|
Authors: María Caballero Sastre & Raquel Pillado González “Currently, performing any cryopreservation technique results in some degree of damage to the sperm.“ THE HISTORY OF SPERM CRYOPRESERVATION Human semen cryopreservation has a long history that begins at the end of the nineteenth century. After prior observations on sperm surviving cooling at very low temperatures (-150º C) (2), Mantegazza (1866) first suggested the idea of human sperm banks (3). Years later, Mantegazza proved it was possible to extend human sperm lifespan up to four days by cooling at moderate sub-zero temperatures (-17º C) (4). Although a few extra days of storability does not make much difference in practical terms, this was the starting point of further research aimed to develop techniques that would allow for longer storage periods. The next leap forward in this field was the discovery of the cryoprotectant properties of glycerol in 1949. This molecule proved to be an effective cryoprotectant agent (CPA) when combined with bovine sperm, and allowed for the development of new cryopreservation methods through which sperm could preserve their motility and fertilization capabilities even after the freezing-thawing process (5). However, during the following years this new technique was primarily applied to cryopreservation of farm animal semen rather than human semen (6). In 1953, at the University of Iowa, the first case of live birth from cryopreserved sperm was reported (7). This successful birth, along with reports of high survival percentage (67%) of human sperm after cryopreservation, popularized the utilization of glycerol with human semen. The most common method to use glycerol was the protocol described by Bunge and colleagues, which stated the processing of sperm in a 10%-glycerol solution before freezing it with dry ice (7). A decade later, the use of liquid nitrogen was introduced for long-term sperm cryopreservation. This new method led to the progressive normalization of sperm freezing as a widespread practice in healthcare (8). With the availability of long-term storage and the extended use of sperm freezing, new methods and variants were developed over time, such as slow freezing, fast freezing, LN2 vapours or lyophilization, which will be later described. Nowadays, cryopreservation is routinely used in most assisted reproduction centres for numerous reasons:
It is important to possess a clear understanding of the semen cryopreservation process due to its current importance in clinical and research environments. Modern assisted reproduction practices are unthinkable without this tool. CRYOPRESERVATION TECHNIQUES Before being frozen, a sperm sample needs to be appropriately processed in order to separate sperm cells from the seminal plasma. This helps increase the concentration of high quality spermatozoa for a later use. Different techniques for sperm selection have been reviewed in our previous post. Cryoprotectants The process of cryopreservation may involve irreversible cellular damage due to a change in the osmotic balance (11). Upon freezing of the extracellular water, the solute concentration increases in this fraction. As a result, the intracellular water is transported out of the cell to restore the osmotic balance, which may lead to cell dehydration and irreversible membrane damage (12, 13). Consequently, CPAs need to be incorporated along the sperm sample following processing. These molecules will protect spermatozoa by reducing intracellular ice formation and by decreasing the freezing point of the medium and the solute concentration present in the extracellular environment (14). There are two kinds of cryoprotective agents: permeable and non-permeable. The permeable ones, such as glycerol, dimethyl sulfoxide or ethylene glycol, protect the intracellular structures and biomolecules. Non-permeable agents, like sucrose, polysaccharides and some proteins, contribute to keeping the osmotic equilibrium, thus preventing cellular dehydration (11). Procedures - Cryopreservation in liquid nitrogen (LN2) This is the most commonly used method to cryopreserve sperm. It can be subdivided into three methods:
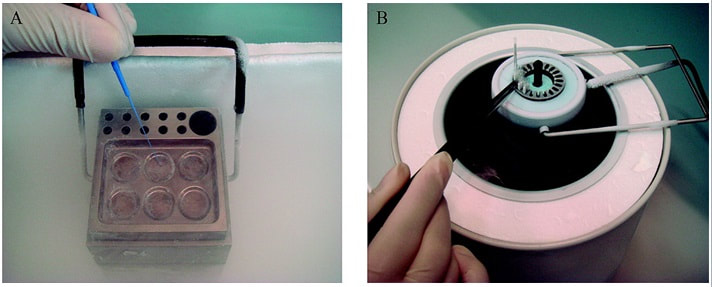
- Cryopreservation in microdroplets Microdroplets are sometimes used in the clinic to cryopreserve sperm in small volumes. This is preferred in cases such as epididymal sperm aspiration. Around 50-100 μL of the sperm-CPA solution are placed on a dry ice plate up to freezing (Fig. 2); droplets formed in such a way are then kept in vials and plunged into LN2 (18). - Vitrification This technique consists of cooling the sample at ultrarapid rates, so that the water solidifies (vitrifies) as a glass-like structure rather than forming ice crystals (19). However, even though vitrification should cause minimal damage, this is not always the case in clinical practice. One of the most frequent problems is the requirement for very high CPA concentrations that sperm do not tolerate well (20). Despite this, some studies have demonstrated it is possible to perform vitrification without using CPAs (20-22). Also, the large volume of sperm typically used impedes the cooling of the sample at the appropriate speed, causing ice formation (4). Despite these limitations, a vast proportion of clinics use vitrification as a routine practice due to its practical advantages. In recent years, a new variant of this technique has been developed. This evolved version is called Solid Surface Vitrification (SSV) (23, 24). For this procedure, the sample or tissue is directly exposed to a metal surface previously precooled at -160º C before the use of LN2 (Fig. 1). This method prevents the apparition of nitrogen bubbles and evaporation that would slow the cooling rate (24). This technique has been previously applied to animal mature oocytes and human gametes and embryos, yielding successful results (23, 24). OUTCOME COMPARATIVE BETWEEN TECHNIQUES Because every cryopreservation technique shows different advantages and disadvantages, it is important to bear in mind how sperm will be affected during the thawing process. Parameters such as motility, viability, morphology and DNA integrity are evaluated accordingly. Regarding LN2 techniques, no studies so far have directly compared post-thaw sperm quality following slow and rapid freezing. The literature shows agreement between studies on the main problem in both processes, which is controlling cooling rates (reviewed in 16). If the cooling rate is too fast, ice crystals may be formed inside the cells. By contrast, if it is too slow, the result could be cell contraction due to osmotic stress (25). This issue can be faced by using an automated programmable freezer, but only when keeping a large number of samples (26). Even though several reports have compared fast and slow freezing in animal reproduction, conclusions are controversial. Some studies in semen from horse (27) and buffalo (28) have reported better results when using fast thawing, whereas other authors have found no difference between fast and slow thawing rates (29). Nevertheless, it seems clear that the critical point lays on thawing matching the freezing process. Considering rapid freezing, thawing is recommended to be also carried out at a fast rate to avoid formation of intracellular ice crystals. Likewise, for the slow-rate cooling procedure, the sample needs a slow thawing protocol, since cells need more time to rehydrate (30). On the other hand, all studies on the use of LN2 show similar or slightly better results regarding the aforementioned parameters when using nitrogen vapours (31-33). These results, however, are obtained after short-term storage of samples in nitrogen vapours of up to three months. When stored for longer, sperm quality decreases. Consequently, this method is only recommended for short-term storage (further research would be needed in order to support its application for long-term storage) (34). Upon comparison between vitrification and LN2 techniques, different results can be highlighted. Certain authors determined that results of sperm parameters such as motility, viability and normal morphology were similar between vitrification and rapid freezing techniques (21). On the contrary, different results were found for DNA fragmentation rates. Whilst some groups found that DNA fragmentation was significantly higher for the rapid freezing technique (12, 21) or for LN2 vapours (24), other groups obtained contradictory or uncertain results (35, 36). For instance, DNA fragmentation has been observed to increase over time when analyzing semen 6h after thawing, compared to recently-thawed samples (35). Despite the different results obtained, vitrification shows important advantages compared to other available techniques. Some of these advantages are: 1) unnecessary use of CPAs; 2) the technique is simpler and faster compared to conventional slow freezing, due to the fact that once the sample is kept in a proper container (such as cryoloop or straws), it is rapidly plunging into LN2 to be stored; 3) no requirement for programmable freezers; 4) the sample is free of seminal plasma and potential pathogens (vitrification is usually performed after swim-up); 5) no requirement for post-thaw processing (reviewed in 6). TRENDS IN IMPROVING SEMEN CRYOPRESERVATION Currently, performing any cryopreservation technique results in some degree of damage to the sperm (4). The severity may differ depending on the initial quality of the sample, being greater in poor quality semen. Luckily, the application of ICSI allows for the successful use of low quality sperm (if necessary) even after having been cryopreserved. Side effects of cryopreservation on sperm include reduced motility, vitality, viability and increased DNA damage. Although motility is the most affected parameter, DNA damage entails greater detrimental effects regarding embryo viability (6). The majority of the harm produced by cryopreservation occurs during the freezing and thawing phases, the crucial moments being between -15º C and -60º C. It is worth mentioning that a considerable damage is produced by CPAs themselves; these agents cause oxidative stress that derives in the formation of reactive oxygen species (ROS) (8, 10, 37). Furthermore, these components affect the polyunsaturated fatty acids in plasmatic membranes due to lipid peroxidation (10). Consequently, phospholipids reservoirs such as egg yolk are usually added to the freezing media. As a more direct countermeasure, several current research lines look into numerous antioxidant components in order to be added to freezing media, thus avoiding the damage caused by ROS. Examples of these antioxidants are TAT-peroxiredoxin-2 fusion protein, quercetin or melatonin. Preliminary research indicates that their presence results in higher motility and viability rates post-thawing, along with a reduction of intracellular ROS levels (8, 10). Other approaches consider the utilization of protocols that may directly disregard the use of CPAs, such as certain vitrification protocols previously mentioned. Sperm freezing entails other associated problems, too, such as the loss of chromatin and acrosome integrity that had been observed post-thaw. Recent data have reported differences in the levels of DNA and acrosome integrity after cryopreservation depending on the freezing technique used (24). The difference in DNA integrity levels is suspected to be due to the cold shock faced by the samples (24). This issue could be amended by the development of media able to preserve sperm without freezing, an avenue that is currently being pursued. For instance, Riel and colleagues have reported that the use of an electrolyte-free medium for short-time (1 week) storage of semen yields better levels of DNA integrity in comparison to traditional cryopreservation. If the storage period capacity could be further improved, this might become a rather attractive alternative (38). Lyophilization or freeze drying is an experimental technique that has been proven less harmful to the DNA (4, 10, 38). In order to perform this method, the sample must be cooled below the triple point of water (Fig. 3). At this temperature solid water (ice) sublimates when the pressure is decreased and exits the cell, leaving it fully dehydrated (4, 10). However, this process irreversibly damages the sperm membrane, thus resulting in non-motile or even non-viable (dead) sperm. Nevertheless, studies on mouse sperm have shown that lyophilized spermatozoa can be used for fertilization with the assistance of ICSI (39). Although the first attempts to use lyophilization on human sperm were in the 50s, today there is still a lacking protocol for this technique that is able to preserve both sperm motility and viability (4). 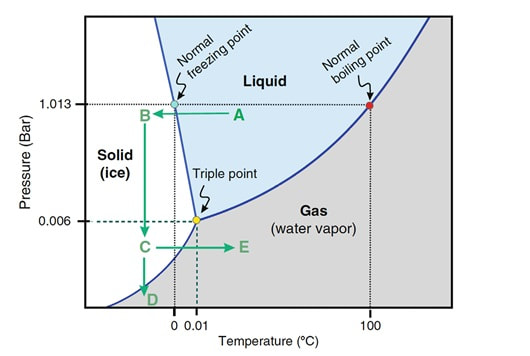 Figure 3. Water phase diagram showing the relation of the conditions of temperature and pressure for freeze-drying (not in scale). Samples are frozen by reducing the temperature (A to B) and then the pressure is also reduced by aspiration (creating vacuum) so the sample lies below the ‘triple’ point (C) for both temperature and pressure (this is the point where all states co-exist). From here on the sample is subjected to a controlled increase of temperature or to a further decrease of pressure to sublimate ice (for detailed current sperm freeze-drying protocols, see (40) and (41)). Modified from (41). The main advantages of freeze drying are: the possibility to preserve spermatozoa with high DNA integrity for at least a year and a half (39), the inactivation of viruses that may be present and the fact that liquid nitrogen is not required. Additionally, samples can be stored at 4º C and transported at room temperature (4). To date, this method still remains experimental regarding humans, due to the lack of actual data on the matter (4, 10). CONCLUSIONS Cryopreservation has gone a long way. Its use in reproductive medicine got to revolutionize the horizon for infertile couples. New doors opened decades ago, and it is fair to reason new ones will open in the near future. Egg donation, social freezing, embryo cryopreservation. Times and timing have changed for patients, and clinics and reproduction centres faced the need for evolution in order to cope with rising approaches. In spite of the variety of options for semen cryopreservation, all of them present their own limitations. Continuous research allows for the discovery of new ways to correct these flaws; however, there lies a long path ahead, and further studies will be required before any improvement can be incorporated to routine practice. REFERENCES
1. Wang X, Catt S, Pangestu M and Temple-Smith P. Live offspring from vitrified blastocysts derived from fresh and cryopreserved ovarian tissue grafts of adult mice. Soc Reprod Fert.2009;138(3): 527–535. 2. Varghese AC, Nandi P, Mahfouz R, Athayde KS, Agarwal A. Human Sperm Cryopreservation. In: Varghese, AC., Nandi, P., Mahfouz, R., Athayde, KS., Agarwal A, editor. ANDROLOGY LABORATORY MANUAL [Internet]. Cleveland Clinic. 2014; p.196–206. 3. Bunge, GR., & Sherman, KJ. Fertilizing Capacity of Frozen Human Spermatozoa. Nature.1953; 172(4382):767-8. 4. Mocé E, Fajardo AJ, Graham JK. Human sperm cryopreservation. EMJ. 2016;1:86–91. 5. Polge C, Smith AV, and Parkes AS. Revival of Spermatozoa after Vitrification and Dehydration at Low Temperatures Nature.1949; 164, 666. 6. Sharma R, Kattoor AJ, Ghulmiyyah J, Agarwal A, Sharma R, Kattoor AJ, et al. Effect of sperm storage and selection techniques on sperm parameters. Syst Biol Reprod Med. 2015;61(1):1–12. 7. Bunge RG, Sherman JK. Fertilizing capacity of frozen human spermatozoa. Nature. 1953;172:767–768. doi: 10.1038/172767b0. 8. Rozati H, Handley T, Jayasena C. Process and Pitfalls of Sperm Cryopreservation. J Clin Med. 2017;6(89):1–13. 9. Tiwari A, Tekcan M, Sati L, Murk W, Stronk J. A new media without animal component for sperm cryopreservation : motility and various attributes affecting paternal contribution of sperm. J Assist Reprod Genet. Journal of Assisted Reproduction and Genetics; 2017;34:647–57. 10. Karimfar MH, Niazvand F, Haghani K, Ghafourian S, Shirazi R, Bakhtiyari S. The protective effects of melatonin against cryopreservation-induced oxidative stress in human sperm. Int J Immunopathol Pharmacol. 2015;28(1):69–76. 11. Sieme H, Oldenhof H, Wolkers WF. Mode of action of cryoprotectants for sperm preservation. Anim Reprod Sci [Internet]. Elsevier B.V.; 2016;1–14. 12. Elliott GD, Wang S, Fuller BJ. Cryobiology Cryoprotectants : A review of the actions and applications of cryoprotective solutes that modulate cell recovery from ultra-low temperatures. Cryobiology [Internet]. Elsevier Ltd; 2017;76:74–91. 13. Royere D, Barthelemy C, Hamamah S, Lansac J. Cryopreservation of spermatozoa: a 1996 review. Hum Reprod Update. 1996;2(6):553-9. 14. Thachil JV, and Jewett, MA. Preservation techniques for human semen. Fertil Steril. 1981;35:546–8. 15. Holt, WV. Basic aspects of frozen storage of semen. Anim Reprod Sci. 2000; 62(1–3):3-22. 16. Sherman, JK. Cryopreservation of human semen. In: Handbook of the Laboratory Diagnosis and Treatment of Infertility. 1990; Keel B. and Webster BW, eds. Boca Raton, Fla, USA: CRC Press, pp. 229–59. 17. Fountain D, Ralston M, Higgins N, Gorlin, J, Uhl L, Wheeler C, et al. Liquid nitrogen freezers: a potential source of microbial contamination of hematopoietic stem cell components. Transfusion, 1997;37:585–91. 18. Abdelhafez F, Mohamed B, El-nashar S, Sabanegh E, Desai N. Techniques for cryopreservation of individual or small numbers of human spermatozoa : a systematic review. Hum Reprod. 2018;15(2):153–64. 19. Kuleshova LL, Lopata A. Vitrification can be more favorable than slow cooling. Fertil Steril. 2002;78(3):449-54. 20. Isachenko E, Isachenko V, Katkov II, Dessole S, Nawroth F. Vitrification of mammalian spermatozoa in the absence of cryoprotectants: from past practical difficulties to present success. Reprod Biomed Online. 2003;6(2):191-200. 21. Agha-Rahimi A, Khalili MA, Nabi A, Ashourzadeh S. Vitrification is not superior to rapid freezing of normozoospermic spermatozoa: effects on sperm parameters, DNA fragmentation and hyaluronan binding. Reprod Biomed Online. 2014;28(3):352-8. 22. Nawroth F, Isachenko V, Dessole S, Rahimi G, Farina M, Vargiu N et al. Vitrification of human spermatozoa without cryoprotectants. Cryo Letters. 2002;23:93–102. 23. Kamath, MS., Muthukumar, K., Appendix B: Solid Surface Vitrification. Methods Mol. Biol.2017;1568:297-307. 24. Rahiminia T, Hosseini A, Anvari M, Ghasemi-esmailabad S, Talebi AR. Modern human sperm freezing: Effect on DNA , chromatin and acrosome integrity. Taiwan J Obs Gynecol. 2017;56(Feb):472–6. 25. Said TM, Gaglani A, Agarwal A. Implication of apoptosis in sperm cryoinjury. Reprod Biomed Online. 2010;21(4):456-62. 26. Pugliesi G, Fürst R, Carvalho GR. Impact of using a fast-freezing technique and different thawing protocols on viability and fertility of frozen equine spermatozoa. Andrologia. 2014;46(9):1055-62. 27. Fürst R, Carvalho GR, Fürst MCO, Ruas JRM, Borges AM, et al. Efeito do resfriamento do sêmen eqüino sobre sua congelabilidade. Arq Bras Vet Zootec. 2005;57:599–607. 28. Shah SA, Andrabi SM, Qureshi IZ. Effect of equilibration times, freezing, and thawing rates on post-thaw quality of buffalo (Bubalus bubalis) bull spermatozoa. Andrology. 2016;4(5):972-6.b. 29. Vidament M, Yvon JM, Couty I, Arnaud G, Nguekam- Feugang J, et al. Advances in cryopreservation in modified INRA 82. Anim Reprod Sci 68:201–218. 30. Mazur P. Basic concepts in freezing cells. In: Proc. 1st International Conf. Deep Freezing Boar Semen. Uppsala, Sweden, 2005;91–111. 31. Amesse LS, Srivastava G, Uddin D, and Pfaff-Amesse T. Comparison of cryopreserved sperm in vaporous and liquid nitrogen. J Reprod Med. 2003;48:319–24. 32. Saritha KR, and Bongso, A. Comparative evaluation of fresh and washed human sperm cryopreserved in vapor and liquid phases of liquid nitrogen. J Androl. 2001;22:857–62. 33. Satirapod C, Treetampinich C, Weerakiet S, Wongkularb A, Rattanasiri S, et al. Comparison of cryopreserved human sperm from solid surface vitrification and standard vapor freezing method: on motility, morphology, vitality and DNA integrity. Andrologia. 2012;44(Suppl. 1):786–790. 34. Lim JJ, Shin TE, Song S, Bak CW, Yoon TK and Lee DR. Effect of liquid nitrogen vapor storage on the motility, viability, morphology, deoxyribonucleic acid integrity, and mitochondrial potential of frozen-thawed human spermatozoa. Fertil Steril. 2010;94:2736–41. 35. Gosalvez J, Nunez R, Fernandez JL, Lopez-Fernandez C, Caballero P. Dynamics of sperm DNA damage in fresh versus frozen–thawed and gradient processed ejaculates in human donors. Andrologia- 2011;43:373–377. 36. Isachenko E, Isachenko V, Katkov II, Rahimi G, Schondorf T, et al. DNA integrity and motility of human spermatozoa after standard slow freezing versus cryoprotectant-free vitrification. Hum. Reprod.2004;19:932–939. 37. Wu Y. Successful delivery derived from cryopreserved rare human spermatozoa with novel cryopiece. Am Soc Androl. 2017;5:832–7. 38. Gianaroli L et al. DNA integrity is maintained after freeze-drying of human spermatozoa. Fertil Steril. 2012;97(5):1067-73. 39. Ward MA, Kaneko T, Kusakabe H, Biggers JD, Whittingham DG and Yanagimachi R. Long-term preservation of mouse spermatozoa after freeze-drying and freezing without cryoprotection. Biol, 2003. 40. Arav A and Saragusty J. Directional freezing of sperm and associated derived technologies. Anim Reprod Sci; 2016, S0378-4320(16):30045-8. 41. Keskintepe L and Eroglu A. Freeze-Drying of Mammalian Sperm. In: Wolkers, W F and Oldenhof, H eds. Cryopreservation and Freeze-Drying Protocols. 3 ed. New York Heidelberg Dordrecht London: Springer Humana Press. 2015;.489-97.
0 Comments
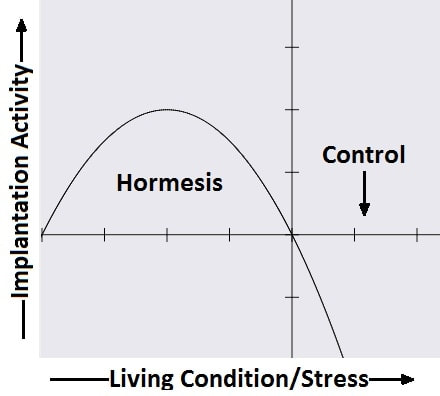
Authors: Javier del Río, Noemi Díaz and Belén Gómez INTRODUCTION What is cryopreservation? The first successful in vitro fertilization (IVF) treatment was in 1978. Since that, there have been a remarkable number of advances in assisted reproductive technologies (ART). Initially, all available embryos were transferred in IVF treatments owing to its low success rate. However, improvements on clinical and laboratory aspects led not only to increased pregnancy rates, but also to increased risk of multiple pregnancies. To prevent this, fewer embryos are transferred and leftover embryos are cryopreserved for potential future cycles use (1). The first pregnancy resulting from transferring a thawed cryopreserved human embryo was reported in 1983 in Australia (2), and the first live birth following embryo cryopreservation was reported in 1984 in The Netherlands (3). Subsequently, the need for an effective cryopreservation program arose from rapid development and improvements of assisted reproductive technology protocols (1). Cryopreservation is a method that requires cells and embryos to be exposed to non-physiological ultra-low temperatures (from -20°C to -196°C) (Fig.2). It aims to achieve “cryogenic suspension of life” through multiple steps, although this puts the elements at risk of damage or “cryoinjury” during temperature changes and phase transitions. These damages could be chilling injury or ice crystal formation, for instance, as a result of the water exchange between the intra- and extracellular compartments, consequence of dramatic changes in osmotic potential (osmotic shock). Therefore, vitrification requires the use of cryoprotectants to avoid the formation of ice crystals in the cells. Two types of cryoprotectants are necessary: permeating and non-permeating. Mixing both at different relative concentrations reduces intracellular ice formation by removing water from inside the cell. Additionally, it creates an osmotic gradient that helps restrict water movement across the cell membrane, thereby preventing osmotic shock (4). There are two typical methods used for cryopreservation: slow freezing and rapid freezing to achieve vitrification. Vitrification is a term used to describe the transformation of a solution into glass by a dramatic increase in viscosity. This method requires to minimize the time for the sample to be exposed to temperature ranges associated with chilling injury and ice crystal formation. As slow freezing, vitrification causes cell dehydration using cryoprotectants. However, unlike that, there is no attempt to maintain equilibrium on both sides of cell membrane (4). The time frame required to reach ultralow temperatures by vitrification is very brief, almost instantaneous. But, the main concern is the need for using high concentrations of cryoprotectant solutions. These might lead to osmotic shock and it could be toxic to cells, affecting embryo survival. Nevertheless, it is possible to limit toxicity by mixing different cryoprotectants, thereby decreasing their relative concentration and the exposure time of embryos to the solution (5). How efficient is the vitrification? This technique seems to be more attractive than slow freezing because it does not require expensive equipment. It uses small amount of liquid nitrogen and it is a simpler technique to perform once the embryologist has gained enough experience in it (6). A recent research performed by Viladimoiv et al. suggests advantages arising from the freezing and thawing process; the authors hypothesize a theory about “cryo-treatment of the embryo”. According to these authors, as a result of freezing or thawing of the embryos there is a decrease in reactive oxygen species levels, in the rate of mitochondrial DNA mutation and cells detoxification is carried out. Also, the authors describe another mechanism involved in restoring the mitochondrial activity (“jumping effect”) which is part of the physiological process of implantation. However, current available data cannot confirm the hypothesis yet (7). Advantages and disadvantages of fresh and frozen cycles Nowadays, fresh embryo transfers (ET) are the most common choice in IVF cycles (8). Nevertheless, in the last years, controlled ovarian stimulation has increased the uncertainty on the possible adverse effects of the ovarian hyperstimulation syndrome (OHSS), and also on possible deleterious effects on the endometrium and implications in obstetric and perinatal results (9). In spite of this, recent developments in cryopreservation of oocytes and embryos have led to substantial improvement in IVF outcomes. This resulted in a significant increase in the number of cycles with frozen embryo transfer (FET), which subsequently led to the enhancement of live births rate (10). What are the advantages of a frozen cycle? Ovarian hyperstimulation syndrome The first strong argument for FET strategy is the prevention of OHSS, that results from an increase in vascular permeability (11,8). OHSS is a medical condition affecting the ovaries of some women who take fertility medication to stimulate oocyte growth. OHSS arguably remains a major cause of morbidity in IVF treatment (10). During a fresh cycle, a woman has to undergo hormonal treatment to regulate her menstrual period, to stimulate the development of multiple oocytes (superovulation), and to encourage their maturation (11, 12). However, in a frozen cycle (FC) the patient does not have to go through ovarian stimulation or egg retrieval depending on their circumstances (13). Many people find that FETs are less stressful than fresh cycles because they do not have to worry about oocytes production or whether there will be viable embryos, since those procedures have already been done (9). Deleterious effects on the embryo The optimization of vitrification protocols has reduced the deleterious effects that this process may produce in embryos. Also, it have been observed similar survival and embryo development in FCs compared to fresh cycles (10). Moreover, best quality embryos, morphologywise, can be stored and transferred in a future cycle in better conditions. These data have allowed for an increment of success rates and the confidence of sanitary personnel and patients over FCs (5). Endometrial receptivity The implantation process, one of the crucial steps in the success of ART, requires a reciprocal interaction between the embryo and the endometrium during a small period of time called window of implantation. This interaction involves the embryo, along with its inherent molecular program of cell growth and differentiation, as well as differentiation of endometrial cells into an adequate uterine receptivity (11). Some patients may find easier to turn to FCs, since dealing with the whole process of medication during a normal cycle for ovarian stimulation may result psychologically and emotionally overwhelming. In this regard, FC may also provide a better outcome (3). The importance of an adequate endometrial environment in ART is highlighted in those patients who resort to oocyte donation, where there must be a synchronization between donor and recipient in fresh cycles. Those cases that require an improvement in endometrial receptivity to stimulate implantation of these donor oocytes seem to obtain better results in frozen cycles or in the next fresh cycle (8). Multiplet pregnancies are one of the major safety concerns of IVF due to the increased risk of neonatal and maternal complications. To achieve good results, to would be ideal to select the optimal single embryo to be transferred. Elective single embryo transfer (eSET) is the most effective way to reduce those risky pregnancies (14). How can cryopreservation damage embryos? Upon analyzing some ART studies and results, embryos are able to adapt and develop in a large range of culture media, showing different gene expression models in different environments. Cryopreservation causes stress in embryos and it is known as “hormesis”(5) (Fig.3). However, if the conditions are too unfavorable or toxic, mitochondrial activity is suppressed below the threshold necessary for the development of the embryo, so that implantation in the endometrium will be affected (5). Results of embryo transfer in fresh cycles vs. frozen cycles The main current objective of IVF professionals is to improve pregnancy rates in both fresh and frozen-thawed cycles. It is clear that embryo and endometrial receptivity are important factors to promote pregnancy rate. Recently, many researches showed FET can enhance the embryo utilization rate and improve the success rate in contrast to other research lines (15). In Roque et al. systematic meta‐analysis for 633 cycles in women aged 27-33 years old showed that FET resulted in a statistically significant increase in the ongoing pregnancy rate and clinical pregnancy compared with the fresh transfer group (8). Interestingly, the fresh group showed a higher miscarriage rate, but no statistical difference was found when compared with the frozen group. According to these data, it seems that the results of IVF-ICSI cycles can be improved by performing the FET especially in patients with normal or high follicular response. This advantage could be explained thanks to a more physiological preparation of endometrium. Several studies have also shown good results with cryopreservation of all embryos and subsequent FET in those patients most susceptible to develop OHSS (8, 16-19). In contrast, Shavit et al. found lower rates of clinical pregnancy and live births in the vitrified-warmed blastocyst group. The difference in implantation and pregnancy rates could be attributed to a higher proportion of good-quality embryos in the fresh blastocysts transfer group. They suggest that in fresh cycles highest quality blastocyst is selected for transfer and the rest are usually vitrified. Thus, vitrified-warmed blastocysts may have slightly poorer grade after warming and prior to transfer (20). In addition, it is necessary to take into account those cycles with frozen oocytes. Braga et al. found that warmed oocytes transferred in endometrial prepared cycles yield better clinical outcomes than fresh ETs. Indeed, they found that fertilization rate, embryo quality, and developmental competence was decreased in embryos derived from vitrified oocytes (12). Conversely, previous studies have suggested that the results of oocyte vitrification followed by ICSI are not inferior with regard to fertilization, embryo developmental competence, pregnancy rates, and live birth (21, 22, 23). An interesting point found in Braga et al. research is that even with lower embryo developmental quality, warmed oocytes transferred in endometrial prepared cycles resulted in higher pregnancy and implantation rates compared with transfer in fresh cycles. This finding strongly suggests that controlled ovarian stimulation impacts endometrial receptivity, which may be a possible cause of implantation failure after ovarian stimulation (12). Indeed, some studies have suggested that pregnancy rate is inversely related to serum progesterone levels on the day of hCG administration (24-27). It has been demonstrated that elevated progesterone levels on hCG trigger day negatively influence the pregnancy, regardless of the oocyte quality. Raised concentrations of progesterone in the late follicular phase are likely to influence the secretory changes of the endometrium, leading to an asynchrony between embryo and endometrial dialogue, which may result in reduced implantation rate (12). Another issue to consider is the obstetric and perinatal outcomes of frozen-thawed cycles. Maheshwari et al. quantified in a meta-analysis the obstetric and perinatal risks for singleton pregnancies after FET and compared it with those after fresh embryo transfer (28). They indicated better perinatal outcomes in singleton pregnancies after the transfer of frozen‐thawed embryos when compared to fresh IVF embryos. This could be explained by antepartum hemorrhage, very preterm birth (delivery at <32 weeks), preterm delivery (delivery at <37 weeks), small for gestational age, low birth weight (birth weight <2500 g), and perinatal mortality significantly lower in women who received frozen embryos than those transferred with fresh embryos (29, 28). It is important to note that most studies comparing perinatal outcome of singleton births conceived after fresh and cryopreserved ETs included both single and multiple ETs. Therefore, part of the adverse perinatal outcome may be attributed to the vanishing twin phenomenon, which occurs in up to 10% of multiple ETs resulting in a singleton live birth (20). What can we conclude? Elective embryo cryopreservation followed by single FET has attracted increasing attention and has been regarded as a potential innovation of IVF treatment. Choosing the well-selected embryo could further increase the chance of live birth of a eSET, which is of high clinical significance. However, there are great gaps in the literature about the risk/benefit ratio of this strategy, which includes multiple steps of treatment (30). The good outcomes in FC might be associated with having a well‐balanced embryo‐endometrium interaction in FC, and also with lacking controlled ovarian hyperstimulation, which may adversely affect endometrial receptivity during fresh IVF cycles. In addition, when hormone replacement cycles were applied in FETs, estrogen and progesterone were given in physiological doses to mimic natural cycles, while supraphysiological doses of gonadotropins were given in fresh cycles (31). On the other hand, other authors find fresh cycles as the best choice, especially in patients who resort to oocyte donation. In fact, it seems that there is a higher proportion of good-quality embryos in fresh blastocysts compared to vitrified-warmed blastocysts, which may have slightly poorer grade after warming and prior to transfer. (8, 20). In conclusion, each case must be individualized in relation to clinical characteristics of the patients and to oocyte, seminal and embryo quality. By doing so, results will be optimized in each cycle and the chances of achieving a live birth will be highly improved. REFERENCES:
1. Wong KM, Mastenbroek S, Repping S. Cryopreservation of human embryos and its contribution to in vitro fertilization success rates. Fertil Steril. 2014;102(1):19-26. 2. Trounson A, Mohr L. Human pregnancy following cryopreservation, thawing and transfer of an eight-cell embryo. Nature. 1983;305(5936):707-9. 3. Zeilmaker GH, Alberda AT, van Gent I, Rijkmans CM, Drogendijk AC. Two pregnancies following transfer of intact frozen-thawed embryos. Fertil Steril. 1984; 42(2):293-6. 4. Sparks AE. Human embryo cryopreservation-methods, timing, and other considerations for optimizing an embryo cryopreservation program. Semin Reprod Med. 2015;33(2):128-44. 5. Konc J, Kanyó K, Kriston R, Somoskői B, Cseh S. Cryopreservation of embryos and oocytes in human assisted reproduction. Biomed Res Int. 2014;2014:307268. 6. Loutradi KE, Kolibianakis EM, Venetis CA, Papanikolaou EG, Pados G, Bontis I, et al. Cryopreservation of human embryos by vitrification or slow freezing: a systematic review and meta-analysis. Fertil Steril. 2008;90(1):186-93. 7. Vladimirov IK, Tacheva D, Diez A. Theory about the Embryo Cryo-Treatment. Reprod Med Biol. 2017;16:118–125. 8. Roque M, Lattes K, Serra S, Solá I, Geber S, Carreras R, Checa MA. Fresh embryo transfer versus frozen embryo transfer in in vitro fertilization cycles: a systematic review and meta-analysis. Fertil Steril. 2013;99(1):156-62. 9. Gurbuz AS, Gode F, Ozcimen N, Isik AZ.Gonadotrophin-releasing hormone agonist trigger and freeze-all strategy does not prevent severe ovarian hyperstimulation syndrome: a report of three cases. Reprod Biomed Online 2014;29:541-544. 10. Lattes K, Prat M, Robles A, Carreras R, Brassesco M, Checa MA. Ciclos de criopreservación y vitrificación de ovocitos y embriones: indicaciones y transferencia diferida. Guía 21 de Práctica Clínica de la SEF y de la SEGO. 11. Lessey BA. Endometrial receptivity and the window of implantation. Baillieres Best Pract Res Clin Obstet Gynaecol. 2000;14(5):775-88. 12. Braga D, Setti A, Figueira R, Azevedo M, Iaconelli A, Lo Turco E et al. Freeze-all, oocyte vitrification, or fresh embryo transfer? Lessons from an egg-sharing donation program. Fertil Steril. 2016;106(3):615-622. 13. Shapiro BS, Daneshmand ST, Garner FC, Aguirre M, Hudson C. Clinical rationale for cryopreservation of entire embryo cohorts in lieu of fresh transfer. Fertil Steril. 2014;102:3-9. 14. Tobias T, Sharara FI, Franasiak JM, Heiser PW, Pinckney-Clark E. Promoting the use of elective single embryo transfer in clinical practice. Fertil Res Pract. 2016;2(1):1-9. 15. Shen C, Shu D, Zhao X, Gao Y. Comparison of clinical outcomes between fresh embryo transfers and frozen-thawed embryo transfers. Iran J Reprod Med. 2014. Jun;12(6):409–14. 16. Griesinger G, von Otte S, Schroer A, Ludwig AK, Diedrich K, Al-Hasani S, et al. Elective cryopreservation of all pronuclear oocytes after GnRH agonist triggering of final oocyte maturation in patients at risk of developing OHSS: a prospective, observational proof-of-concept study. Hum Reprod. 2007;22(5):1348-1352. 17. D'Angelo A. Ovarian hyperstimulation syndrome prevention strategies: cryopreservation of all embryos. Semin Reprod Med. 2010;28(6):513-518. 18. Griesinger G, Schultz L, Bauer T, Broessner A, Frambach T, Kissler S. Ovarian hyperstimulation síndrome prevention by gonadotropin-releasing hormone agonist triggering of final oocyte maturation in a gonadotropin-releasing hormone antagonist protocol in combination with ‘‘freeze-all’’ strategy: a prospective multicentric study. Fertil Steril. 2011;95(6):2029-2033. 19. Devroey P, Polyzos NP, Blockeel C. An OHSS-Free Clinic by segmentation of IVF treatment. Hum Reprod. 2011;26(10):2593-2597. 20. Shavit T, Oron G, Weon-Young S, Holzer H, Tulandi T. Vitrified-warmed single-embryo transfers may be associated with increased maternal complications compared with fresh single-embryo transfers. Reprod Biomed Online. 2017;35(1):94-102. 21. Trokoudes KM, Pavlides C, Zhang X. Comparison outcome of fresh and vitri- fied donor oocytes in an egg-sharing donation program. Fertil Steril. 2011; 95:1996-2000. 22. Herrero L, Pareja S, Aragones M, Cobo A, Bronet F, Garcia-Velasco JA. Oocyte versus embryo vitrification for delayed embryo transfer: an observational study. Reprod Biomed Online. 2014;29:567-72. 23. Rienzi L, Romano S, Albricci L, Maggiulli R, Capalbo A, Baroni E, et al. Embryo development of fresh ‘versus’ vitrified metaphase II oocytes after ICSI: a prospective randomized sibling-oocyte study. Hum Reprod. 2010;25:66-73. 24. Xu, B., Li, Z., Zhang, H., Jin, L., Li, Y., Ai, J. et al, Serum progesterone level effects on the outcome of in vitro fertilization in patients with different ovarian response: an analysis of more than 10,000 cycles. Fertil Steril. 2012;97 (1321-7.e1-4). 25. Wu, Z., Li, R., Ma, Y., Deng, B., Zhang, X., Meng, Y. et al, Effect of HCG-day serum progesterone and oestradiol concentrations on pregnancy outcomes in GnRH agonist cycles. Reprod Biomed Online. 2012;24:511–520. 26. Bosch, E., Labarta, E., Crespo, J., Simon, C., Remohi, J., Jenkins, J. et al, Circulating progesterone levels and ongoing pregnancy rates in controlled ovarian stimulation cycles for in vitro fertilization: analysis of over 4000 cycles. Hum Reprod. 2010;25:2092–2100. 27. Hamdine, O., Macklon, N.S., Eijkemans, M.J., Laven, J.S., Cohlen, B.J., Verhoeff, A. et al, Elevated early follicular progesterone levels and in vitro fertilization outcomes: a prospective intervention study and meta-analysis. Fertil Steril. 2014;102:448–454.e1. 28. Maheshwari A, Pandey S, Shetty A, Hamilton M, Bhattacharya S. Obstetric and perinatal outcomes in singleton pregnancies resulting from the transfer of frozen thawed versus fresh embryos generated through in vitro fertilization treatment: a systematic review and meta-analysis. Fertil Steril. 2012;98:368–77.e1. 29. Qiao J, Zhang L, Yan L, Zhi X, Yan J. Female Fertility: Is it Safe to "Freeze?". Chin Med J (Engl). 2015;128(3):390. 30. Wei D, Sun Y, Liu J, Liang X, Zhu Y et al. Live birth after fresh versus frozen single blastocyst transfer (Frefro-blastocyst): study protocol for a randomized controlled trial. Trials 2017; 18(253): 1-7. 31. Zhang L, Yan LY, Zhi X, Yan J, Qiao J. Female Fertility: Is it Safe to “Freeze?” Chin Med J. 2015;128 (3):390-7. |
Entries
March 2019
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media