|
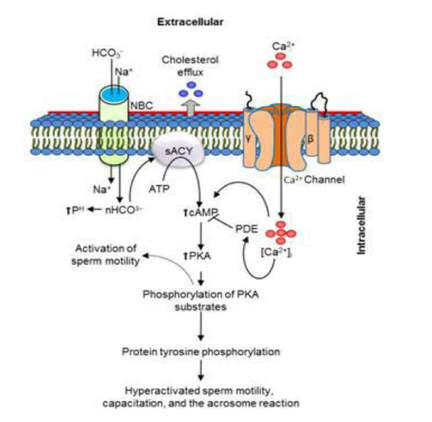
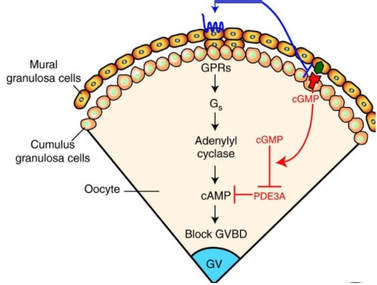
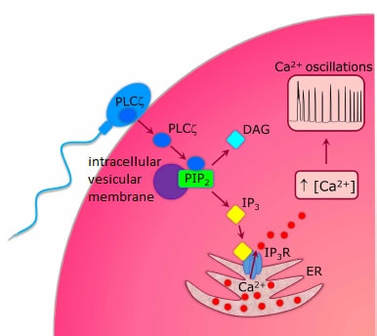
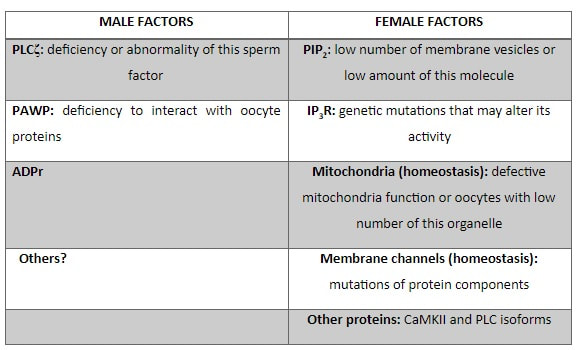
Authors: María Caballero & Lidón Carretero Vilarroig "The main cause for unsuccessful ICSI seems to be failure in oocyte activation mechanisms" 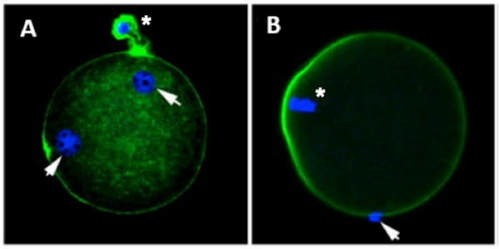 Fig 1. Zona-free mouse oocytes prepared for IVF. Staining with Draq 5 for DNA (blue) and phalloidin-Alexa Fluor 488 for filamentous actin (green) has been performed. (A) An oocyte with two pronuclei (arrows) and a polar body (asterisk) is shown, meaning activation has already begun after fertilization. (B) A spermatozoon (arrow) is observed bound to the oocyte not having penetrated the cortical actin. The equatorial plate in the meiotic spindle (asterisk) suggests the oocyte remains arrested at MII (polar body has presumably been lost during oocyte denudation), which indicates that oocyte activation has not yet been produced, given fertilization has actually not been achieved (modified from [1]). INTRODUCTION Fertilization involves the fusion of male and female gametes. However, for this to occur both cells must undergo certain morphological and physiological changes in order to be able to fuse. The sperm cell must undergo a process known as capacitation, which implies a series of changes regarding motility and plasma membrane composition (among others), essential to acquire the ability to fertilize. The oocyte in turn must not only complete several molecular changes involved in maturation, but also a combination of processes initiated by the sperm entry that culminate in the so-called oocyte activation (OA), which prepares the newly formed zygote for embryogenesis (2). In the context of in vitro fertilization (IVF) cycles, oocyte activation failure (Fig. 1) has been observed to occur in about 1-3% (3), resulting in visibly identifiable non-fertilized oocytes. In order to overcome this problem, assisted oocyte activation (AOA) has recently gained interest for those couples with a history of fertilization failure through intracytoplasmic sperm injection (ICSI). To better understand oocyte activation failure and AOA, here we review gamete maturation, focusing on oocyte activation mechanisms and where/when main problems may arise that cause failure in this process. GAMETE PREPARATION FOR FERTILIZATION Sperm Fresh ejaculated sperm is still not capable to fertilize. In order to be able to interact with the oocyte, the spermatozoon needs a final maturation step known as capacitation. This process takes place into the female reproductive tract, culminating in the acrosome reaction (4). During capacitation, the sperm cell undergoes biochemical changes related to the acquisition of fertilization ability, mainly involving the molecular reorganization and hyperpolarization of the plasma membrane, as well as hyperactivation (5). Changes in membrane organization are necessary in order to increase its fluidity, which is achieved by removing cholesterol. The mechanisms related to the efflux of cholesterol are not well understood yet, but albumin and high-density lipoproteins located in the female genital tract have been recently pointed out as the main responsible actors for this process (6). Sperm membrane reorganization leads to the exposure of some important channels and receptors, which increases the permeabilization of sperm to certain ions. Some of the most important channels are CaSper and NBC, Ca2+ and Na+/HCO3- channels, respectively. Calcium influx plays an important role in the process of capacitation, contributing to acrosomal reaction and aiming to the exocytosis of the acrosomal vesicle (8). Another milestone of capacitation is hyperactivation, referred to as the acquisition of increased and differential sperm motility. This is activated by phosphorylation of protein kinase-A (PKA) in a (HCO3−)- and (Ca2+)-dependent manner, being these ions transported through the cell membrane via CaSper channel and NBC cotransporter, respectively (9). Additionally, tyrosine phosphorylation levels of a multitude of proteins rises significantly in the sperm while in the female tract. Such an increment causes an increase in the activity of the adenylate cyclase, which in turn causes intracellular cyclic adenosine monophosphate (cAMP) levels to rise, ultimately resulting in a differential oscillating movement of the flagellum (hyperactivation). This particular pattern of flagellar beating provides the sperm cell with the proper motility to move forward towards the ovum (Fig. 2). Finally, when the sperm cell reaches the oocyte, acrosome reaction (AR) is triggered, upon interaction with the zona pellucida (ZP). AR refers to the regulated exocytosis of the acrosome content (including several crucial enzymes) and exposure of surface antigens necessary for oocyte-sperm recognition. The acrosome and plasma membranes fuse, tipping out the content and enabling lateral contact between the sperm head plasma membrane and the oolemma. This is followed by fusion of the plasma membranes of both gametes and delivery of the sperm nuclear material into the oocyte. Oocyte Oocytes begin meiosis during foetal development and arrest at a special diplotene stage of prophase I called dictyotene or dictyate, so women are born with a limited number of primary oocytes. There are two mechanisms used by the oocyte to achieve meiotic arrest. The first one can be explained due to the low activity of M-phase promoting factor (MPF), promoted by the oocyte itself via endogenous production of cAMP (10). The oolemma exposes G-protein receptor (GPR3) leading to G-protein-coupled GPR3 and GPR12 activation (11), which in turn stimulates cAMP, a cyclase responsible for AMP synthesis. The second mechanism uses the cumulus-oocyte complex (COC) as a barrier that prevents meiosis reactivation. COC cells produce guanosine 3′, 5′-cyclic monophosphate (cGMP), which enters into the oocyte through gap junctions. Once inside the oocyte, cGMP inhibits the phosphodiesterase responsible for cAMP hydrolysis (PDE3A or cGMP-inhibited phosphodiesterase), which increases cAMP activity, thus inhibiting MPF (10) (Fig. 3). Primary oocytes remain at meiotic arrest until puberty, when the pituitary gland releases luteinizing hormone (LH) during the menstrual cycle and before ovulation. LH acts as a molecular key and activates MPF. LH receptors are located on the surface of granulosa cells, so they transfer the meiotic arrest release signal into the oocyte. Nevertheless, after the resumption of first meiotic division, the maintained activity of MPF promotes the oocyte arrest at metaphase II (12,13). OOCYTE ACTIVATION As a result of the sperm penetration into the oocyte, the female gamete undergoes a process called oocyte activation (OA). This refers to the resumption of the transcriptional activity of the oocyte (inactive until fertilization), essential to allow the subsequent embryo development to progress (14). OA is characterized by the triggering of several events, such as the resumption of meiosis, pronuclei formation, second polar body extrusion, cortical granule exocytosis and genetic material replication, among others (15,16). All these events are the result of an increase in intracellular calcium (Ca2+) concentration, which takes place upon gamete fusion. In mammals, this Ca2+ release is known as Ca2+ oscillations, due to the fact that this molecule is gradually released over time. Every species shows a specific pattern of frequency and duration for these oscillations (17). The “sperm factor” The idea of the sperm containing some kind of OA trigger has been on the table for decades. Evidences showing that sperm extracts are able to fertilize eggs from different species (even phyla) suggested the existence of a common factor that would act as a universal activator of the oocyte (18, 19). Independent observations from different models revealed that the so-called “sperm factor” could effectively be a soluble calcium releasing agent, role that has been attributed to several molecules to date, including phospholipases, second messengers and others. Even involvement of adenosin diphosphate ribose (ADPr) has been discussed in this context, since it has been shown to trigger calcium release through a nitric oxide-based pathway (20, 21). Other molecules such as citrate synthase (22) or post-acrosomal WWP-domain binding protein (PAWP) (23) have been considered potential candidates for years, after their involvement in triggering OA had been demonstrated. However, several pieces of evidence have shown that the key sperm factor responsible for intracellular Ca2+ release and the subsequent oocyte activation may be a testis-specific PLC isoform named PLCζ (24). Although PLCζ is mainly located in the spermatozoon head, it remains unclear if the triggering isoform localizes in the post-acrosomal, acrosomal or equatorial region (17). Once PLCζ is delivered into the ooplasm, it induces hydrolysis of phosphatidylinositol 4,5-biphosphate (PIP2), a membrane-bound substrate located in an intracellular vesicular membrane. The hydrolysis generates two second messengers: inositol triphosphate (IP3) and diacylglycerol (DAG). Thereupon, release of Ca2+ is induced by binding of IP3 to its receptor (IP3R), localized in the endoplasmic reticulum (ER) membrane. This molecular activity causes the characteristic Ca2+ pattern responsible for triggering subsequent events explained below (Fig. 4) (18, 25). Although IP3 causes the first Ca2+ oscillations, DAG has been reported to interact with protein kinase C (PKC), thus increasing its activity. PKC also seems to be involved in events such as the production of Ca2+ oscillations, meiotic resumption or triggering of cortical granule exocytosis (26). The role of oocyte in activation Increased levels of intracellular Ca2+ have two consequences (Fig. 5). First, it enhances Ca2+ production by stimulating PLCζ and generating higher levels of IP3 (26). Secondly, it causes hyperpolarization of the plasma membrane, which results in the opening of specific channels present in the oolemma such as the store-operated Ca2+ entry (SOCE), the TRP family proteins and the plasma membrane (Ca2+)-ATPase (PMCA). Opening of these channels activates the influx of Ca2+, which is essential to keep Ca2+ oscillations and reload the intracellular stores (22, 28). 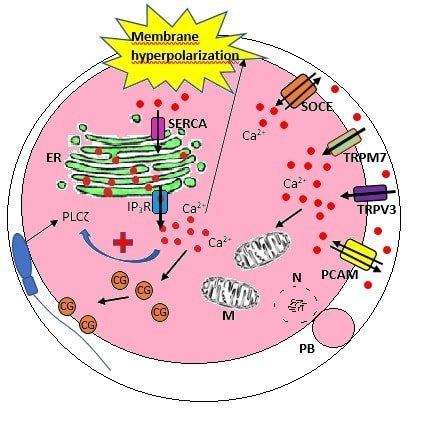 Fig. 5. Representation of the two consequences of Ca2+ increase in the fertilized oocyte. First, high Ca2+ concentration stimulates PLCζ activity and causes the hyperpolarization of the oolemma, which results in the opening of channels like SOCE, TRPM7, TRPV3 and PCAM. Secondly, whereas part of the released Ca2+ is retained by mitochondria, the rest enters into the ER through SERCA or leaves the oocyte through PCAM and SOCE channels. ER: endoplasmic reticulum. N: nucleus. M: mitochondria. CG: cortical granules. Ca2+: calcium. PB: polar body. Regarding intracellular Ca2+ release, it is necessary to keep a balance to prevent prolonged exposures to the ion that may be detrimental to the egg. Although part of this Ca2+ will be pumped back to the ER by sarcoplasmic/endoplasmic reticulum calcium-ATPase (SERCA) or the protein SOCE system, mitochondria are the actual major regulators of intracellular Ca2+ homeostasis, this means mitochondria retain Ca2+ during fertilization, so that they will act as a passive buffer regulating Ca2+ release. Ca2+ in turn activates oxidative phosphorylation (ATP synthesis). Moreover, it has been demonstrated that ATP generation may regulate intracellular Ca2+ release by making the IP3 receptors more sensitive to (Ca2+)-mediated activation (29). The remaining Ca2+ would then be expelled from the cell through PMCA and the Na+/Ca2+ exchanger (Fig.5) (30). "[...] the female gamete undergoes a process called oocyte activation (OA) [...] characterized by the triggering of several events, such as the resumption of meiosis, pronuclei formation [...] and genetic material replication, among others." Egg activation events As previously mentioned, the increase of intracellular Ca2+ concentration triggers certain key events that enable embryo development: Cortical granule exocytosis and changes in the ZP Once fertilization has occurred, the ZP needs to be modified in order to prevent binding and penetration of additional spermatozoa (“polyspermia”). This is achieved by releasing the content of the cortical granules (CGs), specialized secretory vesicles located in the periphery of the oocyte (31). First, CGs are translocated to the plasma membrane by two calmodulin-dependent proteins, CaMKII and MLCK. CaMKII is responsible for releasing the CGs attached to the cytoskeleton, whereas MLCK promotes CG translocation by phosphorylating the motor protein myosin II (14). Upon translocation, CG fuse with the oolemma in a calcium-dependent manner and deliver their enzymatic content to the perivitelline space (PS). The so secreted glycosidases, proteases and cross-linking enzymes modify the structural network of the ZP ("zona hardening"), thus hindering the entry of multiple sperm (14). Resumption of meiosis As aforementioned, MPF is directly involved in maintaining the MII arrest of human mature oocytes (32). MPF is a heterodimer composed of cyclin B and Cdc2 kinase, a regulatory and a catalytic subunit, respectively (33). Following fertilization, Ca2+ oscillations induced by PLCζ activate a phosphorylation cascade that eventually results in the destruction of cyclin B, the concomitant MPF deactivation and the subsequent resumption and completion of meiosis (14). Pronuclei formation and second polar body extrusion With the completion of meiosis, the secondary oocyte produces the second polar body and reorganizes its own chromosome complement in the form of a visible and spherical pronucleus. Formation of the paternal pronucleus in turn is dependent on maternal proteins; since the sperm genetic material is highly condensed due to its association with protamins (14), such condensation needs to be loosened in order to make DNA replication and gene expression possible. Maternal histones then replace protamins and the paternal pronucleus becomes appropriately remodelled. Then, maternal and paternal pronuclei fuse together in a process known as singamy or karyogamy (34). Maternal RNAs and zygote genome activation (ZGA) Upon egg activation, embryo development commences. However, during early stages, the embryo genome remains transcriptionally inactive. Therefore, the maternal mRNAs and proteins present in the oocyte take charge of early development (14). Maternal mRNAs are modified to regulate gene expression at the postranscriptional level, which includes edition, splicing, translation and degradation (35). Subsequently, during the so-called maternal-to-zygotic transition (MZT) nuclear genome becomes transcriptionally active. Two main molecular activities are involved in this period: (1) maternal clearance, which means the deletion of maternal mRNAs and proteins necessary for oocyte maturation and first stages of embryogenesis; and (2) gene expression with new instructions for embryo development. This process is activated by the maternal genome and it is known as zygotic genome activation (ZGA) (14). ZGA is not a sudden event, but it has been found to occur in a wave-like fashion. Similar studies in mice and humans have shown that ZGA commences as early as the 1-cell stage; concomitant with maternal mRNA degradation, a minor zygotic gene expression is observed, followed by the major ZGA and the mid-preimplantation gene activation (MGA) (compaction and cavitation processes are also accompanied by waves of gene expression activation) (36, 37). ICSI FOLLOWING OOCYTE ACTIVATION FAILURE Between 10-16% of couples worldwide are unable to have children without draw on assisted reproduction techniques (38). Both conventional IVF and ICSI have been proven to be highly successful in ART treatments worldwide, however, the use of ICSI has been reported to be of 66% in 61 countries between 2008 and 2010 and it continues increasing, even in cases with normal semen parameters (39). This success may be explained by to the fact that ICSI shows the highest success rates (around 97-99%) (38), and also because it represents a useful method to rescue fertility for men with suboptimal semen parameters, or even for couples whose fertilization rates after conventional IVF are close to zero (3). Despite the low failure rates, ICSI cycles do sometimes fail. There exist several possible causes that may explain failure of ICSI cycles, such as technical factors, failed sperm head decondensation, oocyte spindle defects or poor sperm chromatin condensation (40). Nevertheless, the main cause for unsuccessful ICSI seems to be failure in oocyte activation mechanisms, due to either sperm or oocyte defects (17, 39) (Table 1). Sperm factors associated with oocyte activation failure Several independent studies have found deficient PLCζ levels when both morphologically normal and abnormal sperm (such as in cases of globozoospermia) from different patients were analysed using mouse oocytes (41, 42). Results have shown reduced or absent levels of PLCζ in the sperm head to be associated with two mutations found in the active domain-coding regions of the gene. Although a deficiency in PLCζ has been associated with most cases of men infertility, other molecules might also be involved in oocyte activation failure. Evidences exist that have linked PAWP, a sperm-specific protein located in the sperm perinuclear theca, to the process of OA. Previous studies have shown this protein to induce pronuclei formation and meiosis resumption when injected into swine mature oocytes. The authors suggested one of PAWP domains to interact with PLCγ, a PLC isoform present in the oolemma, thus contributing to the generation of Ca2+ oscillations (23). This suggests that defective levels of the protein or defective interactions with oocyte factors may eventually lead to oocyte activation failure (23). Oocyte factors associated with oocyte activation failure However, neither PLCζ nor any other sperm factor deficiency is always the main responsible for ICSI failure. This points to the existence of oocyte factors that contribute to such failure, as well. Unfortunately, evaluation of human oocyte-related problems is usually quite difficult due to ethical issues and restrictions. As a consequence, the number of available oocytes destined for research is actually low. Therefore, an in-depth study of these potential oocyte-related factors is still necessary nowadays (39). Despite such problems, there are some actual hypotheses about oocyte-related factors that might be involved in activation failure. As it has been previously exposed, PIP2 plays an essential role, since it is the first element to interact with PLCζ. Some studies have suggested that either a small number of oocyte vesicles containing PIP2 or even a low amount of PIP2 in them could lead to oocyte activation failure (43). Once PIP2 is hydrolysed, IP3 binds to its receptor. Certain genetic mutations in IP3R may block this interaction, thus preventing Ca2+ oscillations to occur. Such mutations may also be responsible for abnormal protein folding or abnormal protein trafficking (39). Moreover, high levels of PLCζ are likely to lead to overproduction of DAG, which may contribute to oocyte activation failure by creating unusual patterns of Ca2+ oscillations (26). Even when Ca2+ oscillations occur normally, it is important to keep a balance within and outside the oocyte; any mistake in homeostasis regulatory mechanisms may compromise OA (39). As the main regulators of homeostasis, mitochondria function needs to be guaranteed; defective mitochondrial activity due to a low number of these organelles may alter ATP production and generation of Ca2+ oscillations, leading to defects in OA (44). On the other hand, homeostasis is also regulated by the correct function of specific membrane channels. For instance, SOCE is responsible for the introduction of Ca2+ into cellular stores (45). Both components of this system, STIMI and ORAII, may also result mutated, in which case their compromised function would negatively affect the normal occurrence of Ca2+ oscillations (39). There also exist other oocyte proteins that could lead to activation failure. For instance, the above-mentioned CaMKII is a calcium/calmodulin-dependent protein kinase II that induces cyclin B1 degradation, allowing resumption of meiosis (46). CaMKII is dependent on Ca2+ oscillations, so defects in calcium homeostasis might impair the proper function of the protein and, in consequence, the meiotic progression (39). Additionally, other PLC isoforms present in the oolemma and different from PLCζ like β, γ or δ also seem to have a role in Ca2+ oscillations; in particular isoform γ has been found to interact with PAWP (29, 47). ASSISTED OOCYTE ACTIVATION Being oocyte activation failure the main cause for fertilization failure in ICSI cycles, some private clinics perform a special technique to test whether sperm is responsible for failure in oocyte activation. This test is known as MOAT test (Mouse Oocyte Activation Test) (48), and it consists in performing ICSI using sperm sampled from the patient and mouse oocytes. Sperm samples with proven fertility are used as positive controls, while the negative control is represented by injection of culture medium. Based on the results, patients are classified into one of three groups depending on the oocyte activation rate: (i) 0-20% (non-activation group); (ii) 21-84% (intermediate activation group); and (iii) 84-100% (sperm-related infertility refused). In addition, a calcium pattern analysis is also performed. Calcium plays a very important role in all forms of cell signaling. As already explained, calcium contributes to acrosomal reaction and enables the spermatozoon to penetrate into the oocyte (49). When the spermatozoon fertilizes the ovum, it triggers the generation of calcium waves leading to OA and embryonic development (50). A recent article published by Ferrer-Buitrago et al discusses whether calcium analysis can predict the response to AOA in patients with a history of failed fertilization after ICSI. Even though the sample size was limited, the results shown suggest that calcium analysis may actually foretell the response of ICSI-AOA, only in patients with abnormal capacity to generate Ca2+ oscillations (51). There have been several protocols used for AOA depending on the mechanisms used to trigger Ca2+ oscillations, which can be classified into electrical, mechanical or chemical (52). The first one actually involves electroporation of the oocyte, which generates micropores in the cell membrane allowing Ca2+ influx (53). The mechanical approach consists in oolemma breakage by vigorous aspiration of the ooplasm. The spermatozoon is then introduced into the oocyte, followed by energetic ooplasm aspiration and re-injection during ICSI. This way extracellular Ca2+ from the medium is introduced into the oocyte, resulting in its activation (54). Nevertheless, chemical-based protocols are the most extended techniques. Treatment with ionomycin, calcimycin, ethanol, strontium chloride (StCl2) or calcium ionophores can restore normal calcium oscillations (55). Among the later, the use of A23187 is extended about patients with globozoospermia, whose sperm lack the acrosome and may carry a mutant form of PLCζ. According to the protocol by Rybouchkin et al., oocytes are exposed to the ionophore in the IVF medium after ICSI, and cleavage stage can be assessed the next day (55, 56). Several cases of successful pregnancy have been reported following this ICSI-AOA approach. Few years ago, Hye Jin Yoon and coauthors published results from 185 ICSI AOA cycles using a calcium ionophore ending up in thirty-eight healthy babies with no congenital birth defects (57). The same year, Kim et al. (58) reported five successful pregnancies following ICSI-StCl2 treatment that resulted in the birth of eight healthy children. These results point to AOA as a new therapeutic tool for couples experiencing complete fertilization failure after ICSI. CONCLUSIONS Although ICSI cycles show a high percentage of success (97-99%), there is still certain probability of not achieving fertilization. Among the different causes responsible for this, the most common is the oocyte activation failure. For fertilization to succeed, it is important that both female and male gametes undergo a series of changes. In the case of spermatozoa, they need to be capacitated before oocyte penetration. On the other hand, upon fertilization, the mature oocyte needs to be activated in order to start embryo development. This process, known as oocyte activation, is characterized by internal Ca2+ oscillations produced by sperm factors like PLCζ, which is liberated into the ooplasm after fertilization. Ca2+ oscillations trigger the following events: (1) resumption of meiosis, (2) pronuclei formation, (3) second polar body extrusion, (4) cortical granule exocytosis and (5) maternal mRNA replication. This complex process needs a control in the homeostasis that will be basically regulated by the mitochondria present in the oocyte and by some channels in the ER and the oolemma. Oocyte activation failure is caused by the absence of Ca2+ oscillations or by alteration of their frequency. This failure can be related with both gametes. Regarding sperm, deficiencies in PLCζ have been reported to be directly associated with the absence of Ca2+ oscillations. As for oocytes, and even though studies are scarce due to ethical restrictions, certain hypotheses have proposed activation failure to be associated with deficiencies in mechanisms of homeostasis. This actually means failures in mitochondria and related channels, and also with some molecules like PIP2, DAG, PLC isoforms and IP3R. In order to avoid oocyte activation failure, assisted reproduction clinics have developed certain protocols for assisted oocyte activation, being those with calcium ionophores the most widely extended in fertility centres. REFERENCES
14 Comments
Ved Prakash
2/5/2018 22:59:03
Great topic. I sure it will give me better understanding of the subject.
Reply
María
2/6/2018 14:22:22
Thank you very much Ved!
Reply
P.Papitha
2/8/2018 07:51:52
So informative at molecular level. Good explanation. Thanks for sharing. All the topics which is published in embryologist media is good to read.
Reply
LIDÓN
2/8/2018 20:25:02
Thank you for your comment, P. Papitha. We're glad to see people appreciate and enjoy our effort. Please keep visiting and commenting! Kind regards :)
Reply
Fred Miller
2/10/2018 18:23:44
Thank you ! An excellent review of some of the crucial events of mammalian reproductive biology. Nice to see such a clear presentation of a synthesis of years, decades really, of important research. Best, Fred Miller
Reply
LIDÓN
2/12/2018 18:07:35
Thanks a lot for your nice comment, Fred.
Reply
2/14/2018 05:55:25
Thoroughly enjoyed while reading. Excellent review Article. Thanks for sharing.
Reply
María
2/15/2018 17:15:07
We are glad to see such a good response to our text, we so much appreciate your kind words. Kind regards.
Reply
Jessica
4/29/2018 17:04:51
Although much of this is beyond me as I am not medically trained, thank you for publishing it. My husband and I have had total fertilisation failure with both standard ivf and icsi. We had some success with UK AOA and two oocytes fertilised, both making it to day 5, one fresh and one frozen transfer but sadly neither stuck. We had results from a trial at Oxford university stating my husband’s sperm has reduced levels of plc zeta but moat testing revealed normal fertilisation rates, indicating it may be an oocyte issue. Our next attempt will be with a clinic in Belgium who have a much more advanced ionophore for AOA. Any other advise is greatly appreciated.
Reply
María
5/17/2018 20:10:27
Thanks for your comment Jessica. Please excuse our delay in replying. We are glad to know you found the text informative. Since we are not medical professionals, we cannot provide any specific indication for your case. However, we do encourage you to keep asking for consultation with your doctor. We wish you all the best and good luck!
Reply
prof.dr.med.g.o.ajayi
5/6/2018 15:49:29
Thanks to the the 2 Authors.Very well written,understandable with molecular explanations. Good for sharing
Reply
María
5/9/2018 19:36:40
Thank you very much!
Reply
Ved Prakash
5/17/2018 21:26:18
Your group is doing very good work.
Reply
LIDÓN CARRETERO
5/21/2018 15:25:37
Thank you for your nice comment. We will try to review embryo culture techniques soon :)
Reply
Leave a Reply. |
Entries
March 2019
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media