supports the hypothesis of sperm maturation before becoming fertile after ejaculation (16). Nowadays, such process is known as capacitation, as opposed to maturation in the male tract explained above. Capacitation and fertility Some of the factors involved in sperm capacitation are steroid hormones such as oestrogens and progesterone, both produced by the follicle. These steroids play different roles: they act as chemoattractants, facilitate triggering of hyperactivation, regulate trafficking of cGMP or modulate the potential for completing acrosome reaction (17-19). Semen liquefaction following ejaculation is mainly modulated by prostate derived peptidase KLK3. In females, KLKs 5–8, 10–11, and 13–15 are expressed at very high levels in the cervix and vagina compared to other adult tissues (20, 21). Moreover, KLK1 and KLK3 transcripts are expressed at the highest level in human endometrium when circulating estradiol (E2) is elevated. These findings suggest that KLKs are expressed in the human reproductive tracts and that some of the KLKs in the uteri are regulated by E2. Abnormal E2 signalling in the female reproductive tract leads to semen liquefaction defects, associated with defective SEMG cleavage and sperm transport, which may result in some cases of infertility. It is known that mice lacking ESR1 (one of the oestrogen receptors) in the epithelial cells are infertile (22), partly due to a reduction in the number of sperm able to reach the oviduct (23). However, the effect of ESR1 loss in the epithelial cells on sperm transport in the uterus has not yet been investigated. Similarly, other potential research lines could investigate liquefaction defects caused by diminished KLK activity in females or regulation of KLKs by molecular signalling in the female tract. Once semen trespasses the cervix, sperm are known to achieve capacitation in an asynchronous fashion during the interaction with the epithelium, which results in a continuous flow of fertile spermatozoa towards the Graafian follicle (24). The ability to bind to the epithelium, in turn, may be indirectly related to the sperm DNA integrity, and so DNA fragmentation levels would be indicative of the fertility potential of the sperm (25). Elements involved in sperm capacitation Even though capacitation had traditionally been regarded as a two-step process, through which changes in the cell membrane would lead to the acrosome reaction (AR) (26), capacitation is currently considered as a continuous process that culminates in the AR. It would be difficult to describe all capacitation-related events separately because all of them are connected to each other in time. However, the most important changes in the sperm during the process are (27-37):
Sperm capacitation is a complex process with multitude of interconnected and highly regulated molecular pathways. One of the first events is the alteration of the permeabilization of the sperm plasma, so that the influx and intracellular concentration of certain ions are increased. The main molecules involved are probably Ca2+ and HCO3-; the net intake by the sperm cell triggers alkalinisation of the pH and the concomitant activation of the soluble adenylyl cyclase (sAC) (38, 39). As an immediate consequence, cAMP levels increase followed by activation of the protein kinase A (PKA) (40, 41). The rise of cAMP causes redistribution of certain phospholipids and proteins of the membrane, and so exposing cholesterol, which accumulates in lipid rafts (42). The organization of these rafts promotes the removal of cholesterol and its translocation to extracellular acceptors like albumin (43). Also, increased cAMP activates PKA, which in turn activates SRC kinase (44). Eventually, SRC kinase activity triggers tyrosine phosphorylation, which results and a wide range of proteins been modified and relocated in capacitating sperm. This has been described in several species, including humans (45). The end result of capacitation is the acrosome reaction (AR), the process by which the content of the acrosome is released to the extracellular environment. In natural conditions, this environment is actually the cummulus cells, whose connections will be broken by the chemical reactions of the acrosomal content, mainly proteases like acrosin and hyaluronidase, also exposed to the right membrane domains during lipid redistribution (46). It is not surprising that mutations affecting any of these processes will result in multiple causes for infertility (47). FUTURE PERSPECTIVES Findings like the one regarding post-ejaculated liquefaction, mutations on acrosome protease-encoding genes or other molecular mechanisms of sperm capacitation are crucial to progress in the field of reproductive medicine, and can lead to: (i) potential diagnostic tools for unexplained infertility cases, (ii) the development of a novel contraception technology to entrap sperm (48), (iii) or even revolutionary new methods for human sperm capacitation in the laboratory (49), which could significantly improve live birth rates for fertility treatments. In the following post... different methods for sperm selection in the laboratory will be explained, paying attention to advantages and disadvantages under different circumstances. The importance of different sperm features like DNA fragmentation or morphology will be discussed in relation to the best sperm selection method to achieve optimal clinical outcomes. REFERENCES:
2 Comments
|
Entries
March 2019
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media

