|
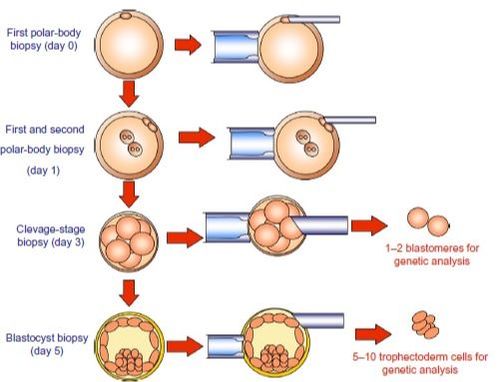
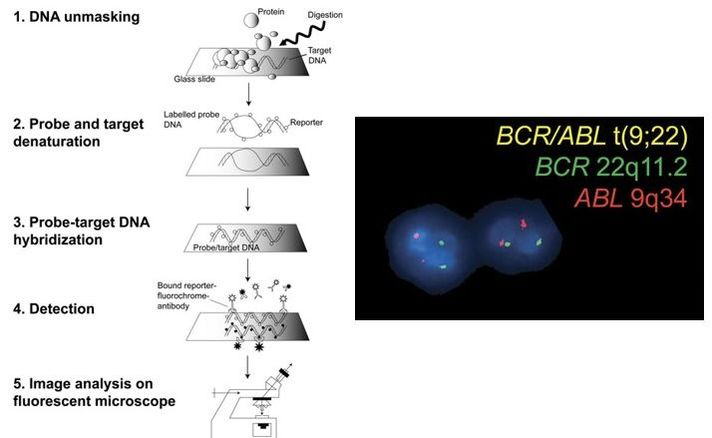
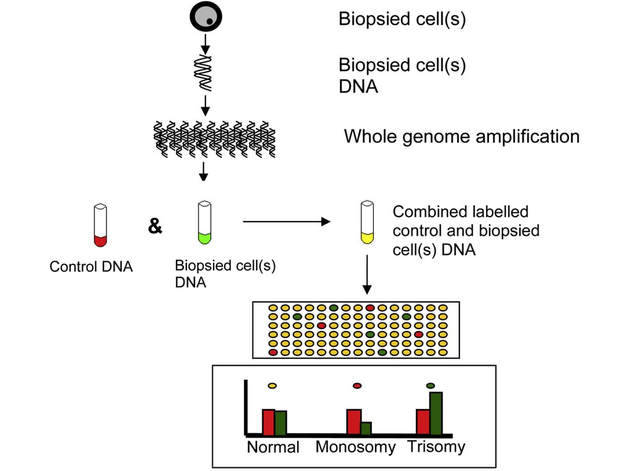
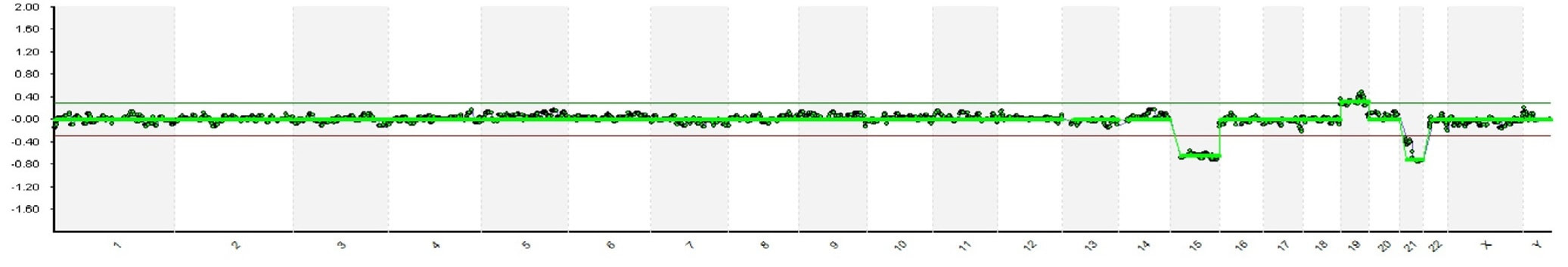
Authors: Javier Del Río and Sara Sanz Genetic problems in the embryo are one of the most important causes of pregnancy loss and miscarriage. However, identifying embryo mosaicism as the cause of genetic problems during development is not an easy task. WHAT IS A MOSAIC EMBRYO? The term mosaicism refers to the presence of more than just one cell line, which present different chromosome count (1). Additionally, the most common situation in these cases is the presence of a mixture of distinct aneuploid cells, rather than of a variety between euploid and aneuploid cells. These are the embryos that may be at risk of misdiagnosis (2). There are four possible types of mosaic embryos (3,4): 1. Embryos with a mix of euploid and aneuploid cells in the trophectoderm (TE), and with aneuploid cells in the inner cell mass (ICM). 2. Embryos with a mix of euploid and aneuploid TE cells and euploid ICM. 3. Embryos with euploid TE cells and aneuploid ICM. 4. Embryos with aneuploid TE cells and euploid ICM. Even though there exists no specific cut-off to determine mosaicism, the Preimplantational Genetic Diagnosis International Society (PGDIS) suggests an embryo with more than 20% of aneuploid cells to be considered as mosaic. This means lower levels of mosaicism should be treated as normal (euploid) (5). It has been traditionally thought that only genetic problems in the oocyte or the sperm could be responsible for embryo mosaicism. Nevertheless, it is currently postulated that this also occurs during the first mitotic divisions, when maternal transcripts control the cell cycle of the early embryo (6). NORMAL INDICATIONS FOR CHROMOSOMAL TESTING TO DETECT EMBRYO MOSAICISM 1. Advanced maternal age. This is the most common cause for aneuploid problems (7). A recent study has shown that women between the ages of 35 and 43 years have more probabilities (an increase ranging from 28 to 78%) of presenting mis-segregation for the most clinically relevant aneuploidies, namely chromosomes 13,16,18,21 and 22 (8). 2. Severe male factor infertility. Even though levels of sperm aneuploidy are associated with increased levels of chromosomal abnormalities in embryos (9), such abnormalities could also arise from certain males who do not present any chromosomal abnormality a priori. Such could be specific cases of oligoastenozoospermic patients (10). 3. Recurrent implantation failure (RIF). In spite of the lack of specifications for such diagnosis, it is usually defined as the occurrence of three or more failed IVF attempts due to an unidentified cause. RIF is the usual diagnosis in those cases in which after a cumulative transfer of more than 10 good-quality embryos, the eventual result is IVF failure (7,11,12,13). 4. Recurrent miscarriage. The definition of this concept may vary for every country. However, generally speaking it can be defined as the occurrence of 3 or more consecutive miscarriages once pregnancy has reached at least 14 weeks (14). The main cause for this problem seems to be aneuploidy, which has been identified as the leading cause in a high percentage of miscarriages (15,16). 5. Previous trisomic pregnancy. Cases in which there has been a previous trisomic pregnancy entail higher probability of suffering from another aneuploid conception. Therefore, it is in this group of patients in which it would be beneficial to conduct a study to find out possible related causes (17). BIOPSY TECHNIQUES TO STUDY A CASE OF MOSAICISM Although there are different biopsy strategies, depending on the embryo stage, blastocyst biopsy is recommended over both polar body and blastomere biopsies in those cases in which mosaicism is suspected. Blastocyst biopsy is less invasive, it is possible to extract a higher number of cells, which increases the probability to confirm mosaicism, and it is cheaper than the other techniques due to the lower number of embryos required for biopsy (7). Furthermore, if cells with different chromosome complements are widely distributed throughout the trophectoderm, there might be a good chance of capturing a representative sample. If cells are clustered, mosaicism could not be easily detected, thus providing false normal results (18). TECHNIQUES EMPLOYED TO DIAGNOSE MOSAICISM The earliest trials of PGD involved the use of karyotyping and PCR. By the mid-90s, the use of cytogenetic techniques such as FISH allowed for the progress of preimplantation diagnosis. This very own approach was later shown to impose important technical limitations to the analysis, and so it was encouraged the development of new technologies that could minimize the errors in diagnosis (19). Fluorescence in situ hybridization (FISH) It allows for the analysis and identification of chromosomes or chromosome fragments with 5-10 fluorescently labelled molecular probes from one cell (blastomere) (Fig. 2). This cell can be biopsied from day-3 embryos, it could belong to the trophectoderm from blastocysts or it could also be a polar body biopsied from an oocyte or a zygote. Therefore, PGS–FISH diagnosis is limited to the most common abnormalities involving chromosomes 13, 15–18, 21, 22, X and Y. Some studies of preimplantation embryos diagnosed by using this technique estimate a 5-7% error caused by mosaicism when embryos are reanalyzed. Additionally, FISH is being rapidly replaced by other DNA analysis methods with higher efficiency (20,21). Array Comparative Genomic Hybridization (aCGH) This technique relies on whole genome amplification from one or more blastomeres (Fig. 3). It provides a quantitative analysis based on the comparison between the relative amount of tested DNA and the control DNA. Thus, chromosome imbalances such as aneuploidies, unbalanced translocations, deletions and duplications are easily detected. However, since balanced chromosome rearrangements such as reciprocal translocations or inversions do not affect copy number, such alterations cannot be identified (19,20). When blastomeres are analyzed by aCGH, the error rate measured is merely 2% (21,23). Notably, some studies have found that PGS-aCGH after blastocyst biopsy provides higher implantation and pregnancy rates than PGS-FISH (24). Next Generation Sequencing (NGS) Next Generation Sequencing belongs to the group of Massively Parallel Sequencing (MPS) methods that allow for parallel processing of an extremely large number of nucleic acid molecules (Fig. 4). As a result of sequencing on a microspace scale, it has been possible to drastically increase the amount of information collected during one test up to an entire human genome. Also, it is the only method that allows for analysis of all chromosomes (aneuploidies or translocations) and mutations responsible for any single-gene disease, just using one biopsy and in a single step (20). Although clinical results have documented high pregnancy rates following transfer of screened embryos, further data along with an extended use in clinical application are required to better define the role of NGS in PGS. Nevertheless, it seems that this method may actually lead to reduced costs per patient, thus allowing IVF couples a wider use of PGS for choosing the most competent embryo for transfer (26). DIFFICULTIES WHEN MANAGING RESULTS The detection of mosaicism at an early stage does not mean that it will spread along embryo development (27). However, the utilization of chromosome identification techniques as part of the IVF process makes it possible to identify embryos “at risk of mosaicism’’ in order to select those that are suitable for transfer (18). Mosaic embryos are supposedly less competent than others due to a reduced implantation potential. Therefore, by discarding mosaic embryos implantation rates should be improved and, simultaneously, embryo loss rates reduced. Nevertheless, mosaic embryos may still have reproductive potential, and consequently they could still be viable. Furthermore, discarding embryos capable of producing healthy children will decrease pregnancy rates in those patients who get a low number of blastocysts in the pool of transferable embryos (18). It is important to take into account the reaction of the patients when they are informed about their embryos being at risk for mosaicism, what may entail genetic abnormalities, reduce implantation rates, increase loss risk and even diminish obstetrical and neonatal outcomes. However, there is not a simple answer when patients decide to transfer a mosaic embryo; either way, the obstetrical team should be informed for future screens (18). SUGGESTED GUIDELINES BY THE PREIMPLANTATINO GENETIC DIAGNOSIS INTERNATIONAL SOCIETY (PGDIS)
This article has been selected for publication in the Scientists in Reproductive Technologies (SIRT) Newsletter of The Fertility Society of Australia: DEL RÍO, J. and SANZ, S. (2017) Mosaic embryos are capable of producing healthy children. How to handle it? Fertility Society of Australia - SIRT Newsletter 4(20): 12-15. REFERENCES
2 Comments
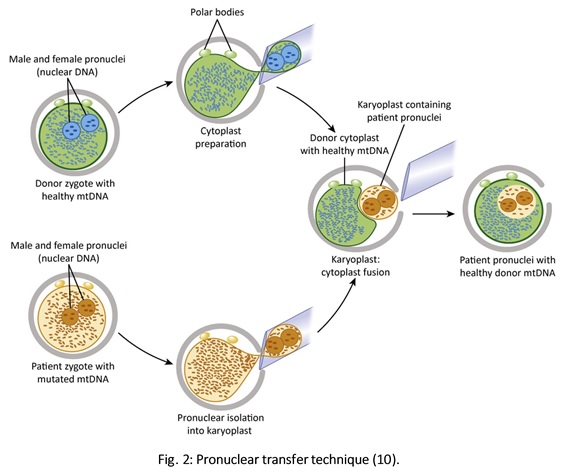
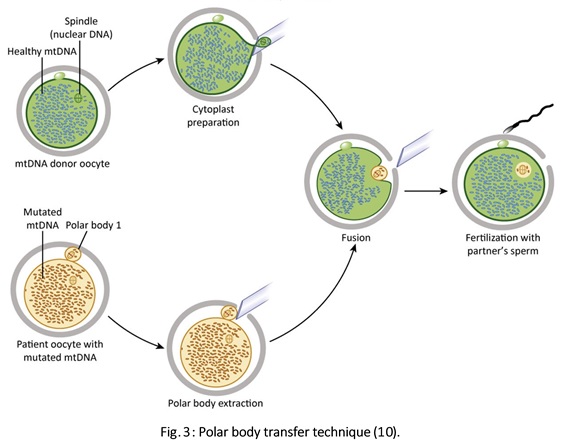
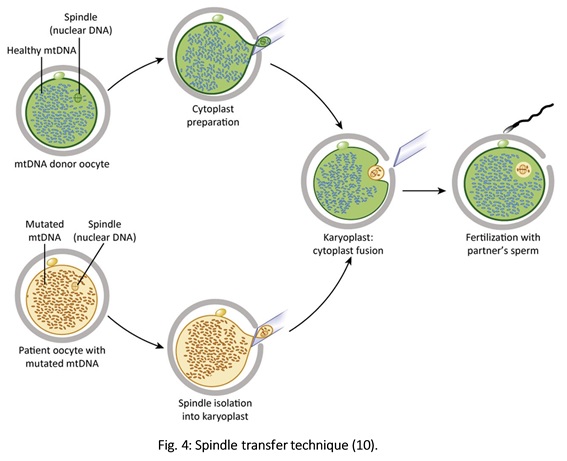
Authors: Javier Del Río and Sara Sanz Although most of the genetic material of eukaryotic cells is located inside the nucleus, mitochondria are organelles that also possess a certain amount of DNA. Mutations in mitochondrial DNA (mtDNA) or nuclear genes involved in mitochondrial function are cause of infertility and diseases, not only in individuals, but also in their offspring. In these cases, one of the solutions known to be efficient in order to conceive and give birth to a healthy child is the "three-parent in vitro fertilization" approach. THE MITOCHONDRIAL GENOME IS MATERNALLY INHERITED During fertilization, mitochondria from sperm are normally eliminated by a ubiquitin-dependent mechanism. As a consequence, in case the father carries the mutation both his health and fertility could become affected, but never his offspring (2). By contrast, mitochondria in the oocyte must present a specific location and distribution pattern (which represents an actual sign of oocyte maturation), and they are solely inherited from the mother. WHY ARE MUTATIONS IN mtDNA OR IN NUCLEAR GENES INVOLVED IN MITOCHONDRIAL FUNCTIONS SO PROBLEMATIC? Mitochondria provide energy to the cells through oxidative phosphorylation, and so mutations in their genome mainly affect structures form the nervous system, heart, skeletal muscle, pancreas, gonads, colon, blood, kidney or liver (3). Why these structures? The higher the energy demand is, the higher the need for more mitochondria in the cells (4). Also, cells that present a slower division process are more likely to present some kind of mtDNA mutation (5). HOLOPLASMY vs. HETEROPLASMY The situation in which all cells from an individual contain identical mtDNA (mutated or otherwise) is known as holoplasmy. By contrast, heteroplasmy is defined as the condition in which part of the mitochondria from the same individual present a DNA content that is different from the other. These cases are the most common among patients affected by mitochondrial DNA diseases (6). WHY DOES HETEROPLASTY REPRESENT A PROBLEM? Even though heteroplasmy implies the presence of two different DNA contents in the cell, cells with great amounts of mutant (or affected by a specific condition) mitochondria respond to proliferate their entire DNA. This is why the percentage of mutant/affected mtDNA tend to increase in certain tissues (7). THE "BOTTLENECK EFFECT" AND THE "THRESHOLD EFFECT" During oogenesis, only a subset of molecules of mtDNA are eventually amplified and passed on to the offspring (8). This effect explains why it is possible to obtain homoplasmic individuals in just a few generations (2). Previous reports on human diseases caused by an mtDNA mutation have shown that the mutation needs to be present at a certain percentage in order to manifest pathological effects. Typically, this percentage should be higher than 60-80% (8,9), although it also depends on age, affected tissues, type of mutations, etc. (4) WHY TO APPLY THE "THREE-PARENT IN VITRO FERTILIZATION" APPROACH? It might be reasonable to think of other possibilities to treat patients suffering from mitochondrial diseases in order to achieve pregnancy. Rather than prenatal diagnosis or preimplantation genetic diagnosis, the "three-parent in vitro fertilization" technique because (4): In the first case: 1. It needs a uniform mtDNA distribution in the extra-embryonic and foetal tissues. 2. It needs the mutant DNA load to remain constant over time. 3. There must be a close relation between the severity of the disease and the amount of mutant DNA. As for preimplantation genetic diagnosis (6,9): 1. It is not applicable to patients with high levels of heteroplasmy. 2. It reduces but does not eliminate the risk of suffering from a mitochondrial disease-related condition. 3. The amount of tDNA found in blastomeres or the trophectoderm does not represent the whole embryo. 4. This approach is not an efficient diagnosis due to the combination of heteroplasmy and the "bottleneck effect". KNOWB TECHNIQUES TO BE USED FOR MITOCHONDRIAL REPLACEMENT Pronuclear transfer It involves the transfer of the two pronuclei from a zygote affected by diseased (or mutated) mitochondria into an enucleated zygote containing healthy mitochondria. Even though this technique has not yet been performed in humans, the efficiency of pronuclear transfer in mice has been adversely affected by descendants bearing high levels of carryover mtDNA (10). Polar body transfer Since the polar body has a lower proportion of mitochondria around it, this is currently considered the best method for preventing the transmission of mutated mtDNA on to the next generation (9,10). Embryos derived from polar body transfer support normal fertilization and are capable of producing live offspring in mouse. Polar body transfers leading to a minimal amount of affected mtDNA carryover have demonstrated the great potential of this technique for preventing inherited mitochondrial DNA diseases (9,10). When applied in mice, this technique has shown the best success rate so far due to the transfer of mitochondria being lowered to a minimum (9). Spindle transfer This technique involves transferring the meiotic spindle along with the associated chromosomes, the spindle-chromosome complex (SCC), from an unfertilized oocyte with affected mitochondria into an enucleated healthy mitochondria-containing oocyte (11). This technique recently became popular when performed by Dr. John Zhang and his team, hitting the media within the latest weeks. However, potential problems could arise; just as for the previous techniques, the spindle is also surrounded by mitochondria, and so they could too be introduced into the ooplasm, thus causing heteroplasmy (12). WHAT DOES LAW STATE REGARDING MITOCHONDRIAL REPLACEMENT? So far, scientific societies are very skeptical about experimental techniques. Thus, this particular approach is specifically prohibited by the Food and Drug Administration (FDA) in the US. It can only be performed in countries such as Mexico, where legislation is more flexible, or in UK, where it was approved for application in very specific cases (14). CURRENT DATA ON MITOCHONDRIAL REPLACEMENT As it has been previously mentioned, cases of cytoplasm transfer have been performed. In fact, there have been around 30-50 live births from this technique. However, newborns presented certain genetic defects, and so this technique was banned and replaced by pronuclear transfer and meiotic spindle transfer (14). Such data demonstrate the potential damage that could be inflicted to the embryo when performing these techniques (12,15). In addition to this, ethical issues must also be taken into account, which means the sole possibility of successfully applying a specific procedure does not imply its moral appropriateness. In order to guarantee so, a committee of experts should pronounce their opinions and reach a consensus about it. On a related note, long-term effects derived from these procedures are still unknown, and so it would be necessary to monitor all babies born through these techniques. This post has been published in the Scientists in Reproductive Technologies (SIRT) newsletter, a special interest group representing the scientific membership of The Fertility Society of Australia. REFERENCES
|
Entries
March 2019
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media