|
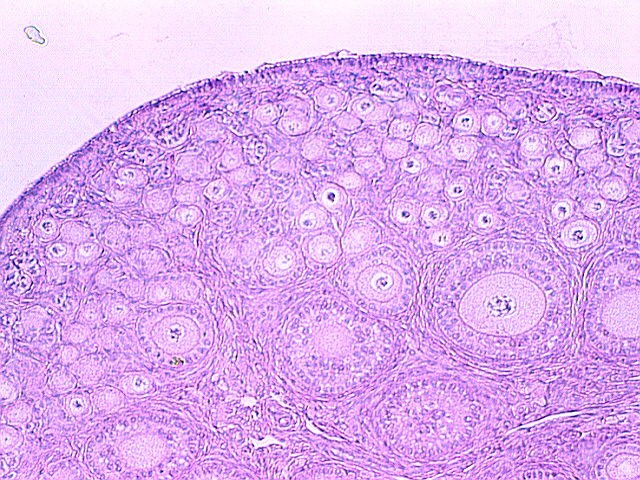
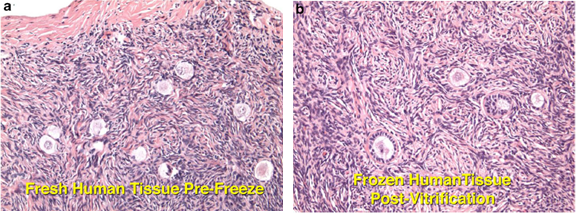
Authors: Iñaki Arroyos, Lidón Carretero Vilarroig & Belén Gómez Giménez INTRODUCTION Cryopreservation is a basic procedure in the daily work with assisted reproduction techniques. It is routinely and successfully applied to sperm samples, spare embryos from in vitro fertilization (IVF) cycles and oocytes. In the past, slow freezing procedures were carried out with no reliable clinical outcomes, especially in egg freezing programs. After the recent introduction of vitrification it is now possible to cryopreserve embryos and oocytes, and to assure patients that this process will not decrease the quality and the biological potential of the cells. At present, there is a growing trend towards a “vitrify-all” strategy after IVF with a single embryo transfer of a warmed embryo in a subsequent cycle (1, 2). There have been reports of improved implantation and pregnancy rates with frozen embryo transfers as compared to fresh autologous embryo transfer, suggesting superior endometrial receptivity in the absence of ovarian stimulation (2, 3). Oocyte cryopreservation The mature oocyte is the biggest cell in the body and contains a high proportion of water. The amount of water compromises the viability of the ovocyte during the slow freezing because ice crystals can be formed. The spindle of the mature oocyte is very sensitive to damage from ice crystal formation, and so it may easily become disrupted. Also, the oocyte is extremely vulnerable to mild chilling, which may severely compromise its integrity, and even cause complete degeneration. Innovation and improvement of vitrification was not easy in the beginning. It took a lot of research and experimentation on different protocols for the developers, as well as long time to set the right process. The procedure itself is complex, and it involves numerous critical steps for successful performance (4). Egg freezing is an increasing phenomenon because of its many advantages. It may provide an easier solution in cases of legal and/or ethical issues between the parents, such as divorce or decease. Freezing eggs rather than embryos can also avoid dealing with the complexities of having an increasing number of frozen embryos in nitrogen tanks unclaimed by the progenitors. Moreover, certain medical treatments to battle cancer can jeopardize the ovarian function, and so an egg freezing program seems suitable for such patients. Regarding social aspects, egg freezing allows women to postpone their motherhood. In addition, egg freezing also makes synchronization between donors and recipients much easier. Finally, this technique is suitable for patients who have low ovarian reserve and need PGD treatments: it allows patients to accumulate oocytes from several cycles, which increase the success of the treatments (5). Sperm cryopreservation This technique offers the possibility of freezing sperm to preserve fertility in those men subjected to treatments such as chemotherapy or radiotherapy, which may compromise gametogenesis. Additionally, sperm cryopreservation makes it possible to create sperm banks, which offer a valuable alternative to azoospermic patients willing to father children. Embryo cryopreservation Embryo freezing represents an essential method for any assisted reproduction centre, since it allows to save spare embryos from an IVF cycle and preserve them in liquid nitrogen for future uses. Ovarian Tissue Cryopreservation Ovarian tissue cryopreservation (OTC) represents another technique to preserve fertility in women. It has been carried out for more than 18 years now (6, 7) and it represents the main option available for women who require cancer treatment but cannot delay chemotherapy. Moreover, it is the only possibility for pre-pubertal patients (8). Currently, embryo and oocyte cryopreservation are the only fertility preservation techniques that are considered to be non-experimental by the American Society for Reproductive Medicine (9). Unfortunately, both approaches require previous preparation and stimulation with gonadotropins for oocyte retrieval, which usually requires 2-4 weeks or longer (10). Furthermore, oocyte harvest typically involves the use of transvaginal ultrasound and needle oocyte retrieval techniques, which could require general anaesthesia. This delay is often not possible or appropriate for females requiring urgent therapy or in breast cancer patients, because high estrogen levels might have detrimental effects on the primary tumor. Additionally, not all patients have partners with whom to create embryos for cryopreservation (10). Most clearly, these techniques are not indicated for young and pre-pubertal female patients due to the inability of their immature hypothalamic-pituitary axis to produce mature eggs (11) Under these circumstances, the possibility of cryopreservation of ovarian tissue (cortex) has become an urgent and highly-demanded technology for two types of young female cancer patients. First, those who must undergo advanced chemotherapy and/or radiotherapy. Second, those with non-oncological systemic diseases such as autoimmune or haematological conditions, that sometimes require chemotherapy, radiotherapy, or bone marrow transplantation (12). OTC does not require ovarian stimulation, and it allows to preserve gonadal function in pre-pubertal and adult patients. This procedure offers a promising option for women at high risk of premature ovarian failure and sterility (12). Furthermore, transplanting ovarian tissue not only restores fertility but also restores endocrine function (13). Ovarian cortical tissue contains the primordial follicles, which are located in the ovarian cortex, near the surface epithelium. These are the smallest female fertility unit, including 90% of the ovarian follicular reservoir. Although there are other types of follicles present in ovaries removed from the patient, primordial follicles are the ones that are considered for ovarian cryopreservation. Among other reasons, these are the most resistant follicles to cryoinjury due to of the small size of the oocytes, the reduced cytoplasmic content and the absence of meiotic spindle that could potentially be damaged (14). OVARIAN CORTEX CRYOPRESERVATION TECHNIQUE The most common steps to be followed for this approach are summarized below: 1. Biopsy. The removal of ovarian tissue is a relatively simple procedure. In order to ensure optimal penetration of cryoprotectants, approximately twelve small biopsies of ovarian cortical tissue (3x3x1 mm) are collected (15). This procedure can be performed by using minimally invasive techniques like laparoscopy, or even in conjunction with other open procedures such as primary tumor resection. Regardless of the technique, any area away from the hilum and lacking predominant follicles and luteal tissue is preferred for biopsy. The goal is to maximize the number of primordial follicles per specimen without compromising vascular supply to the remaining ovarian tissue. In order to optimise results, it is best to remove the ovarian cortex from the medulla, which helps extreme penetration of cryoprotectants into the cortical tissue (14). Normally, extraction of a single block of cortical tissue through partial excision technique can be performed in either one or both ovaries (11). 2. Histological examination. Ovarian fragments are immediately observed by a pathologist to simultaneously confirm the presence of follicles and the absence of a massive cancer infiltration (16). 3. Cryopreservation. Ovarian tissue can be cryopreserved independently of the menstrual phase, which represents no delay for the oncological therapy. Centres offering OTC allow for the procedure to be performed even the first day after the patient's first visit (17), and it can be carried out using one of two established techniques: slow freezing or rapid freezing (vitrification) (Fig. 3). The slow freezing method has already resulted in dozens of live births worldwide, whereas vitrified tissue has only led to a few reported live births to date (18, 19). The well-established standard method for human OTC is slow equilibrium freezing. It begins with exposing cells to low concentrations of one of the cryoprotective agents (CPAs) (propanediol, dimethyl sulfoxide or ethylene glycol) in combination with human serum albumin, and temperature must be slowly decreased. In contrast, vitrification requires higher concentrations of CPAs, which reduces the risk of ice nucleation, and it is significantly faster. When the solution is rapidly cooled, the tissue turns into a glassy, vitrified state, avoiding extracellular and intracellular ice crystallization (19). You can read our previous article to know more about these techniques here. The current main problem of ovarian banking is to restore ovarian function and fertility using frozen/thawed tissue with both high efficiency and minimal risks (16). 4. Biopsies are stored in the vapour phase of liquid nitrogen at -176 °C, thus allowing for long-term storage of potentially large numbers of primordial follicles (15). 5. Re-implantation. The stored ovarian tissue can be re-implanted orthotopically (into the remaining ovary, ovarian fossa, or broad ligament), heterotopically (into the subcutaneous space of the forearm, subcutaneous tissue of the abdomen, anterior wall of the abdomen, just beneath the peritoneum, or in the rectus muscle), or as recent studies have reported, xenografted or matured entirely in vitro (15, 19). Orthotopic transplantation may provide the possibility to achieve natural pregnancy; however, it requires abdominal surgery with general anaesthesia. In contrast, heterotopic is advantageous in cases of severe pelvic adhesions, distorted pelvic anatomy, and poor pelvic vasculature due to previous irradiation. Furthermore, it implies the possibility of creating long-term ovarian endocrine function with a less invasive surgical approach for transplantation, which does not require general anaesthesia, and thereby creating a more cost-effective option. However, this option may produce oocytes (and therefore, embryos) with reduced quality, as compared to orthotopic transplantation sites. This outcome is likely related to the suboptimal environment of heterotopic sites in regard to local factors. These include: external pressure, temperature, vascularization (decreased blood supply), oxygen tension and reduced paracrine factors, which could affect the possibility of sustaining the growth and development of normal follicles and oocytes (16). Thawed ovarian fragments do not need vascular anastomosis when transplanted into a well-vascularized tissue. Genes for angiogenesis factors seem to be upregulated in the ovary, compared to other tissues (16). However, there is a significant risk of the tissue suffering from hypoxia until neovascularization starts, normally following the first 48 hours after the procedure. Survival of primordial follicles (around 90% after thawing) ranges between 5 and 50% after grafting, the most crucial factor being the degree of ischaemic injury after transplantation (16). Although restoration of ovarian function has been reported for both approaches, live births following bilateral oophorectomy have only been documented from orthotopic transplantations (19). CASE REPORTS Successful studies in experimental animals with live births following transplantation of cryostored ovarian tissue have been previously reported in rodents and sheep (20, 21, 22, 23). Ovarian tissue cryopreservation was first described by Hovatta et al in 1996 (6). The first human ovarian transplantation with cryopreserved ovarian tissue was later performed by Oktay in 2000 (24). Donnez and colleagues reported the first human live birth from orthotropic transplantation of frozen human ovarian tissue in 2004 (25), with another successful live birth achieved by Meirow in 2005 (26). Live births following cryopreservation of pre-pubertal ovarian tissue had not been described until the 2015 case report by Demeestere et al (27). During all these years, authors have been discussing about whether vitrification is better than slow freezing. Some of them found apoptotic cells in vitrified ovarian tissue, meanwhile others showed higher primordial follicle density using the slow freezing method. Sanfilippo et al found no significant differences between both techniques (28). The American Society of Clinical Oncology advises that OTC for fertility preservation is still considered an experimental technique (9). Consequently, it should only be performed in centres with the appropriate expertise. This means performance under the Institutional Review Board (IRB) approved protocols, which include follow-up for recurrent cancer (29). However, in some clinical situations it remains the only available option. Here we describe some reported cases in different scenarios: fertility wishes and puberty induction, autotransplantation and allotransplantation. Fertility preservation OTC is the only available option for fertility preservation before menarche. Demesteree et al (2015) reported a spontaneous pregnancy case of a woman who had undergone ovarian tissue transplantation shortly before turning 14 (27). She had been diagnosed with sickle-cell anaemia at the age of 5 and treated with hematopoietic stem cell transplantation at 11. Her parents and herself were offered OTC in order to preserve fertility. The patient started puberty at the age of 10 (breast development) and the procedure was performed when she was 13 years and 11 months. At the age of 25 she interrupted hormonal supplementation and underwent ovarian tissue transplantation. Two years later she got pregnant and gave birth to a healthy boy in November 2014. Andersen 2012 et al described a case of ovarian tissue allotransplantation between identical twins at the age of 38 (30). One of them had undergone menopause at the age of 22, whereas the other had already had 2 children. The woman who was transferred the ovarian tissue had three natural pregnancies: eight months after transplantation, at age 42 and at 45. She gave birth to a healthy girl and two healthy boys. Puberty induction A.K. Jensen et al recommend OTC to all young girls who present a high risk of developing ovarian insufficiency and/or infertility following high-dose chemotherapy and/or irradiation (31). The ovary serves a double function: folliculogenesis and production of sex hormones, which play a main role in the woman’s body such as the development of secondary sexual characteristics. Ernst et al (2013) described a case of puberty induction in a girl treated with chemo- and radiotherapy at the age of 9 (32). Due to the treatment, her ovary had been damaged, leading to a pre-pubertal stage with postmenopausal levels of FSH. Four and a half years after OTC, tissue was grafted and she recovered normal levels of FSH and oestradiol, which let the ovaries reach the pubertal stage. Clinical effectiveness and outcomes Since the lifespan of a piece of transferred ovary varies from a few to several years (33), hormone levels can turn to those corresponding to a pre-pubertal stage some months after transplantation. The effectiveness of this technique also depends on the woman’s own ovarian reserve, which is linked to the age at which her ovary tissue was cryopreserved. In all cases reported in which pregnancy has been achieved, patients had only cryopreserved tissue of one of their ovaries, whereas the other one had been kept during the chemotherapy treatment. Thus, it has not been possible to determine whether pregnancy was a product of the cryopreserved and transferred tissue or to the remaining intact ovary. On the other hand, cases have been described in which infertility due to hormone failure could be restored by ovarian tissue allotransplantation, thus leading to healthy pregnancies (34). Even though numerous successful births have been reported after OTC, re-implantation of malignant cells (potentially causing metastatic processes), along with grafted ovarian tissue in a subject previously treated for the same cancer, still remains a serious concern. CONCLUSIONS - It is well known that treatments that expose patients to gonadotoxic therapies such as alkylating agents, pelvic irradiation, and/or stem cell transplant could increase the risk for post-therapy infertility, because the ovaries are very sensitive to these cytotoxic drugs. - Although currently OTC still remains an experimental technique, there has been an increasing amount of successfully case reports since 1996. - OTC remains an alternative and the only suitable option to preserve fertility in some special cases such as young female cancer patients. - It is necessary to make efforts in order to develop the best protocol to preserve ovarian tissue, as well as to study some unclear aspects such as the possibility of reintroducing the original disease (no cases reported, through), or to try to improve the survival of primordial follicles after grafting. - Finally, it would be recommended to follow up individuals from live births after OTC in order to analyze any abnormal findings. REFERENCES
1- Roy T, Bradley C, Bowman M et al. Single-embryo transfer of vitrified-warmed blastocysts yields equivalent live-birth rates and improved neonatal outcomes compared with fresh transfers. Fertil Steril 2014;101:1294-301 2- Barnhart K. Are we ready to eliminate the transfer of fresh embryos in in vitro fertilization? Fertil Steril 2014;102:1-2 3- Roque M, Lattes K, Serra S, et al. Fresh embryo transfers versus frozen embryo transfer in in vitro fertilization cycles: A systematic review and meta-analysis. Fertil Steril 2013;99:156-162. 4- Silber S. Foreword. In: Michael J. Tucker and Juergen Liebermann. Vitrification in Assisted Reproduction.Second ed. Boca Raton FL:CRC Press; 2016 5- Chatziparasidou A, Martine N, Moisidou M, Chara O, Loakeimidou C, Pappas C, Christoforidis N. Accumulation of oocytes and/or embryos by vitrification: a new strategy for managing poor responder patients undergoing pre implantation diagnosis. F1000Research 2014,2:240. Last updated 03 JUL 2014. 6- Hovatta O, Silye R, Krausz T et al. Cryopreservation of human ovarian tissue using dimethylsulphoxide and propanediol-sucrose as cryoprotectants. Hum Reprod 1996;11:1268-72. 7- Newton H, Aubard Y, Rutherford A, Sharma V, Gosden R. Low temperature storage and grafting of human ovarian tissue. Hum Reprod 1996;11:1487-91. 8- The Ethics Committee of American Society for Reproductive Medicine. Fertility preservation and reproduction in patients facing gonadotoxic therapies: a committee opinion. Fertil Steril 2013;100:1224-12315. 9- Practice Committee of American Society for Reproductive M. Fertility preservation in patients undergoing gonadotoxic therapy or gonadectomy: a committee opinion. Fertil Steril. 2013;100(5):1214e1223. 10- Medicine PCotASfR, Technology PCotSfAR. Ovarian tissue and oocyte cryopreservation. Fertil Steril. 2006;86(5 Suppl 1):S142-7. 11- Corkum KS, Laronda MM, Rowell EE. A review of reported surgical techniques in fertility preservation for prepubertal and adolescent females facing a fertility threatening diagnosis or treatment. Am J Surg. 2017. [Epub ahead of print] 12- Xiao Z, Zhang Y, Fan W. Cryopreservation of human ovarian tissue using the silver closed vitrification system. J Assist Reprod Genet. 2017. [Epub ahead of print] 13- Silber S. Ovarian tissue cryopreservation and transplantation: scientific implications. J Assist Reprod Genet. 2016;33(12):1595-603. 14- Fathi R, Rezazadeh Valojerdi M, Ebrahimi B, Eivazkhani F, Akbarpour M, Tahaei LS, et al. Fertility Preservation in Cancer Patients: In Vivo and In Vitro Options. Cell J. 2017;19(2):173-83. 15- Anderson RA, Wallace WH, Baird DT. Ovarian cryopreservation for fertility preservation: indications and outcomes. Reproduction. 2008;136(6):681-9. 16- Moffa F, Biacchiardi CP, Fagioli F, Biasin E, Revelli A, Massobrio M, et al. Ovarian tissue cryostorage and grafting: an option to preserve fertility in pediatric patients with malignancies. Pediatr Hematol Oncol. 2007;24(1):29-44. 17- Lotz L, Maktabi A, Hoffmann I, Findeklee S, Beckmann MW, Dittrich R. Ovarian tissue cryopreservation and retransplantation--what do patients think about it? Reprod Biomed Online. 2016;32(4):394-400. 18- Herraiz S, Novella-Maestre E, Rodríguez B, Díaz C, Sánchez-Serrano M, Mirabet V, et al. Improving ovarian tissue cryopreservation for oncologic patients: slow freezing versus vitrification, effect of different procedures and devices. Fertil Steril. 2014;101(3):775-84. 19- Ladanyi C, Mor A, Christianson MS, Dhillon N, Segars JH. Recent advances in the field of ovarian tissue cryopreservation and opportunities for research. J Assist Reprod Genet. 2017;34(6):709-22. 20- Deanesly R. Inmature rat ovaries grafted after freezing and thawing. J Endocrinol 1954;11:197-200. 21- Green SH, Smith AU, Zuckerman S. The number of oocytes in ovarian autografts after freezing and thawing. J Endocrinol 1956;13:330-4. 22- Carroll J, Gosden RG,. Transplantation of frozen thawed mouse primordial follicles. Human Reprod 1993;8:1163-7. 23- Gosden RG, Baird DT, Wade JC, Webb R. Restoration of fertility to oophorectomized sheep by ovarian autografts stored at -196ºC. Human Reprod 1994;9:597-603. 24- Oktay K, Karlikaya G. Ovarian function after transplantation of frozen, banked autologous ovarian tissue. N Engl J Med. 2000;342(25):1919. 25- Donnez J, Dolmans MM, Demylle D, Jadoul P, Pirard C, Squifflet J, et al. Livebirth after orthotopic transplantation of cryopreserved ovarian tissue. Lancet. 2004; 364 (9443): 1405-10. 26- Meirow D, Levron J, Eldar-Geva T, Hardan I, Fridman E, Zalel Y, et al. Pregnancy after transplantation of cryopreserved ovarian tissue in a patient with ovarian failure after chemotherapy. N Engl J Med. 2005;353(3):318-21. 27- Demeestere I, Simon P, Dedeken L, Moffa F, Tsépélidis S, Brachet C, et al. Live birth after autograft of ovarian tissue cryopreserved during childhood. Hum Reprod. 2015;30(9):2107-9. 28- Sanfilippo S, Canis M, Smitz J, Sion B, Darcha C, Janny L, et al. Vitrification of human ovarian tissue: a practical and relevant alternative to slow freezing. Reprod Biol Endocrinol [Internet]. Reproductive Biology and Endocrinology; 2015;13(1):67. 29- Loren AW, Mangu PB, Beck LN, Brennan L, Magdalinski AJ, Partridge AH, et al. Fertility preservation for patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2013;31(19):2500–10. 30- Andersen CY, Silber SJ, Berghold SH, Jorgensen JS, Ernst E. Long-term duration of function of ovarian tissue transplants: Case reports. Reprod Biomed Online [Internet]. Reproductive Healthcare Ltd.; 2012;25(2):128–32. 31- Jensen AK, Rechnitzer C, Macklon KT, Ifversen MRS, Birkebaek N, Clausen N, et al. Cryopreservation of ovarian tissue for fertility preservation in a large cohort of young girls: focus on pubertal development. Hum Reprod. 2016;32(1):154–64. 32- Ernst E, Kjærsgaard M, Birkebæk NH, Clausen N, Andersen CY. Case report: Stimulation of puberty in a girl with chemo- and radiation therapy induced ovarian failure by transplantation of a small part of her frozen/thawed ovarian tissue. Eur J Cancer. 2013;49(4):911–4. 33- Ernst E, Bergholdt S, Jørgensen JS, Andersen CY. The first woman to give birth to two children following transplantation of frozen/thawed ovarian tissue. Hum Reprod. 2010;25(5):1280–1. 34- Silber SJ, Derosa M, Pineda J, Lenahan K, Grenia D, Gorman K, et al. A series of monozygotic twins discordant for ovarian failure: Ovary transplantation (cortical versus microvascular) and cryopreservation. Hum Reprod. 2008;23(7):1531–7.
0 Comments
Leave a Reply. |
Entries
March 2019
Categories
All
 2016-2019. All Rights Reserved by Embryologist Media. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License . |
Embryologist Media